Marangoni bursting takes place in alcohol-water droplets; as the alcohol evaporates, surface tension changes across the liquid surface, generating a flow that tears the original drop into smaller droplets. Here researchers add a twist to the experiment using PMMA, an additive that dissolves well in alcohol but poorly in water. As the alcohol evaporates, the PMMA precipitates back out of the water-rich droplet, forming yarn-like strands. (Image and video credit: C. Seyfert and A. Marin)
Tag: instability

Laser-Induced Jet Break-Up
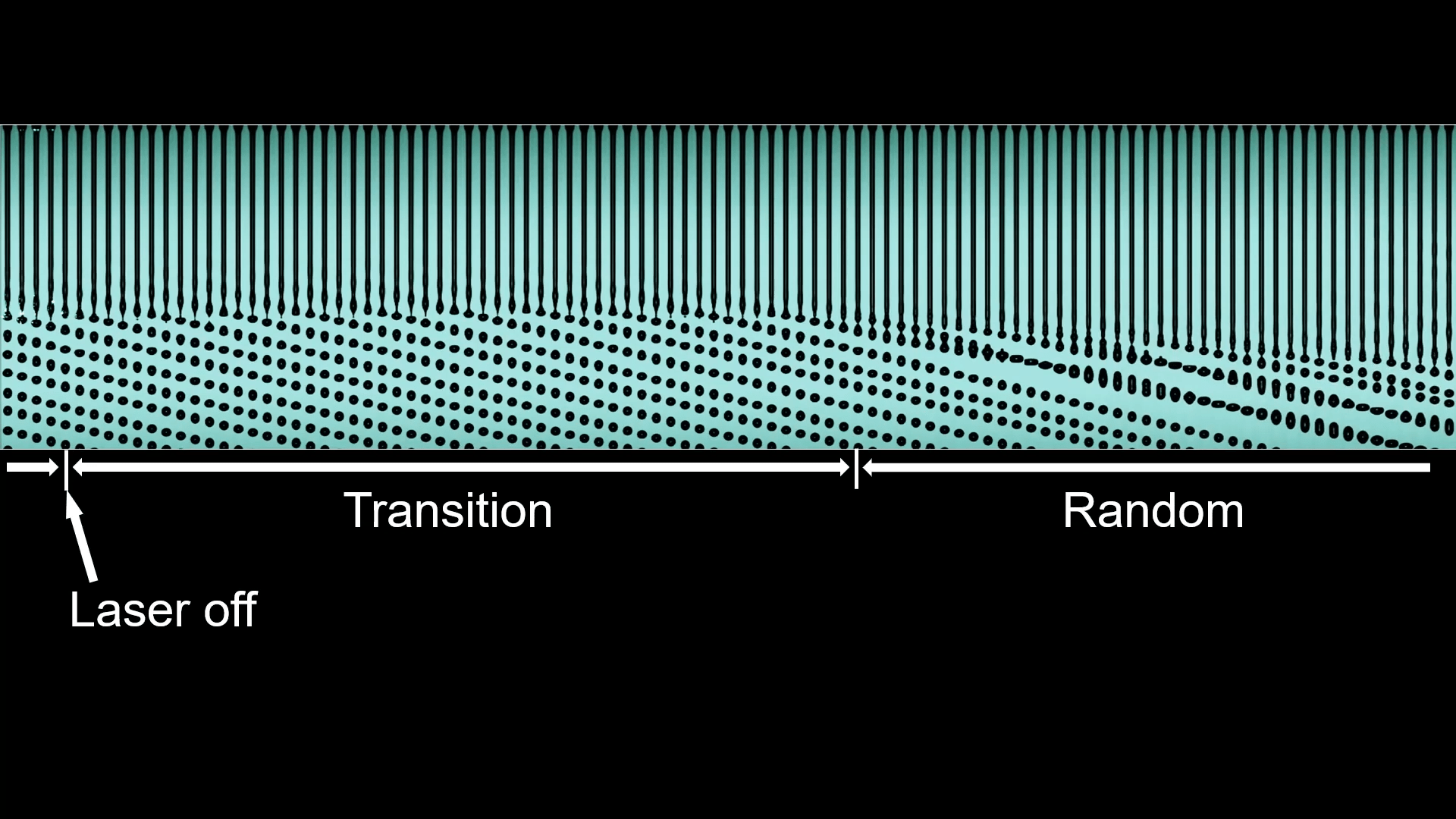
A falling stream of water will naturally break up into droplets via the Plateau-Rayleigh instability. Those droplets are random, unless something like vibration of the nozzle sets their size. In this study, though, researchers found that shining a laser beam on the stream can trigger an orderly break-up with droplets that are consistent in size and spacing.
The optofluidic phenomenon depends on a few different effects. The changing curvature of the liquid stream reflects the laser light, some of which undergoes total internal reflection and travels up the jet as if it were a fiber optic cable. Look closely in the right side of the second image, and you’ll see a periodic flicker of green light at the mouth of the nozzle. Those flashes of green reveal that the liquid jet is guiding the light upstream in bursts, each of which exerts an optical pressure that triggers the Plateau-Rayleigh instability.
When the laser first turns on, there’s a transition period before the orderly break-up begins, and, likewise, turning the laser off triggers a transition from orderly to random (top image). (Image and research credit: H. Liu et al.; via APS Physics; submitted by Kam-Yung Soh)

Where Wind Meets Water
That the wind causes ocean waves is obvious to anyone who has spent time near the water, but the details of that process remain fuzzy. Many of the explanations — like the Kelvin-Helmholtz instability — only explain part of the process, usually the beginning when the waves are very small. As the waves get larger, they affect the wind in turn, complicating matters.
As messy as the theory gets, our ability to measure the wind and water in situ is limited, too. Just look at this wild research platform oceanographers designed to study wind and waves. It’s part of a 355-ft vessel that’s towed out to sea horizontally and then flipped so that 300 feet of it remain underwater to stabilize the remainder for measurements. Even with equipment like this, measuring the turbulent air and water near the ocean-sky interface is incredibly difficult.
This review article gives a nice overview of different historical efforts to explain how wind makes waves and provides a snapshot of the latest research in the area. (Image credit: R. Bilcliff; see also N. Pizzo et al.)

How To: Ink Flow Photography
Ever wanted to try your hand at making these cool billowing ink photos? Photographer Jason Parnell-Brookes has a detailed tutorial over at PetaPixel laying out the necessary tools and set-up. I haven’t tried this out myself, but I hope to! How about you? (Image credit: J. Parnell-Brookes; submitted by clogwog)

Breaking Compound Ligaments
When pulled, viscous liquids stretch into ligaments that thin and then break into droplets. In this video, researchers investigate how these ligaments break up, depending on their composition. The initial views show the break-up of a water-glycerol ligament (Image 1) and an oil ligament (Image 2). By placing a water droplet inside oil, the researchers got quite different results, including oil-encapsulated droplets (Image 3). The technique could be useful for making compound droplets, even with more than two components. (Image and video credit: V. Thiévenaz and A. Sauret)

The Bubbly Escape
Sometimes experiments don’t work as planned and, instead of answers, they lead to more questions. In this video, we see an experiment looking at an air bubble trapped beneath a cone. It’s the same situation you get by holding a mug upside-down in a sink full of water but with inclined walls. As the cone moves downward, it squeezes the trapped air bubble. A film of air gets pushed along the walls of the cone, eventually forming finger-like bubbles that wrap around the edge of the cone and get entrained into the vortex ring outside the cone.
Clearly, there is some kind of instability that drives the air bubble to form these fingers rather than spreading uniformly. But the big question is which one? Is this a density-driven Rayleigh-Taylor instability caused by air getting pushed into water? Or is it a Saffman-Taylor instability causes by the less viscous air forcing its way into the more viscous water? What do you think? (Image and submission credit: U. Jain)


Falling Pancake Drops
Despite their round appearance, the droplets you see here are actually shaped like little pancakes. They’re sandwiched inside a Hele-Shaw cell, essentially two plates with a viscous fluid between them. As these droplets fall through the cell, some remain steady and rounded (Image 1), while others experience instabilities (Images 2 and 3). By varying the ratio of the ambient fluid’s viscosity relative to the drop, the authors found two different kinds of breakup. In the first type (Image 2), droplet breakup occurred due to perturbations inside the drop itself. In the second type (Image 3), the viscosity of the ambient fluid is closer to that of the drop and intrusions of the ambient fluid into the drop break it apart. (Image and research credit: C. Toupoint et al.)

Paint Spinning
In a return to their roots, this Slow Mo Guys video features paint flowing on (and off!) a spinning disk. To help us see what’s going on, Gav uses a trick that’s familiar to many fluid dynamicists: he rotates the high-speed footage at the same speed that the disk rotates. This transformation places the viewer into a reference frame where the disk appears stationary, so that small changes in the flow are apparent.
It makes for a gorgeous view as centrifugal force flings the paint outward and eventually breaks it into drops. The rotation speed is unfortunately so high that the spinning completely dominates all other forces. The few runs with more viscous acrylic paint show some hints of more interesting behaviors that might be visible with a slower rotation rate (which would make the tug of war between inertia/viscosity/surface tension and centrifugal force less one-sided). Anyone got a high-speed camera, some speed control, and a willingness to get messy? (Image and video credit: The Slow Mo Guys)

Solid, Liquid, Both?
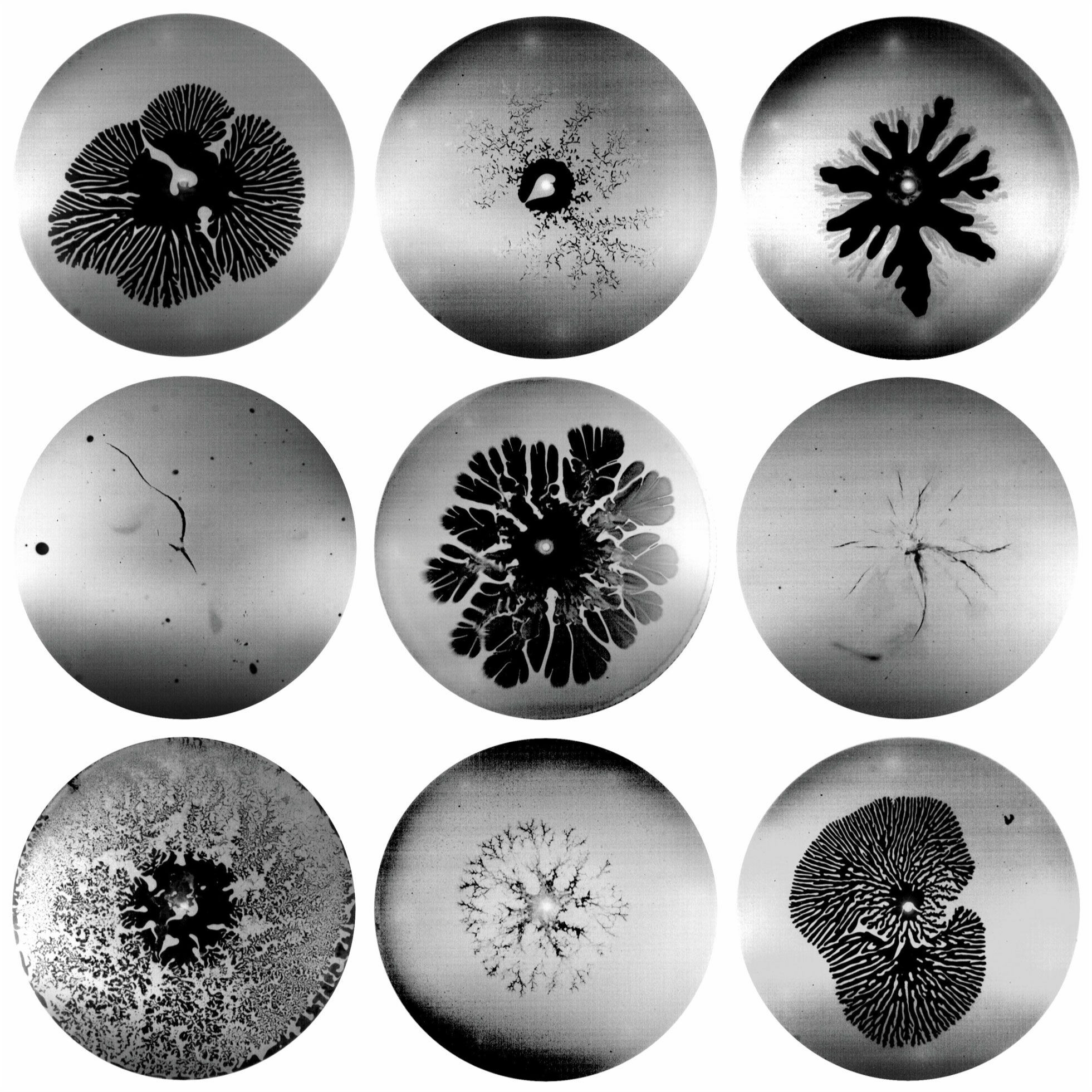
Materials like oobleck — a suspension of cornstarch particles in water — are tough to classify. In some circumstances, they behave like a fluid, but in others, they act like a solid. Here researchers sandwiched a thin layer of oobleck between glass plates and injected air into the mixture. For a fluid, this setup creates a classic Saffman-Taylor instability where rounded fingers of air push their way into the more viscous fluid. And, indeed, for low air pressures and low concentrations of cornstarch, the oobleck forms these viscous fingers. You can see examples in the top row’s first and third image, the second row’s middle image, and the bottom row’s third image.
Injecting air at high pressures and high cornstarch concentrations fractures the oobleck like a solid (middle row, first and third images). At intermediate pressures and concentrations, the oobleck forms a pattern called dendritic fracturing, where new branches can grow perpendicularly to their parent branch. Examples of this pattern are in the top row’s second image and the bottom row’s first and second images. (Image and research credit: D. Ozturk et al.; via Physics Today)

Superfluid Instabilities
Superfluids — like Bose-Einstein condensates — are bizarre compared to fluids from our everyday experience because they have no viscosity. Without viscosity, it’s no surprise that they behave in unusual ways. Here, researchers simulated superfluids moving past one another. In each of these images, the blue fluid is moving to the left, and the red fluid is moving to the right. In a typical fluid, such motion causes ocean-wave-like curls due to the Kelvin-Helmholtz instability.
Instead, with a low relative velocity and high repulsion between atoms of the two layers, the superfluids form a tilted, finger-like interface (Image 1) that the authors refer to as a flutter-finger pattern. (Repulsion essentially sets the miscibility between the superfluids. With a high repulsion, the superfluids resist mixing.)
With a higher relative velocity (Image 2), the wavelength of the ripples becomes comparable to the thickness of the interface, and the superfluids take on a very different, zipper-like pattern. Note how the tips detach and reconnect to the neighboring finger!
With lower repulsion, the interface between the two liquids is thicker and breaks down quickly (Image 3). The authors call this a sealskin pattern. (Image credits: water – M. Blažević, simulations – H. Kokubo et al.; research credit: H. Kokubo et al.; via APS Physics)