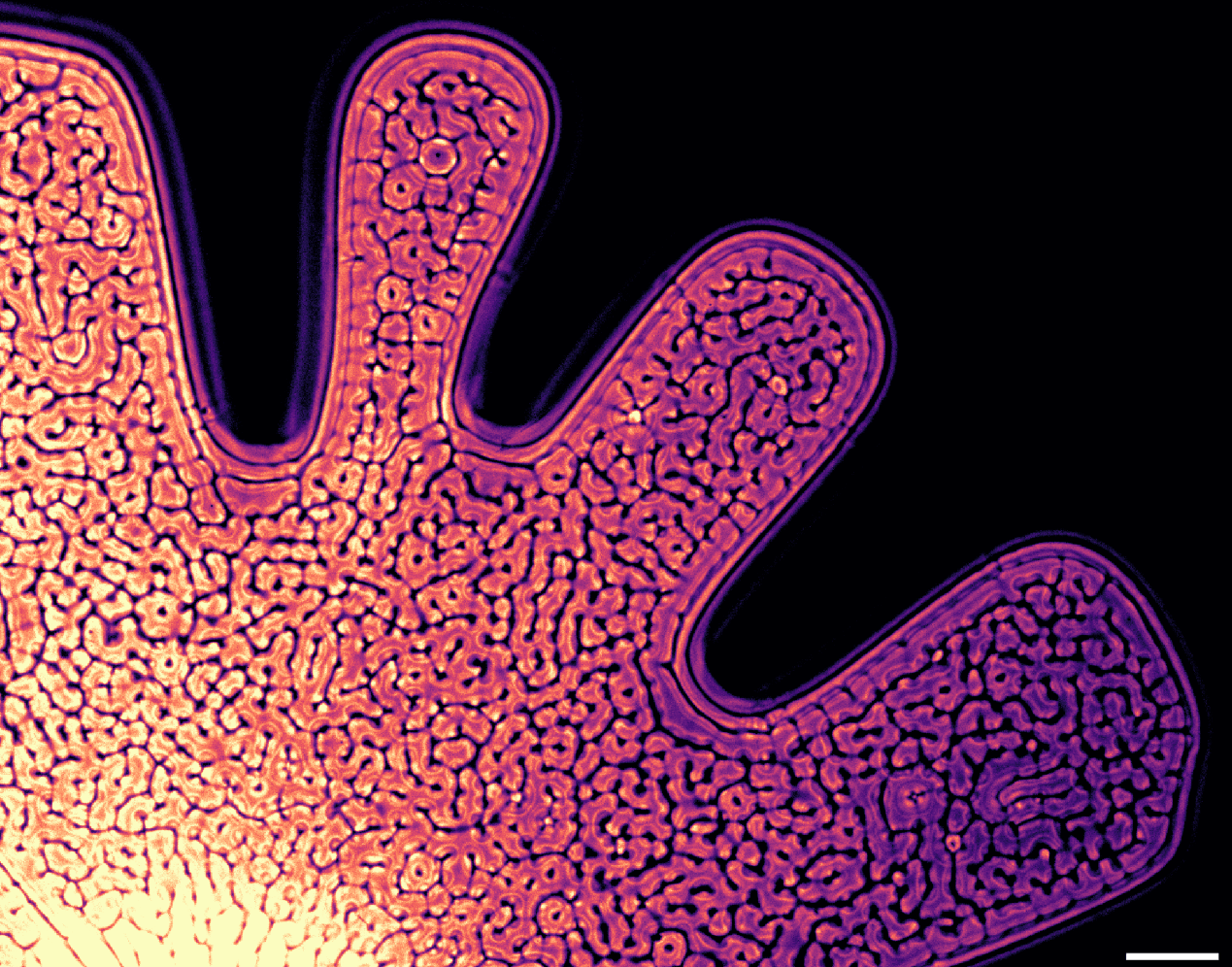
The world of fluid instabilities is a rich one. Combine fluids with differing viscosities, densities, or flow speeds and they’ll often break down in picturesque and predictable manners. Here, researchers explore the Rayleigh-Taylor instability (RTI), which occurs when a denser fluid sits above a less dense one (in a gravitational field). It’s an extremely common instability, showing up in both the cream in your ice coffee and the shape of a supernova’s explosion. It’s very difficult to set up and observe, though, which is where the real cleverness of this experiment stands out.
To study the RTI, these researchers first created another instability, the Saffman-Taylor instability. They filled the space between two glass plates with a viscous fluid, then injected a less viscous one. That created the distinctive viscous fingering pattern seen in the top image. In addition to being less viscous, the injected fluid was also less dense. As it pushed into the original fluid, it displaced some of it, creating a three-layer structure with dense fluid over less-dense fluid over dense fluid. That laid the groundwork for the Rayleigh-Taylor instability form.

Check out the cell-like pattern distributed across the fluid in the top image. These are plumes formed in the RTI as dense fluid sinks into the less-dense fluid below it. From the side (see second image), each plume takes on the distinctive mushroom-like shape of a Rayleigh-Taylor instability. Given time, the two fluids mix and the cellular pattern disappears. But until then, this set-up uses one instability to study a second one. How cool is that?! (Image and research credit: S. Alqatari et al., see also)