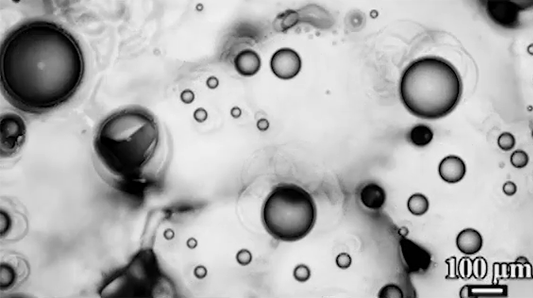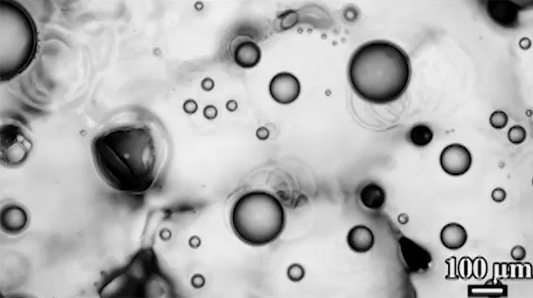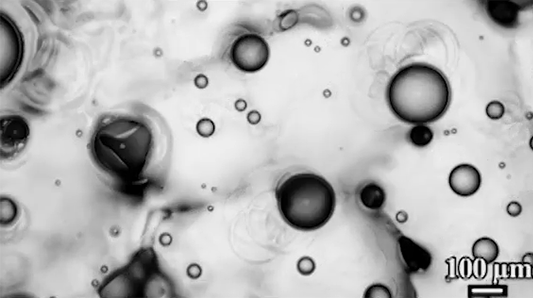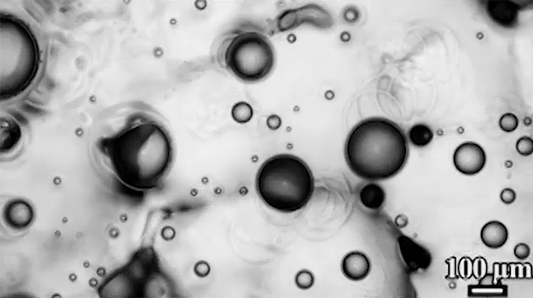
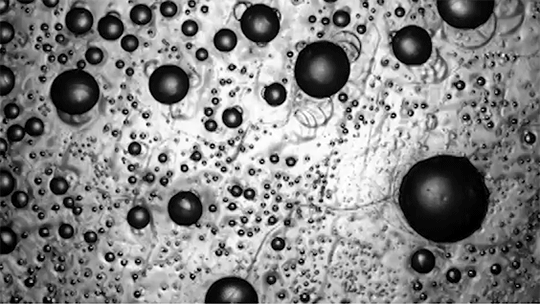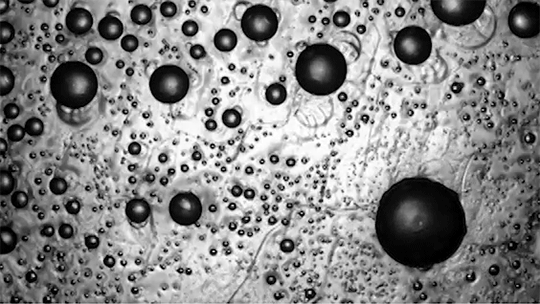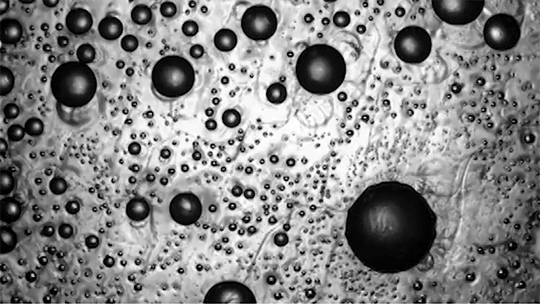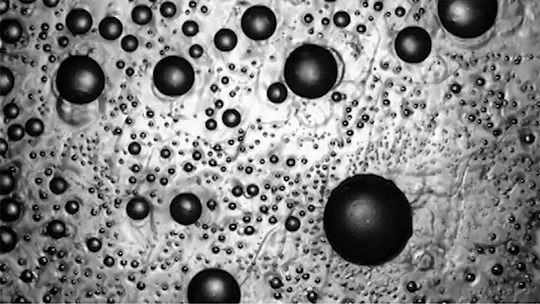
For a droplet to bounce, we expect it to hit a wall or a sharp interface of some kind. But in a new study, researchers demonstrate a droplet that bounces with neither. Shown above is an oil droplet sinking through a stratified mixture of ethanol (toward the top) and water (toward the bottom). Because the oil is heavier than ethanol, it initially sinks, dragging some of the ethanol with it as it falls. Over time, some of that ethanol rises again, forming what’s known as a buoyant jet.
Simultaneously, the gradient of ethanol to water between the top and bottom of the drop creates an imbalance in surface tension. The ethanol near the top of the drop has a lower surface tension than the water at the bottom. This creates a downward Marangoni flow along the drop interface.
The bounce itself happens quickly after a long, slow sinking period. As the drop’s sinking slows, the buoyant jet weakens until it disappears completely. At the same time, the downward Marangoni flow pulls fresh ethanol-rich fluid toward the top of the drop. That increases the surface tension difference and strengthens the Marangoni flow, creating a positive feedback loop. In less than a second, the Marangoni flow increases by two orders of magnitude, pulling so hard that the drop shoots upward.
That resets the cycle by weakening the Marangoni flow and strengthening the buoyant jet. The droplet can continue bouncing for about 30 minutes until the concentration gradient is so well-mixed that the cycle can’t continue. (Image and research credit: Y. Li et al.; via APS Physics; submitted by Kam-Yung Soh)