Artist Kamiel Rongen uses common substances like paint, oil, eggs, and even air freshener to create what he calls “water ballet.” His videos are full of ethereal and surreal landscapes full of color and motion. Buoyancy (or the lack thereof) plays a major role in his work – fluids often spurt upward like alien creatures emerging from a chrysalis. I’ve been debating with myself whether the fluids are actually rising or if they’re falling in front of an upside-down camera, and I’m not completely certain either way! I think that’s a testament both to Rongen’s artistry and to the awesome physics involved. Check out the full video below and you can see many more examples of Rongen’s work on his website. (Image and video credit: K. Rongen; h/t to James H.)
Tag: buoyancy

Impressionist Foams
Imagine taking two panes of glass and setting them in a frame with a small gap between them. Then partially fill the gap with a mixture of dye, glycerol, water, and soap. After turning the frame over several times, the half of the frame will be filled with foamy bubbles. When you flip it again, the dyed glycerol-water will sink and penetrate the bubble layer, creating complex and beautiful patterns as it mixes. Some of the bubbles may get squeezed together until they coalesce into larger bubbles that shoot upward thanks to their increased buoyancy. Other smaller bubbles will wend their way upward as neighboring fluid shifts. If you examine the tracks left by individual bubbles, you can find patterns reminiscent of Impressionist paintings, as seen at the end of this Gallery of Fluid Motion video. (Image credit: A. Al Brahim et al., source)

Riding Across Water
Humans may not be fast enough to run across water, but we’ve found other ways to conquer the waves. It’s even possible (though definitely not recommended) to ride across stretches of water on a dirt bike. To do so, you have to keep the bike (hydro)planing, and to understand what that means, let’s take a moment to talk about boats.
At low speeds, boats stay afloat based on buoyancy, a force that depends on how much water they displace. But when moving at high speeds, modern speedboats lift mostly out of the water and skim the surface instead. At this point, it’s hydrodynamic lift that keeps the boat above the surface and we say that the boat is planing. Calculating that hydrodynamic lift is fairly complicated and depends on many factors – for those who are interested, check out some of David Savitsky’s papers – but, generally speaking, going faster gives you more lift.
This brings us back to the dirt bike. There’s nothing particularly hydrodynamic about a dirt bike. It’s not shaped to provide hydrodynamic lift, but it does come with a high power-to-weight ratio. It’s this ability to create pure speed, and a rider’s keen sense for holding the bike at the right angle, that enables pros to cross open water. Needless to say, this is the kind of stunt that could end really badly, so don’t try it yourself. (Image credits: C. Alessandrelli, source; EnduroTripster, source; via Digg; submitted by 1307phaezr)

Layered Latte Physics
Latte lovers may be familiar with the layered latte, a beverage with distinctive horizontal layers mixing espresso and milk, but you may not have taken the time to wonder how these layers form. Like many layering phenomena in our oceans, the layered latte is the result of double-diffusive convection. This means that there are two variables that both affect density in the fluid mixture and that they act at different rates.
In the latte, those factors are 1) the different densities of the milk and espresso and 2) density changes caused as the latte cools to room temperature. A layered latte forms when the lighter espresso is poured into denser milk. If it’s poured quickly enough, the momentum of the pour forces some of the espresso down into the milk, despite the buoyant force that tries to keep the espresso on top. So that initial pour sets up a density gradient that runs from pure espresso at the top to pure milk on the bottom, with varying mixtures of the two in between.
The distinct layers won’t form until the latte begins cooling off. Along the walls of the container, heat is lost more quickly, causing fluid to cool and start sinking. But a specific bit of fluid can only sink until the fluid surrounding it is the same density. That can carry a cooler bit of latte to the bottom of a layer, but not into the denser layer below. At this point, our bit of latte moves inward, starts to warm up, and circulates up through the center of its layer. As when it sank, the fluid can only move up until it encounters a layer with equal or lesser density, at which point it must move horizontally instead. This thermal convection, combined with the density gradient formed by the initial pour, sets up the distinctive layers of the latte. The layers are quite stable – neither gentle stirring nor taking a sip will disrupt them for long – provided the drink remains warmer than the surrounding air. (Image credits: kopeattugu/Instagram, N. Xue et al.; research credit: N. Xue et al.; via NYTimes; submitted by Kam-Yung Soh)

The Cheerios Effect
You’ve probably noticed that cereal clumps together in your breakfast bowl, but you may not have given much thought as to why. This tendency for objects at an interface to attract is known as the Cheerios effect, although it happens in more than just cereal, as Joe Hanson from It’s Okay to Be Smart explains. The effect is a combination of buoyancy, gravity, and surface tension acting in concert.
When air, a liquid, and a solid meet, they form a meniscus, the curvature of which depends on characteristics of their interaction. Light, buoyant cereal and the walls of your bowl both have upward-curving menisci. Denser objects, like the tacks shown below, stay at the surface only because surface tension holds them up. Their meniscus curves downward.
Objects with a similar meniscus curvature will attract. For cereal approaching a wall, the light Cheerio is buoyant enough that there’s an upward force on it, but it’s constrained to stay at the interface. It cannot rise, but that buoyancy is enough to let it climb the meniscus at the wall. The two tacks attract one another for similar reasons, except this time their weight helps them fall into one another. Check out the full video to see more examples of this effect in nature! (Video and image credit: It’s Okay to Be Smart; research credit: D. Vella and L. Mahadevan, pdf)



Flames in Freefall
Gravity is such an omnipresent force in our lives that we frequently forget how strongly it affects our daily experiences and how differently nature behaves without it. A wonderful example of this is the simple flame of a candle. On Earth, a candle flame is tear-drop-shaped and elongated, burning hotter near the bottom and glowing yellow from soot at the top. But, as Dianna demonstrates with her free-fall experiment, this shape is due entirely to the effects of gravity. Buoyant forces make the hot air near the candle rise, pulling in cooler air and fresh oxygen at the base while stretching out the flame. In microgravity – or free-fall – flames are instead spherical, their shape driven by molecular and chemical diffusion. Check out the full video to see more effects of acceleration on flames. (Video credit: Physics Girl)

Soaring Pelicans
Earlier this summer, I looked up on a bright, sunny day and saw a quartet of black and white figures soaring overhead. Initially, I thought it might be a formation of kites or unmanned aerial vehicles (UAVs) because I saw no flapping as the group wheeled about. With the help of the Cornell Lab of Ornithology’s awesome Merlin app, I was able to identify the soarers as American white pelicans – not a species I’d expected to find flying along the Front Range of the Rocky Mountains! (Turns out, they breed on lakes around here.)
The reason I saw so little flapping is that the birds were riding thermals. As the sun heats the ground, air near the surface warms up and begins to rise due to its buoyancy. Pelicans interested in flying between breeding and foraging grounds will start testing the thermals early in the day, as soon as they begin to form. As the heating continues, the intensity of thermals strengthens and they extend higher into the atmosphere. This is where the birds can really excel at using atmospheric energy for their flight. Pelicans will circle within a thermal until they reach roughly the middle of its height. Then they will glide, gradually losing altitude until they reach another thermal where they can climb without expending their own energy. With a 2.7 meter wingspan and a relatively low drag coefficient, the pelicans can glide and soar remarkably well. Researchers have even suggested using them as a sort of biological UAV for studying atmospheric dynamics! (Image credits: D. Henise, M. Stratmoen; research credit: H. Shannon et al., pdfs – 1, 2)

Tightrope Walkair
A bubble rising through water can get caught on an aerophilic (air-attracting) fiber. The bubble will then adhere to the fiber and be guided to the surface by it. In the poster above, the image is a composite photo of such a bubble every 40 milliseconds. Once captured by the fiber, the bubble first accelerates and then reaches a terminal velocity, indicated by the equal spacing of the bubble photos toward the right end of the picture. The terminal velocity strikes a balance between buoyancy, which pulls the bubble upward, and skin friction between the bubble and the water, which acts like drag on the bubble. At the terminal velocity, these forces are equal; neither is able to speed up or slow down the bubble. (Image credit: H. de Maleprade et al.)

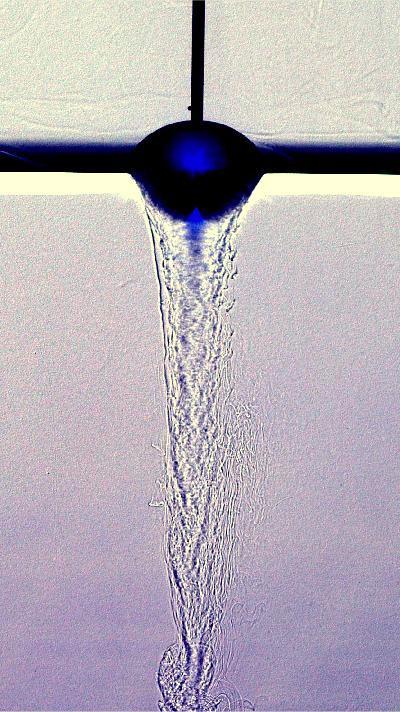
Eroding Candy
When you pop a hard candy in your mouth, you probably don’t give much thought to the fluid dynamics involved in dissolving it. The series above shows a hard candy suspended in water being slowly eaten away. As sugars in the candy dissolve into the water, the fluid becomes denser and falls away. This creates the downward flow visible in the center of the image. As sugar-laden water sinks, fresher water is pulled in alongside the walls of the candy. That flow helps erode the candy, creating a rougher surface. Since rough surfaces have a greater surface area exposed (than a smooth surface), they prompt further and faster dissolution. That strengthens the downward flow, pulls in more ambient water, and keeps the whole process going. (Image credit: M. Wykes)

“Ink in Motion”
In this short film, the Macro Room team plays with the diffusion of ink in water and its interaction with various shapes. Injecting ink with a syringe results in a beautiful, billowing turbulent plume. By fiddling with the playback time, the video really highlights some of the neat instabilities the ink goes through before it mixes. Note how the yellow ink at 1:12 breaks into jellyfish-like shapes with tentacles that sprout more ink; that’s a classic form of the Rayleigh-Taylor instability, driven by the higher density ink sinking through the lower density water. Ink’s higher density is what drives the ink-falls flowing down the flowers in the final segment, too. Definitely take a couple minutes to watch the full video. (Image and video credit: Macro Room; via James H./Flow Vis)