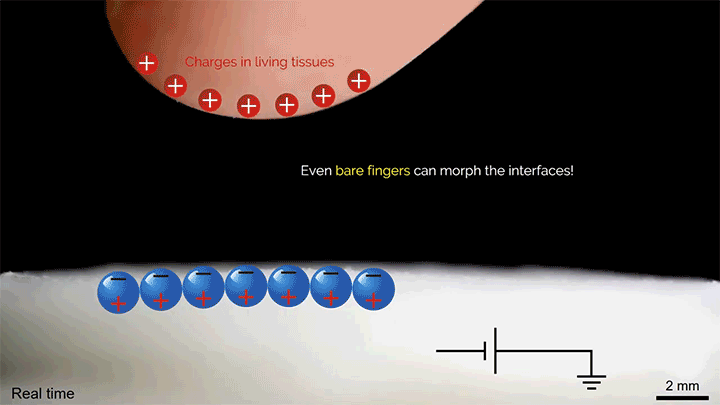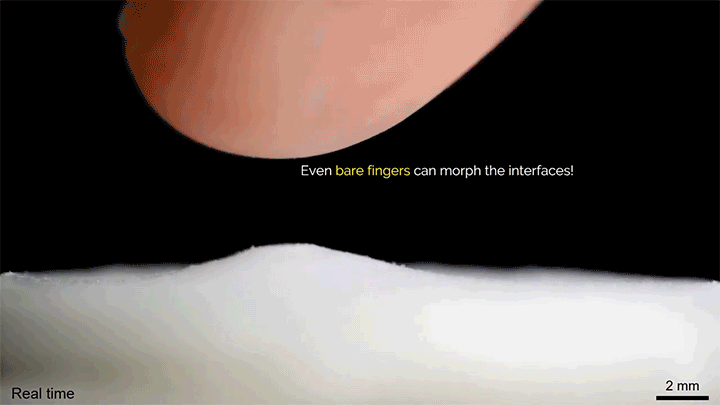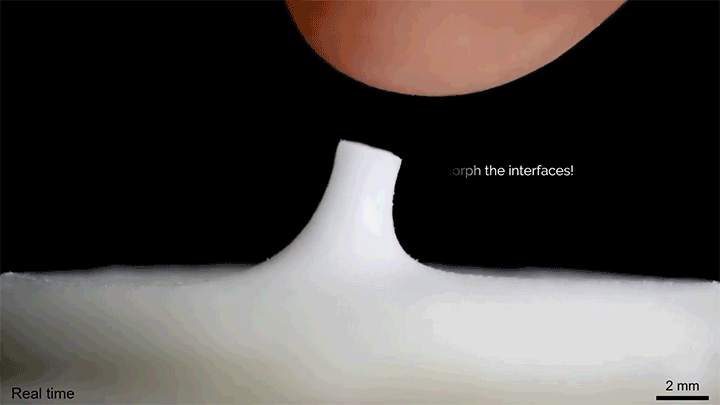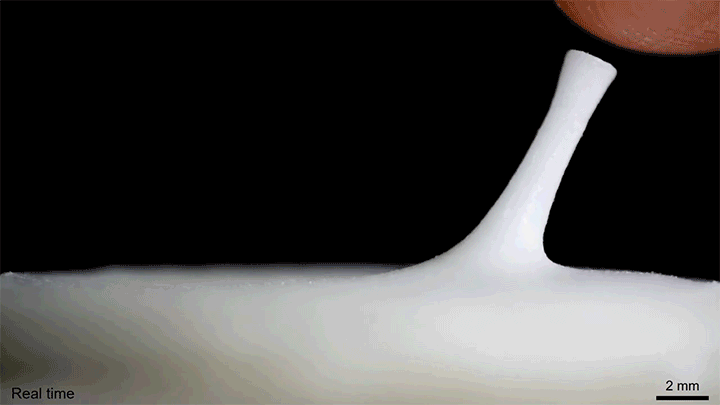
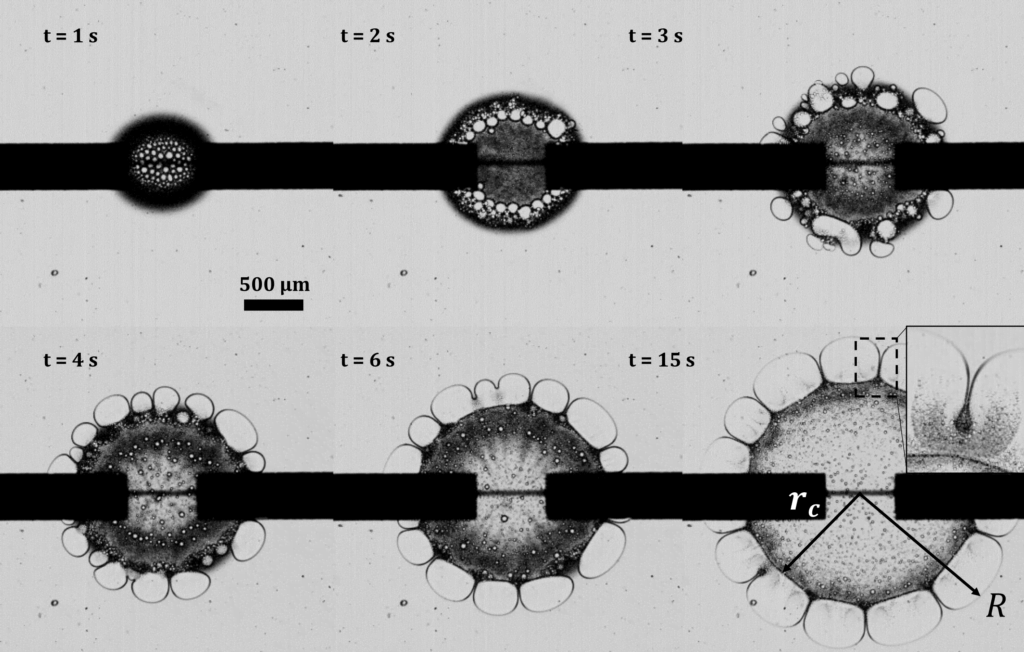
A layer of tiny glass beads sitting atop a pool of castor oil becomes a morphing surface in this video. Applying an electric field creates enough electrostatic force to draw the interface upward against the power of both gravity and surface tension. Moving the electric field — either by shifting the electrode or simply moving a finger over the surface — is enough to pull columns of fluid along! I could imagine this making some very cool human-machine interfaces one day. (Image and video credit: K. Sun et al.)
Search results for: “surface tension”

Wild Patterns in Ionic Liquids
Ionic liquids are essentially salts in a liquid form. In these images, a mixture of water and ionic liquid separates when heated. This phase separation causes the initial mixture to break into two regions: one low in ionic liquid and one rich in ionic liquid. Because the surface tensions of these two phases are different from one another, complex flow patterns form. (Image and research credit: M. Pascual et al.)

Acrylic Paint Fractals
Here’s a simple fluids experiment you can try at home using acrylic paints, ink, isopropyl alcohol and a few other ingredients. When dropped onto diluted acrylic paint, a mixture of black ink and alcohol spreads in a fractal fingering pattern. The radial (outward) flow is driven by the alcohol’s evaporation, which increases the local surface tension and draws fluid outward. The shape and density of the fingers depends, at least in part, on the viscosity of the underlying paint layer; more viscous paint layers grow smaller and denser fractal patterns. (Image and video credit: S. Chan et al.)

The Assassin’s Teapot
The assassin’s teapot is a cleverly designed container that can pour from different reservoirs depending on how it’s held. Steve Mould digs into the physics in this video, and he builds a transparent cutaway version of the pot to show exactly how it works. This design uses two separate reservoirs, each with two holes — one in the spout and one concealed near the pot’s handle. By covering this breather hole, the server blocks air from flowing into the teapot, which also keeps the liquid inside from flowing out.
What holds the liquid in? Air pressure, with an assist from surface tension. Atmospheric pressure is enough to hold the fluid inside the pot, provided air has no separate way in. To get in through the spout, air would have to push into the pot at the same time as water coming out. Surface tension prevents that, though, because the spout is too narrow. The same physics keeps water inside a larger bottle with a wire mesh over its mouth. The mesh’s tiny holes are smaller than the capillary length of water, which is the length scale at which surface tension and gravity balance one another. As long as the spout and holes are smaller than that length, surface tension will keep the liquid from deforming enough to get out. (Video and image credit: S. Mould)

The Yarning Droplet
Marangoni bursting takes place in alcohol-water droplets; as the alcohol evaporates, surface tension changes across the liquid surface, generating a flow that tears the original drop into smaller droplets. Here researchers add a twist to the experiment using PMMA, an additive that dissolves well in alcohol but poorly in water. As the alcohol evaporates, the PMMA precipitates back out of the water-rich droplet, forming yarn-like strands. (Image and video credit: C. Seyfert and A. Marin)

Viscosity and Quantum Mechanics
Viscosity describes a fluid’s resistance to changing its shape. Like surface tension, it’s a fundamental property of a fluid that comes from the interactions between molecules. But viscosity is a slippery beast, and especially so for liquids. There is no generic way to calculate a liquid’s thermodynamic properties from quantum dynamical first principles. But that hasn’t stopped theoretical physicists from making progress on deducing the connections between quantum mechanics and liquids.
Although viscosity changes with temperature, all liquids have a minimum viscosity, and those minima are all fairly close to the same value as water’s (excluding any superfluids, which are their own brand of quantum weirdness). Why would liquids share a similar minimum viscosity? Because it turns out the minimum viscosity is quantum! Physicists found that the minimum viscosity is set by an equation depending on Planck’s constant and the mass of an electron — both fundamental constants.
Physicists sometimes like to conjecture about the habitability of the universe if fundamental quantities like Planck’s constant had a different value. This work shows that changing that value would alter water’s viscosity, completely changing the viability of microscopic life! (Image credit: A. Rozetsky; research credit: K. Trachenko and V. Brazhkin; via Physics Today)

Ink-Based Propulsion
In this video, Steve Mould explores an interesting phenomenon: propulsion via ballpoint pen ink. Placing ink on one side of a leaf or piece of paper turns it into a boat with a dramatic dye-filled wake. It’s not 100% clear what’s happening here, though I agree with Steve that there are likely several effects contributing.
Firstly, there’s the Marangoni effect, the flow that happens from an area of low surface tension to high surface tension. This is what propels a soap boat as well as many water-walking insects. I think this is a big one here, and not just because the ink has surfactants. As any component of the ballpoint ink spreads, its varying concentration is going to trigger this effect.
Secondly, there’s a rocket effect. Rockets operate on a fairly simple principle: throw mass out the back in order to go forward. These dye boats are also doing this to some extent.
And finally there’s some chemistry going on. Some kind of reaction seems to be taking place between one or more of the ink components and the water in order to create the semi-solid layer of dye. Presumably this is why the dye doesn’t simply dissolve as it does in some of Steve’s other experiments.
I figure some of my readers who are better versed in interfacial dynamics, rheology, and surface chemistry than I am will have some more insights. What do you think is going on here? (Video and image credit: S. Mould)

Paint Spinning
In a return to their roots, this Slow Mo Guys video features paint flowing on (and off!) a spinning disk. To help us see what’s going on, Gav uses a trick that’s familiar to many fluid dynamicists: he rotates the high-speed footage at the same speed that the disk rotates. This transformation places the viewer into a reference frame where the disk appears stationary, so that small changes in the flow are apparent.
It makes for a gorgeous view as centrifugal force flings the paint outward and eventually breaks it into drops. The rotation speed is unfortunately so high that the spinning completely dominates all other forces. The few runs with more viscous acrylic paint show some hints of more interesting behaviors that might be visible with a slower rotation rate (which would make the tug of war between inertia/viscosity/surface tension and centrifugal force less one-sided). Anyone got a high-speed camera, some speed control, and a willingness to get messy? (Image and video credit: The Slow Mo Guys)

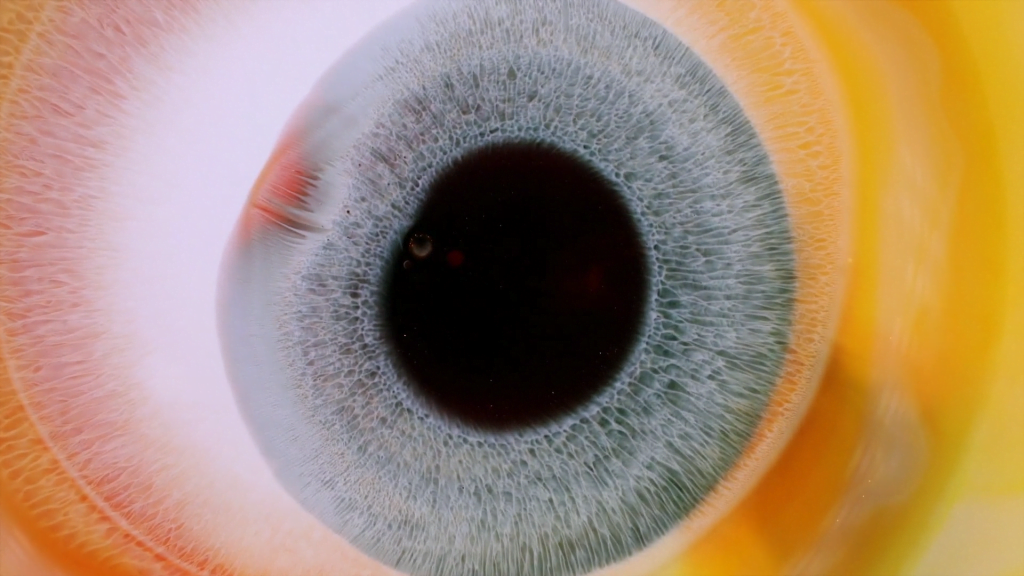
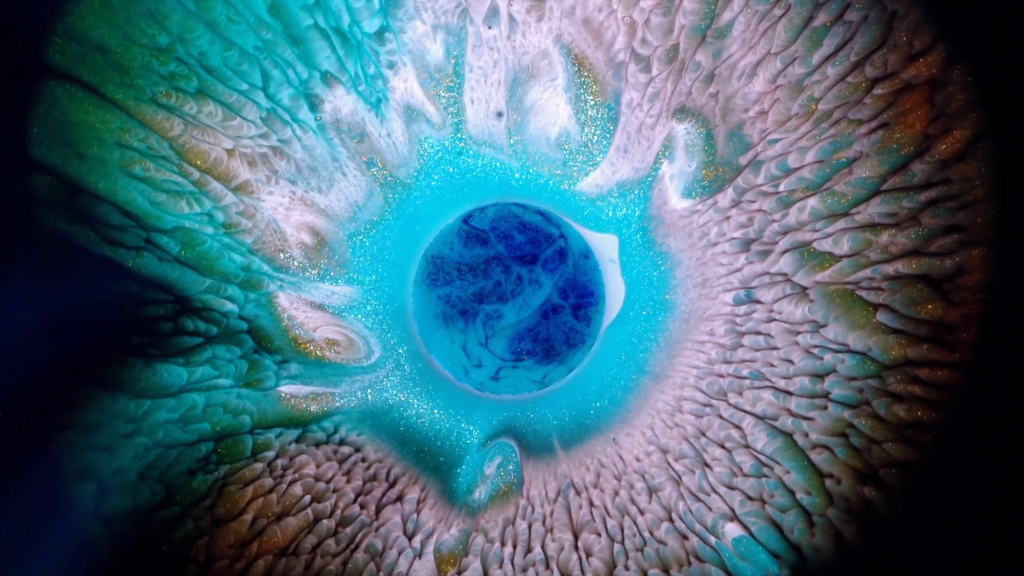
“Heterochromia Iridum”
Heterochromia iridum is the formal name for when a person’s irises are multi-colored, often with streaks or swirls of one color cutting through another. In this short film, photographer Rus Khasanov recreates the effect with glittery inks and paints. Their varying surface tensions help create the eye-like streaks and feathers through the Marangoni effect. Check out the full video to see the effect in action. (Image and video credit: R. Khasanov; via Colossal)

Taking A Turn
Water droplets immersed in a mixture of oil and surfactants will move about, propelled by the Marangoni effect. Surfactant molecules congregate along the interface between the water and oil, but they do not do so uniformly. This uneven grouping causes variations in the surface tension, which in turn creates flows inside the droplet from areas of low surface tension to ones with higher surface tension. Those internal flows then dictate how the droplet as a whole moves.
Researchers found that droplet trajectories in these systems depend on the droplet’s size. Small droplets move in relatively straight lines, whereas larger droplets take highly curved paths. The difference comes from the way surfactants get distributed around the drop’s interface. Larger drops are more sensitive to shifts in surfactant location, making them more prone to take changeable, curving paths. (Image credits: top – P. Godfrey, others – S. Suda et al.; research credit: S. Suda et al.; via APS Physics)