A liquid drop can fold itself up in a thin sheet. The animation above shows a drop of water with an ultra-thin (79nm) circular sheet of polystyrene atop it. As a needle removes water from the underside of the droplet, the shrinking droplet causes wrinkles and folds to form in the sheet. What’s going on here is a competition between the energy required to change the droplet’s shape and the energy needed to bend the sheet. Eventually, the droplet’s volume is small enough that the bending of the sheet overrules surface tension in dictating the droplet’s shape. The result is a tiny empanada-shaped droplet completely encapsulated by the sheet. (Image credit: J. Paulsen et al., source; research paper)
Search results for: “droplet”
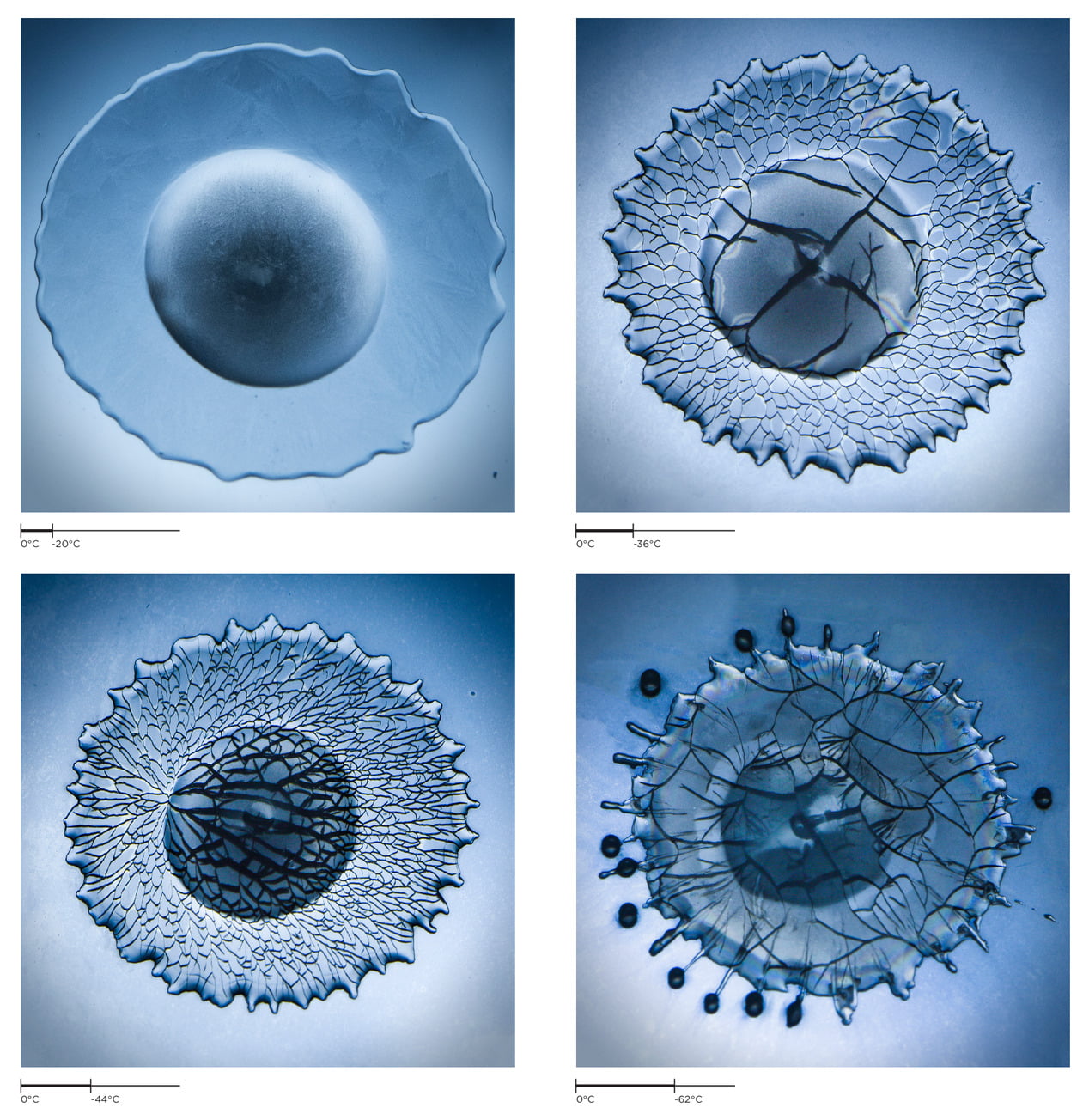
Freezing Impact
When a water droplet hits a frozen surface, what happens depends significantly on the temperature of the substrate. At relatively high temperatures (-20 degrees C), the droplet freezes without any cracking (upper left). As the surface gets colder, drops begin to crack. At first the cracks are relatively large and unstructured (upper right), but at lower temperatures, they grow in a network of smaller cracks with more distinctive structure (lower left). Cold temperatures can also affect the contact line where water, air, and substrate meet. This can cause droplets to splash even as they’re freezing (lower right). (Image credit: V. Thievenaz et al.; see also E. Ghabache et al.)

Popping
Popcorn’s explosive pop looks pretty cool in high-speed video, but just watching it with a regular camera doesn’t show everything that’s going on. If we take a look at it through schlieren optics, the kernel’s pop looks even more extraordinary:
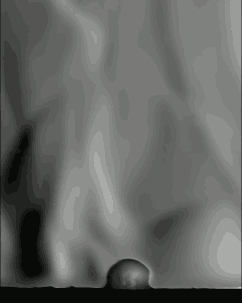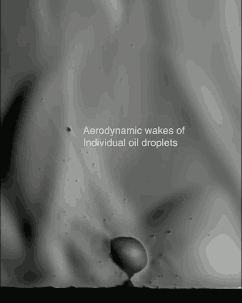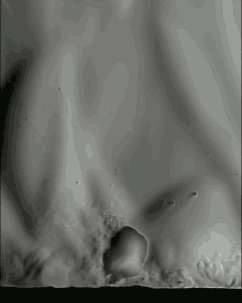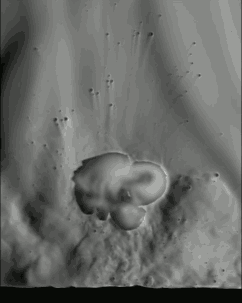
The schlieren technique reveals density differences in the gases around the corn–effectively allowing us to see what is invisible to the naked eye. The popcorn kernel acts like a pressure vessel until the expansion of steam inside causes its shell to rupture. The first hints of escaping steam send droplets of oil shooting upward. The kernel may hop as steam pours out the rupture point, causing the turbulent billowing seen in the animation above. As the heat causes legs of starch to expand out of the kernel, they can push off the ground and propel the popcorn higher. As for the eponymous popping sound, that is the result of escaping water vapor, not the actual rupture or rebound of the kernel! See more of the invisible world surrounding a popping kernel in the video below. (Image credits: Warped Perception, source; Bell Labs Ireland, source; WP video via Gizmodo; BLI video submitted by Kevin)

Vibrated to Bits
Sound and vibration can be powerful tools for controlling liquids. In this animation, a water/glycerin drop violently bursts into a cloud of droplets when it is vibrated vertically 1000 times per second by a piezoelectric actuator. This vibration shakes the drop with accelerations of 150 g. Initially, the amplitude is small enough to simply create ripples around the drop’s circumference. As it increases, the drop deforms more at the edges and starts to eject droplets there. When the vibration hits a critical amplitude, the entire drop explodes into droplets. The technique is called vibration-induced droplet bursting, and its near-instantaneous ability to atomize drops makes it a candidate for applications like spray cooling microprocessors or spray coating a solid surface. (Video credit: B. Vukasinovic, source)
Dissolving
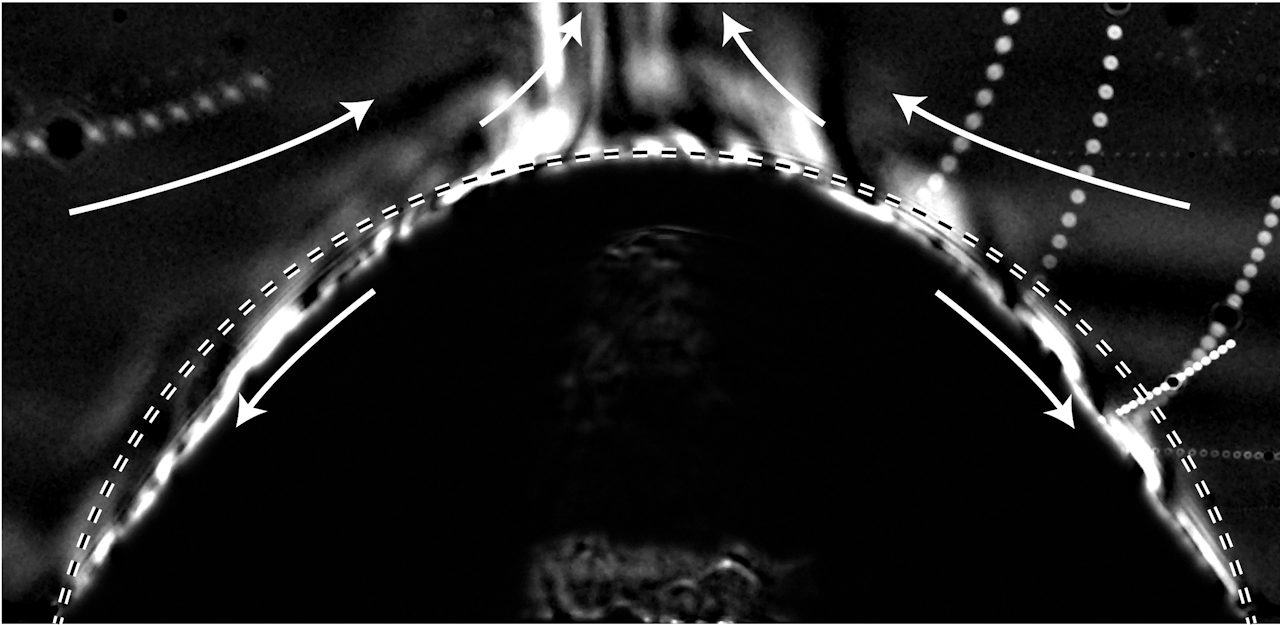
It looks like the fiery edge of a star’s corona, but this photo actually shows a dissolving droplet. The droplet, shown as the lower dark region in this shadowgraph image, is a mixture of pentanol and decanol sitting in a bath of water. Pentanol is a type of alcohol that is fully miscible with decanol and is water soluble, so that it will dissolve into the surrounding water over time. Decanol, on the other hand, is immiscible with water, so that part of the droplet won’t mix with the surrounding water.
The bright swirls along the droplet’s edge show areas with more pentanol. As the alcohol dissolves into the water, it forms a buoyant plume at the top of the droplet that rises due to pentanol’s lower density. That rising plume draws fresh water in from the sides, shown by the upper white arrows. Inside the droplet, flow moves in the opposite direction, from the top toward the outer edges. This is a result of uneven surface tension within the droplet. Scientists are interested in understanding the dynamics of these multiple component drops for applications like printing, where it’s desirable for pigments in an ink drop to be distributed evenly as the drop dries. (Image credit: E. Dietrich et al.)

A Water Balloon on a Bed of Nails
If you dropped a water balloon on a bed of nails, you’d expect it to burst spectacularly. And you’d be right – some of the time. Under the right conditions, though, you’d see what a high-speed camera caught in the animation above: a pancake-shaped bounce with nary a leak. Physically, this is a scaled-up version of what happens to a water droplet when it hits a superhydrophobic surface.
Water repellent superhydrophobic surfaces are covered in microscale roughness, much like a bed of tiny nails. When the balloon (or droplet) hits, it deforms into the gaps between posts. In the case of the water balloon, its rubbery exterior pulls back against that deformation. (For the droplet, the same effect is provided by surface tension.) That tension pulls the deformed parts of the balloon back up, causing the whole balloon to rebound off the nails in a pancake-like shape. For more, check out this video on the student balloon project or the original water droplet research. (Image credits: T. Hecksher et al., Y. Liu et al.; via The New York Times; submitted by Justin B.)


Shot Through a Drop
Shoot a sphere through a drop with sufficient speed, and you’ll see something like the composite photo above. Going from right to left, the projectile is initially coated in liquid and stretches the fluid behind it as it continues flying. This forms a thin sheet of fluid called a lamella with a thicker, uneven rim at its far end. The lamella continues stretching until the projectile breaks through and detaches. Now the lamella starts rebounding back on itself as surface tension struggles to keep the fluid together. A new rim forms on the front, and both the front and back rims thicken as the lamella collapses. Along the rims thicker portions start forming droplets – like spikes on a crown – as the surface-tension-driven Plateau-Rayleigh instability starts breaking the structure down. The untenable sheet of fluid will break up into a cloud of smaller, satellite droplets when it can hold together no longer. (Image credit: V. Sechenyh et al., video)

“Kingdom of Colours”
Oil, paint, and soap combine to create a polychrome landscape in Thomas Blanchard’s “Kingdom of Colours” short film. Colorful droplets of paint coated in oil form anti-bubbles that skim along the liquid surface until they burst, dispersing new colors. One of my favorite touches in this video, though, are the branching fingers of color that appear repeatedly (most often in blue-violet). This is an example of a phenomenon known as the Saffman-Taylor instability. It’s a hallmark of a low viscosity fluid pushing into a higher viscosity one–like air into honey. (Image/video credit: T. Blanchard; via Flow Vis)




Erie Waves
Photographer Dave Sandford braved the cold and turbulent waters of Lake Erie in late fall to capture some remarkable wave action. Like on the ocean, waves in the Great Lakes are largely driven by winds, but lakes don’t develop the constant set of rolling waves that oceans do. Instead their waves are more erratic and unpredictable. Sandford focused on capturing the moment when wind-driven waves coming into shore collided with waves rebounding from piers or rocks along the shore. The results are waves that, through Sandford’s lens, look like exploding mountainsides. Such energetic waves mix sediment and nutrients in the lake, and the spray of droplets can even loft aerosols and pollutants from the water into the atmosphere. (Photo credit: D. Sandford; via Flow Vis)

When Jets Collide
Two liquids that collide don’t always coalesce. The image above shows two jets of silicone oil colliding. On the left, the jets collide and bounce off one another. On the right, at a slightly higher flow rate, the two jets coalesce. This bouncing, or noncoalescence, observed at lower speeds is due to an incredibly thin layer of air separating the two jets. This air layer is constantly being replenished by air that gets dragged along by the flowing oil. But if the oil flows too quickly, that air layer becomes unstable–in the same way that a droplet that falls too quickly will splash on impact. When the separating air layer becomes unstable and breaks down, the jets collide and merge. (Image credit: N. Wadhwa et al., pdf)


