How liquid droplets spread on solid surfaces is pretty well understood, but researchers have looked less at the related problem of how a gas spreads. In a recent paper, scientists have examined the spreading dynamics of bubbles impacting an immersed solid. As the bubble contacts the surface, it quickly squeezes out water trapped between the bubble and the gas layer trapped at the solid surface. The bubble squishes as surface tension tries to flatten the liquid-gas interface. Buoyancy also helps flatten the bubble. The spreading is remarkably fast, taking only about 10 milliseconds. That’s good news for the many insects who use trapped air bubbles like these to breathe underwater. Check out the video below to learn about some of these natural scuba divers. (Image credit: H. de Maleprade et al., source; video credit: Deep Look)
Search results for: “droplet”

Icy Spikes
Water is one of those strange materials that expands when it freezes, which raises an interesting question: what happens to a water drop that freezes from the outside in? A freezing water droplet quickly forms an ice shell (top image) that expands inward, squeezing the water inside. As the pressure rises, the droplet develops a spicule – a lance-like projection that helps relieve some of the pressure.
Eventually the spicule stops growing and pressure rises inside the freezing drop. Cracks split the shell, and, as they pull open, the cracks cause a sudden drop in pressure for the water inside (middle image). If the droplet is large enough, the pressure drop is enough for cavitation bubbles to form. You can see them in the middle image just as the cracks appear.
After an extended cycle of cracking and healing, the elastic energy released from a crack can finally overcome surface energy’s ability to hold the drop together and it will explode spectacularly (bottom image). This only happens for drops larger than a millimeter, though. Smaller drops – like those found in clouds – won’t explode thanks to the added effects of surface tension. (Image credit: S. Wildeman et al., source)
ETA: A previous version of this post erroneously said this was freezing from the “inside out” instead of “outside in”.

How Rainfall Can Spread Pathogens

Rainfall may provide a mechanism for soil bacteria to spread. A new study examines how raindrops hitting infected soil can eject bacteria into the air. When drops fall at the rate of a light rainfall, they form tiny bubbles after impact (upper left). Those microbubbles rise to the top of the water and burst, sending extremely tiny droplets – or aerosols – spraying up into the air (upper right). Soil bacteria can hitch a ride on these aerosols, staying alive for up to an hour while the wind transports them to fresh, new soil. The researchers found that the most aerosols were produced when soil temperature was about 86 degrees Fahrenheit (30 degrees Celsius) – the temperature of tropical soils. Depending on the conditions, a single raindrop could aerosolize anything from zero to several thousands of soil bacteria. (Image and research credit: Y. Joung et al.; video credit: MIT News)

Self-Propelling Drops
Droplets of acetone deposited on a bath of warm water can float along on a Leidenfrost-like vapor layer. The droplets are self-propelling, too, thanks to interactions between the acetone and water. Acetone can dissolve in water, and when acetone vapor beneath the drop gets absorbed into the water bath, it lowers the local surface tension. That drop in surface tension creates a pull in the direction of a higher surface tension; this is what is known as the Marangoni effect. Because of that flow in the direction of higher surface tension, the acetone drop accelerates away. (Image credit: S. Janssens et al., source)
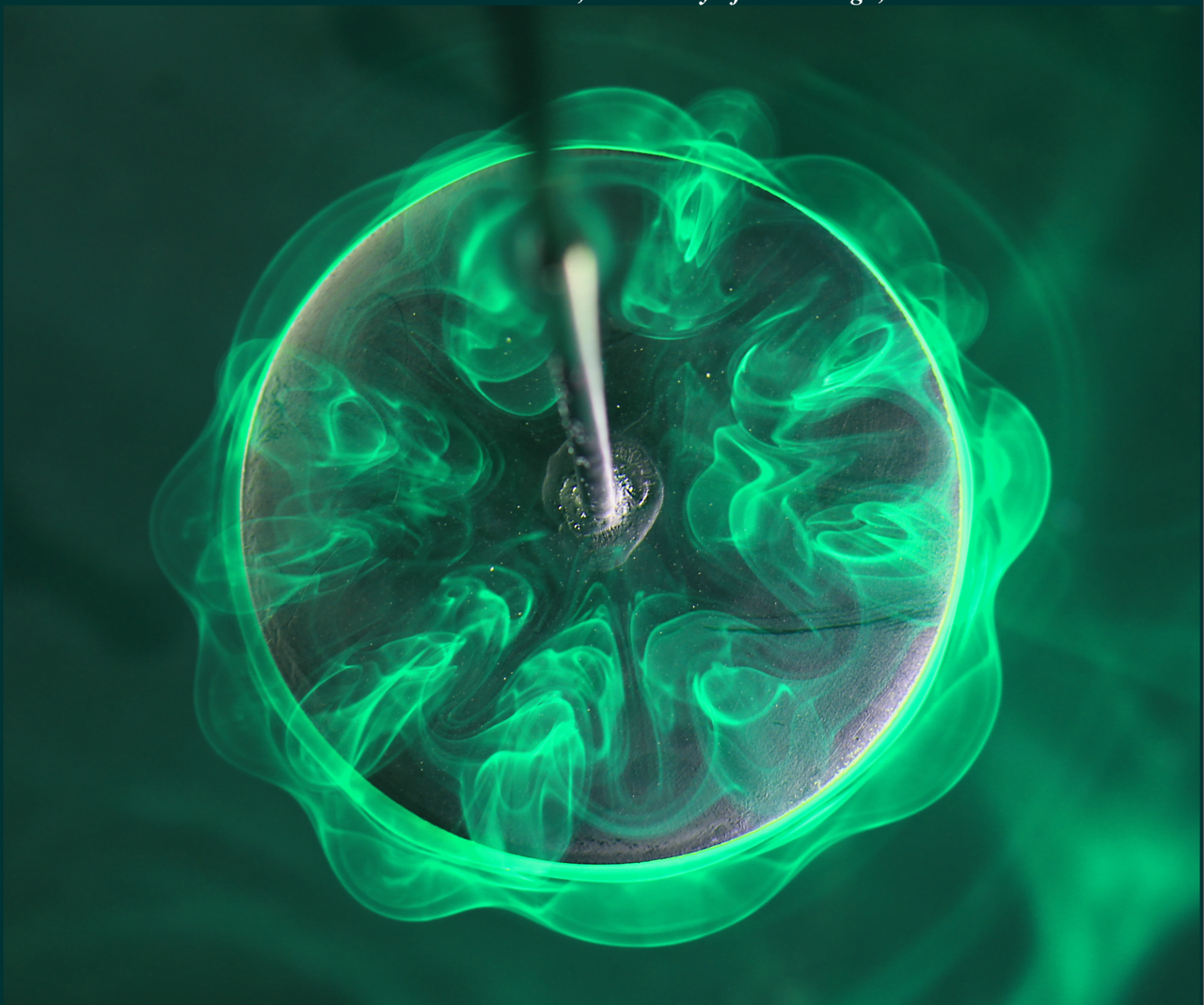
Breaking Down Vortices
Vortex rings are ubiquitous in nature, showing up in droplet impacts, in propulsion, and even in volcanic eruptions. Understanding the interaction and breakdown of multiple vortices with one another is therefore key. The image above shows a circular disk that’s being oscillated up and down (in and out of the page). As the disk moves and changes direction, it generates vortices that interact with one another. Here some of those interactions are visualized with fluorescent dye. The overlapping vortices form complex and beautiful shapes on their way to breakdown. (Image credit: J. Deng et al., poster, paper)

Molten Copper
In this video, the Slow Mo Guys prove that pouring molten copper in slow motion is every bit as satisfying as one would imagine. Because they pour the metal from fairly high up, they get a nice break-up from a jet into a series of droplets; that’s due to the Plateau-Rayleigh instability, in which surface tension drives the fluid to break up into drops. Upon impact, the copper splashes and splatters very nicely, forming the crown-like splash many are familiar with from famous photos like Doc Edgerton’s milk drop. The key difference between the molten copper and any other liquid’s splash comes from cooling; watch closely and you’ll see some of the copper solidifying along the edges and surface of the fluid as it cools. In this respect, watching the molten copper is more like watching lava flow than seeing water splash. (Video and image credit: The Slow Mo Guys)


Supporting Bubbles
Surface tension holds small droplets in a partial sphere known as a spherical cap. But when droplets become larger, they flatten out into puddles due to the influence of gravity. In contrast, soap bubbles remain spherical to much larger sizes. The bubble pictured above, for example, is more than 1 meter in radius and nearly 1 meter in height.
There is a maximum height for a soap bubble, though, and it’s set by the physical chemistry of the surfactants used in the soap. To support itself, the bubble requires a difference in surface tension between the top and bottom of the bubble. A higher surface tension is necessary at the top of the bubble to help prevent fluid from draining away. The difference in surface tension between the top and bottom of the bubble can never be greater than the difference in surface tension between pure water and the soap mixture – thus those values set a maximum height for a bubble. The researchers found their bubbles maxed out at a height of about 2 meters, consistent with their theoretical predictions. (Image credit: C. Cohen et al.; via freshphotons)

Aerodynamic Leidenfrost Effect

If you place a droplet on a surface much hotter than its boiling point, that droplet will skitter and float almost frictionlessly across the surface on a thin layer of its own vapor. This is what is known as the Leidenfrost effect. But you don’t have to heat a surface to get this behavior. There’s also an aerodynamic Leidenfrost effect, shown above, when the surface is moving. As the surface moves, it drags a layer of air along with it, and that layer of air is capable of keeping droplets aloft indefinitely. The thickness of the air layer depends on speed; the faster the plate moves, the thicker the air layer underneath droplets. The aerodynamic forces generated are large enough to drive a droplet up an incline against the force of gravity (bottom image). (Image credit: animation – M. Saito et al., source; chronophotograph – A. Gautheir et al., pdf)
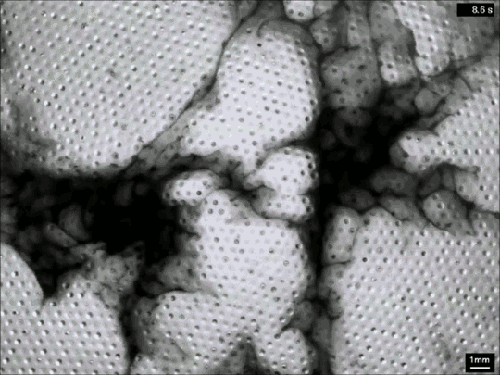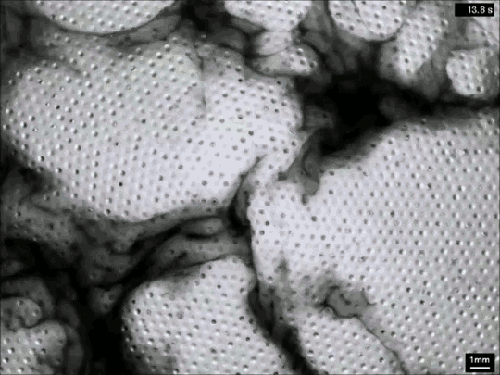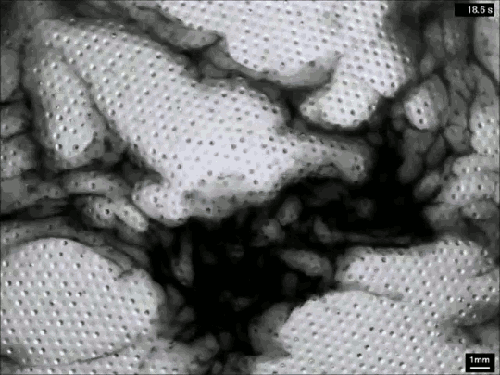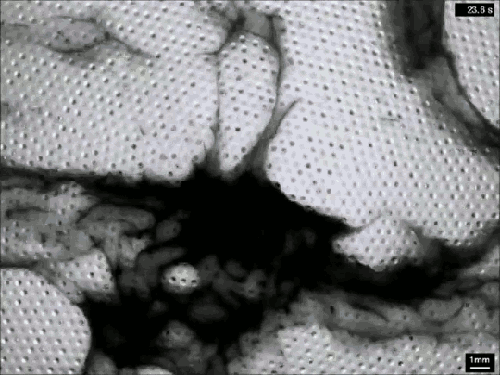
Creating Clouds
What you see here is the formation of clouds and rain – but it’s not quite what you’re used to seeing outside. This is an experiment using a mixture of sulfur hexafluoride and helium to create clouds in a laboratory. Everything is contained in a cell between two transparent plates. Liquid sulfur hexafluoride takes up about half of the cell, and when the lower plate is heated, that liquid begins evaporating and rising in the bright regions. When it reaches the cooled top plate, the liquid condenses into droplets inside the dimples on the plate, eventually growing large enough to fall back as rain. The dark wisps you see are areas where cold sulfur hexafluoride is sinking, much like in the water clouds we are used to. Setups like this one allow scientists to study the effects of turbulence on cloud physics and the formation of droplets. (Image credit: E. Bodenschatz et al., source)
Boston-area folks! I’ll be taking part in the Improbable Research show Saturday evening at 8 pm at the Sheraton Boston. Come hear about the Boston Molasses Flood and other bizarre research!

Leidenfrost Atop a Fluid
Leidenfrost droplets typically hover on a thin layer of vapor above a surface that is much hotter than the boiling point of the liquid. Such drops move almost frictionlessly across these surfaces and can even propel themselves. The question of how hot is hot enough to produce the Leidenfrost effect is still being debated, but recent research suggests that the answer may depend strongly on surface roughness.
To test the role of surface roughness, one group tested drops of ethanol atop a heated pool of silicone oil, as pictured above. Ethanol’s boiling point is 78 degrees Celsius, and the researchers found they could hold the ethanol drop in a Leidenfrost state by heating the pool to 79 degrees Celsius – only 1 degree above ethanol’s boiling point! Thanks to surface tension, a liquid surface is essentially molecularly smooth. The fact that solid surfaces require much higher temperatures before the Leidenfrost effect is observed indicates that even the slightest roughness can have a large impact on the Leidenfrost temperature. (Image credit: F. Cavagnon; research credit: L. Maquet et al., pdf)
Heads-up for Boston-area folks! I’ll be taking part this Saturday evening in the Improbable Research show at the AAAS conference. The show is free and open to the public but fills up quickly, so be sure to come early for a seat.


