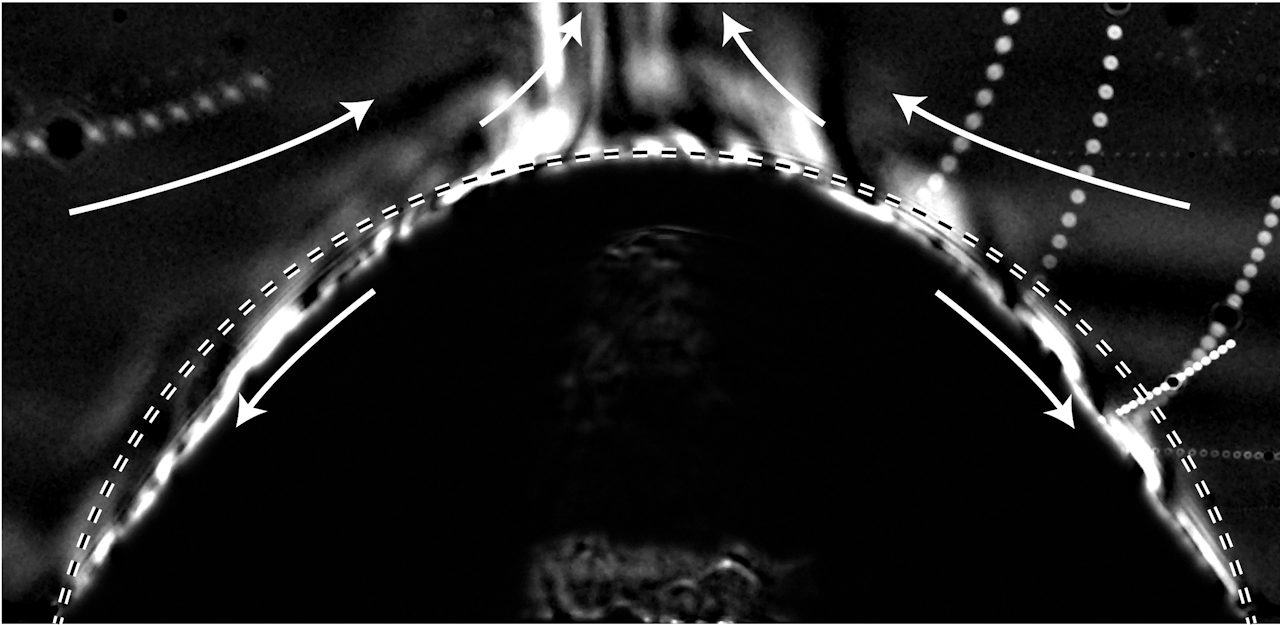
It looks like the fiery edge of a star’s corona, but this photo actually shows a dissolving droplet. The droplet, shown as the lower dark region in this shadowgraph image, is a mixture of pentanol and decanol sitting in a bath of water. Pentanol is a type of alcohol that is fully miscible with decanol and is water soluble, so that it will dissolve into the surrounding water over time. Decanol, on the other hand, is immiscible with water, so that part of the droplet won’t mix with the surrounding water.
The bright swirls along the droplet’s edge show areas with more pentanol. As the alcohol dissolves into the water, it forms a buoyant plume at the top of the droplet that rises due to pentanol’s lower density. That rising plume draws fresh water in from the sides, shown by the upper white arrows. Inside the droplet, flow moves in the opposite direction, from the top toward the outer edges. This is a result of uneven surface tension within the droplet. Scientists are interested in understanding the dynamics of these multiple component drops for applications like printing, where it’s desirable for pigments in an ink drop to be distributed evenly as the drop dries. (Image credit: E. Dietrich et al.)
Leave a Reply