We’re surrounded daily by convection — a buoyancy-driven flow — but most of the time it’s invisible to us. In this video, Steve Mould shows off what convection really looks like with some of his excellent tabletop demos. The first half of the video gives profile views of turbulent convection, with chaotic and unsteady patterns. When he switches to oil instead of water, the higher viscosity (and lower Reynolds number) offer a more structured, laminar look. And finally, he shows a little non-temperature-dependent convection with a mixture of Tia Maria and cream, which convects due to evaporation changing the density. (Image and video credit: S. Mould; submitted by Eric W.)
Search results for: “density”

“Lucid”
Artist Roman Hill made this official music video to go with Thomas Vanz’s “Lucid.” The imagery, formed from ink and other fluids, warps our sense of scale. Though the camera focuses on an extremely small area, to our eyes the results shift from nebulas to oceans and back again. There are likely a whole host of phenomena going on here, but without knowing more about Hill’s ingredients, I can only speculate that there are Marangoni flows driven by variations in surface tension and maybe some density instabilities going on between fluid layers. I’m also fairly confident that Hill has played with time reversal in the video editing. Regardless of the secrets in its making, the film is captivating and gorgeous. (Image and video credit: R. Hill)

Tumbling in Air
When snowflakes and volcanic ash fall, they tumble. Historically, it’s been too hard to observe this behavior first hand — the particles are too small to easily follow with a camera — so scientists instead looked at larger particles falling through water. That change preserves important characteristics of the physics, but it misses out on one key feature: in air, the density of the falling particle is much higher than air’s.

A football-shaped particle wobbles around its stable orientation as it falls through air. To account for that, researchers built a special apparatus that drops particles one-at-a-time through the field of view of four high-speed cameras. This setup gave them a narrow 1-mm band where they could track a falling particle’s orientation — provided the particle fell through the band, which happened about 20% of the time. Their results show that particles in air tumble and oscillate back and forth around their stable orientation more than in water experiments. This difference affects how quickly particles settle, which, in turn, affects how much they tend to clump and grow. (Image credit: snow – A. Burden, experiment – T. Bhowmick et al.; research credit: T. Bhowmick et al.; via APS Physics)

“Alive”
In “Alive,” filmmaker Christopher Dormoy explores acrylic paints and the variety of ways in which the medium can be used. From a fluids perspective, there’s dripping, viscous flow, turbulent eddies, billowing plumes, and “accidental painting” due to density-driven instabilities. It’s a fun tour of fluid phenomena in art. What examples do you spot? (Video and image credit: C. Dormoy)

Understanding Cyanobacteria
Over 2 billion years ago, cyanobacteria emerged as Earth’s first photosynthesizing organisms. Today they are widespread and critical contributors to both carbon and nitrogen cycles. Colonies can form large mats, like those pictured above, but, even at the microscale, cyanobacteria are actively forming patterns among individual bacteria. A recent study considers cyanobacteria as active matter.

At the microscopic scale, cyanobacteria form different patterns, depending on their density. By simulating the cyanobacteria as filaments that interact through a series of simple rules, the researchers were able to reproduce the complex patterns bacterial colonies form. Their physical model also offered an explanation — based on the relative importance of advective and diffusive transport — for the characteristic length scales found in the bacterial patterns. (Image credit: Yellowstone – B. Cappellacci, patterns – M. Faluweki et al.; research credit: M. Faluweki et al.; via APS Physics)

Food-Based Fluid Dynamics
The kitchen is a rich source of fluid physics. From cocktails to coffee, from crepes to tempura, food is full of physics. In fact, it’s not hard to relate almost any fluid phenomenon you can imagine to something that goes on in the kitchen. That’s why scientists managed to write a 77-page review article of culinary fluid dynamics. It’s even structured after a menu, carrying readers from the kitchen sink and cocktails all the way through a meal and the process of washing up afterward! (Image credit: top – S. Hsu, others – A. Mathijssen et al.; research credit: A. Mathijssen et al.; via APS Physics)

Dancing Peanuts
Bartenders in Argentina sometimes entertain patrons by tossing a few peanuts into their beer. Initially, the peanuts sink, but after a few seconds they rise, wreathed in bubbles. Once on the surface, they roll, causing the bubbles to pop, and the peanut sinks once again. The cycle repeats, sometimes for as long as a couple hours.
There are a couple physical processes governing this dance. The first is bubble nucleation. Most beers are carbonated; they contain dissolved carbon dioxide gas that remains in solution while the beer is under pressure. Once poured, that storage pressure is gone and bubbles start to form in the liquid. The shape of the peanut means that bubbles form more easily on it than on the glass walls or in the liquid. And once the peanut is covered in bubbles, buoyancy comes into play. The bubbles attached to the peanut reduce its density relative to the surrounding fluid, enabling the peanut to rise up and float.
This same process is seen with other objects in carbonated fluids, too, such as blueberries in beer and lemon seeds in carbonated water. But it’s also reflected elsewhere in nature. For example, magnetite crystals are thought to float in magma due to a similar nucleation of dissolved gases on their surface. (Image and research credit: L. Pereira et al.; via APS Physics)
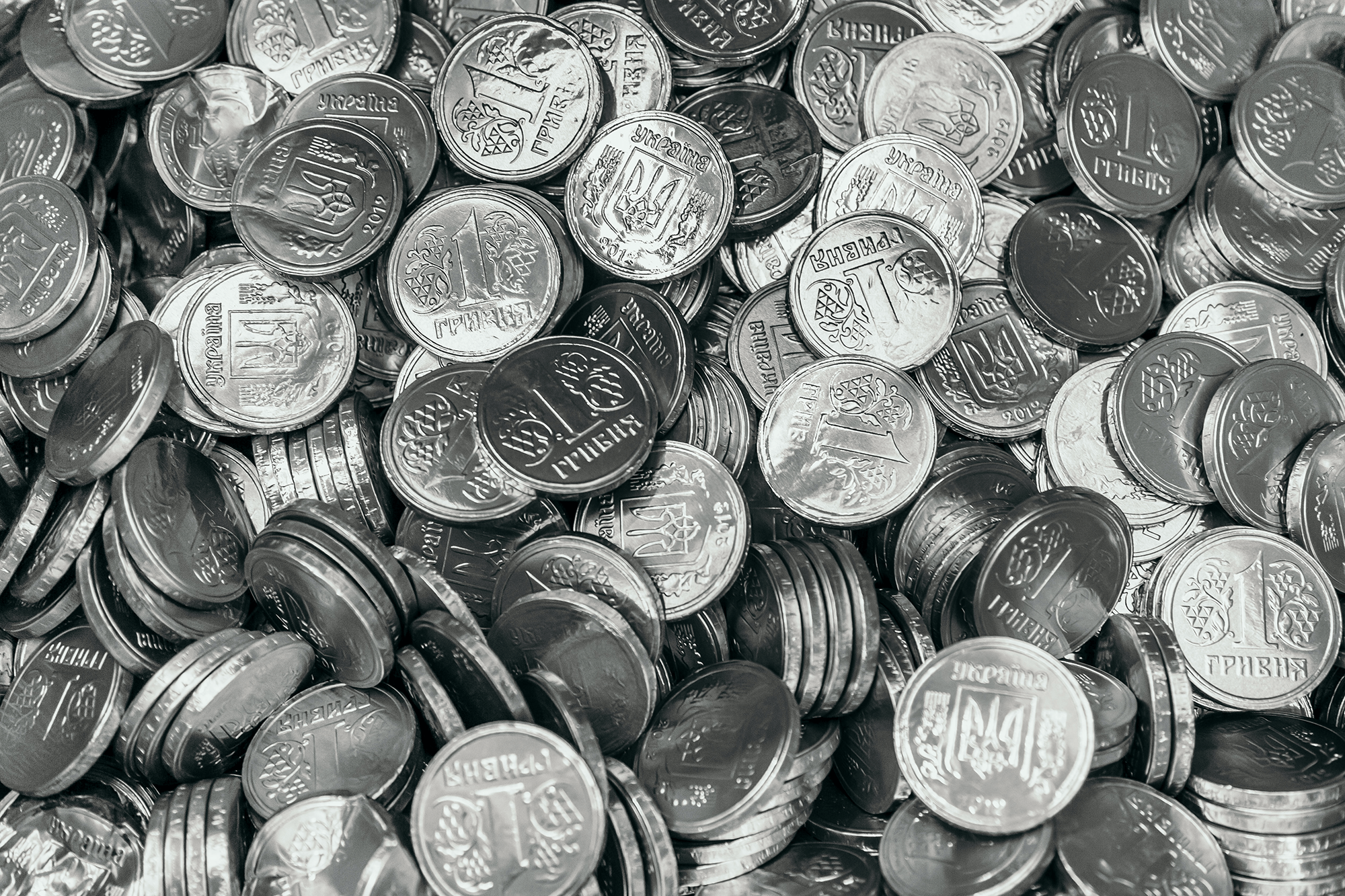
Packing Disks
Liquid crystals, bottles of pills, and hoppers of grains can all involve disk-shaped particles. To better understand how disks pack together, researchers studied how disks in a box orient themselves after shaking. They used MRI to observe the disks’ interior packing.

These reconstructions show the packing found in the experiment. The disks are color-coded by orientation; horizontal disks are redder and vertical ones are bluer. Initially, the packing has many horizontal disks (left), but after shaking, the disks get more compacted (right). The disks form short stacks that are randomly oriented. This increases the overall density but the random orientations reduce the total alignment of disks. The team found that shaking increases the disks’ density, but that increase does not come from disks orienting in the same direction. Instead, the disks form short stacks of similarly-oriented disks. The stacks themselves took on many different orientations, which reduced the system’s overall alignment in orientation. (Image credit: coins – M. Blan, packing – Y. Ding et al.; research credit: Y. Ding et al.; via APS Physics)

“Aquakosmos”
Colorful chandeliers, passing spirits, sprouting mushrooms, and fountains of falling ink appear in Christopher Dormoy’s “Aquakosmos.” Driven by the slight density difference between ink and water, many of these elaborate shapes result from the Rayleigh-Taylor instability. Anytime you see mushroom-like plumes and chandelier-like splitting vortex rings, there’s probably a Rayleigh-Taylor instability behind it. Check out the full video above, and, if you want to give this kind of flow visualization a try yourself, a glass of water and vial of food coloring is a great place to start. (Video and image credit: C. Dormoy)

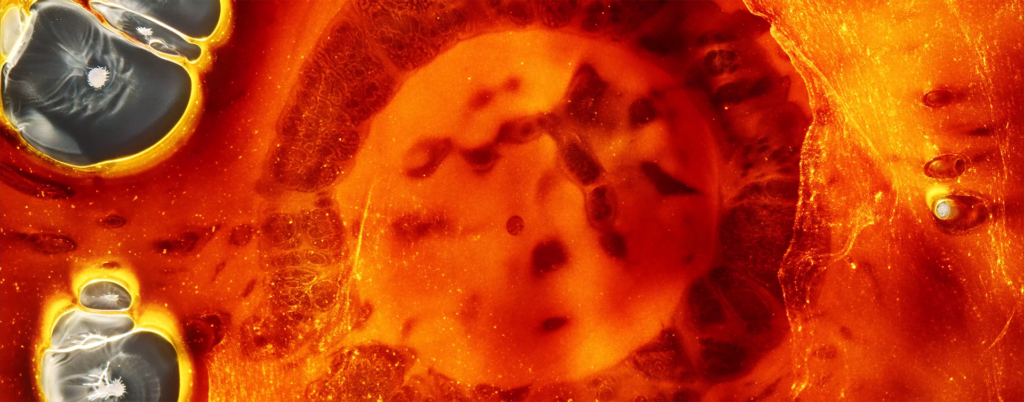
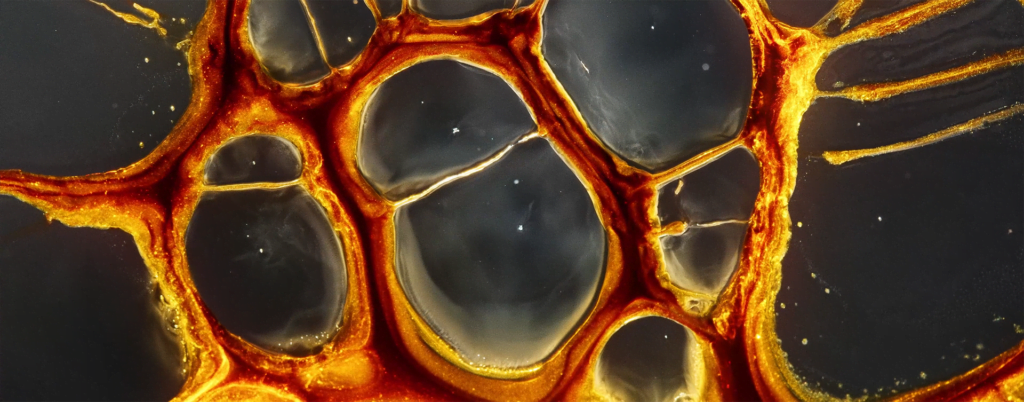
“Space Iris”
Ruslan Khasanov’s “Space Iris” explores the similarities between nebulae and eyes. Made entirely with common fluids like paint, soap, and alcohol, the film shows off the gorgeous possibilities of surface-tension- and density-driven instabilities. Marangoni flows abound! I even see some hints of solutal convection, perhaps? (Video and image credit: R. Khasanov; via Colossal)