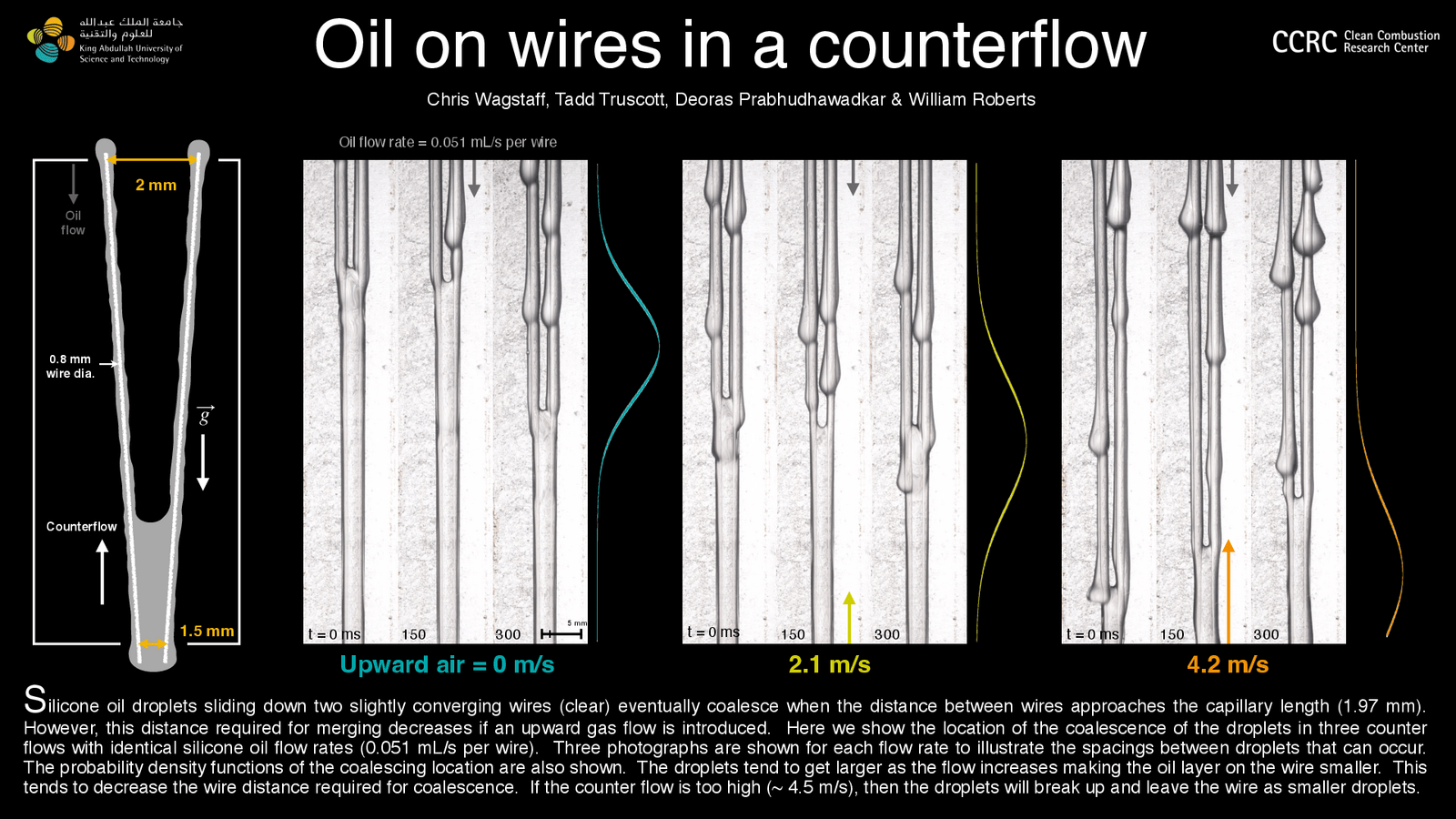
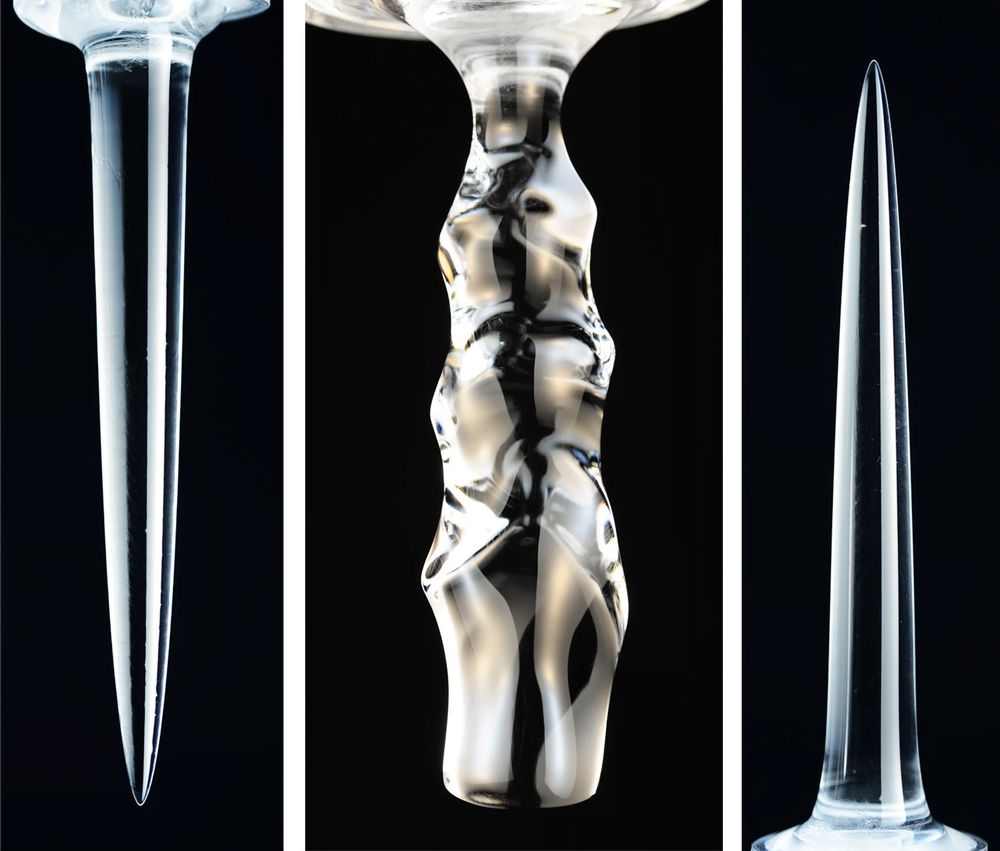
In nature and industry, swarms of bubbles* often encounter turbulence in their surrounding fluid. To study this situation, researchers used numerical simulation to observe bubbles across a range of density, viscosity, and surface tension values relative to their surroundings. They found that density differences between the two fluids made negligible changes to the way bubbles broke or coalesced.
In contrast, viscosity played a much larger role. More viscous bubbles were less likely to deform and break, thanks to their increased rigidity. When looking at small deformations along the bubble interface, both density and viscosity had noticeable effects. With increasing bubble density, they observed more dimples on the interface; increasing the viscosity had the opposite effect, making the bubbles smoother. (Image credit: Z. Borojevic; research credit: F. Mangani et al.)
*We usually think of bubbles as air or another gas contained within a liquid. But this study’s authors use the term “bubble” more broadly to mean any coherent bits of fluid in a different surrounding fluid. Colloquially, this means their results apply to both bubbles and drops.