Hot exhaust gases churn in the wake of this climbing B-1B Lancer. The high temperature of the exhaust changes the density and, thus, the refractive index of the gases relative to the atmosphere. Light traveling through the exhaust gets distorted, making the highly turbulent flow visible to the human eye. Note how the four individual engine exhaust plumes quickly combine into one indistinguishable wake. This is typical for turbulence; it’s hard to track where any given fluctuations originally came from. The airplane’s wingtip vortices are just visible as well, if you look closely. (Image credit: T. Rogoway; submitted by Mark S.)
Search results for: “density”

Twisting to Pack
One conventional method for packing granular materials is to tap them repeatedly, but a new study suggests that twisting is a faster method. Researchers poured thousands of dice into a cylinder, then twisted the container back and forth. When the acceleration caused by the change in direction exceeded a threshold value, the dice worked their way into ordered layers of concentric rings over ~10,000 cycles. Reaching this maximum packing density through tapping requires a specially designed method where the tapping characteristics change over time. With twisting, shear forces transmitted from the walls of the container tend to align the flat surfaces of the dice, providing an efficient method of ordering if the acceleration is large enough. The researchers hope methods like this may be useful where tapping works poorly, such as in microgravity. (Image and research credit: K. Asencio et al.; via APS Physics; submitted by Kam-Yung Soh)

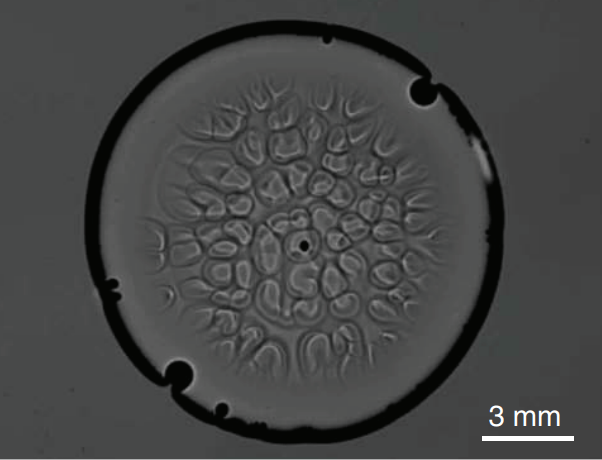
Layered Latte Physics
Latte lovers may be familiar with the layered latte, a beverage with distinctive horizontal layers mixing espresso and milk, but you may not have taken the time to wonder how these layers form. Like many layering phenomena in our oceans, the layered latte is the result of double-diffusive convection. This means that there are two variables that both affect density in the fluid mixture and that they act at different rates.
In the latte, those factors are 1) the different densities of the milk and espresso and 2) density changes caused as the latte cools to room temperature. A layered latte forms when the lighter espresso is poured into denser milk. If it’s poured quickly enough, the momentum of the pour forces some of the espresso down into the milk, despite the buoyant force that tries to keep the espresso on top. So that initial pour sets up a density gradient that runs from pure espresso at the top to pure milk on the bottom, with varying mixtures of the two in between.
The distinct layers won’t form until the latte begins cooling off. Along the walls of the container, heat is lost more quickly, causing fluid to cool and start sinking. But a specific bit of fluid can only sink until the fluid surrounding it is the same density. That can carry a cooler bit of latte to the bottom of a layer, but not into the denser layer below. At this point, our bit of latte moves inward, starts to warm up, and circulates up through the center of its layer. As when it sank, the fluid can only move up until it encounters a layer with equal or lesser density, at which point it must move horizontally instead. This thermal convection, combined with the density gradient formed by the initial pour, sets up the distinctive layers of the latte. The layers are quite stable – neither gentle stirring nor taking a sip will disrupt them for long – provided the drink remains warmer than the surrounding air. (Image credits: kopeattugu/Instagram, N. Xue et al.; research credit: N. Xue et al.; via NYTimes; submitted by Kam-Yung Soh)

Oil Splatters
Most cooks have experienced the unpleasantness of getting splattered with hot oil while cooking. Here’s a closer look at what’s actually going on. The pan is covered by a thin layer of hot olive oil. Whenever a water drop gets added – from, say, those freshly washed greens you’re trying to saute – it sinks through the oil due to its greater density. Surrounded by hot oil and/or pan, the water heats up and vaporizes with a sudden expansion. This throws the overlying oil upward, creating long jets of hot oil that break into flying droplets. These are what actually hit you. This is a small-scale demonstration, but it gets at the heart of why you don’t throw water on an oil fire. (Image credit: C. Kalelkar and S. Paul, source)
Convection Without Heat
Glycerol is a sweet, highly viscous fluid that’s very good at absorbing moisture from the ambient air. That’s why a drop of pure glycerol in laboratory conditions quickly develops convection cells – even when upside-down, as shown above. This is not the picture of Bénard-Marangoni convection we’re used to. There’s no temperature or density change involved; in fact, there’s no buoyancy involved at all! This convection is driven entirely by surface tension. As glycerol at the surface absorbs moisture, its surface tension decreases. This generates flow from the center of a cell toward its exterior, where the surface tension is higher. Conservation of mass, also known as continuity, requires that fresh, undiluted glycerol get pulled up in the wake of this flow. It, too, absorbs moisture and the process continues. (Image credit: S. Shin et al., pdf)

Lighting Engines
Combustion is complicated. You’ve ideally got turbulent flow, acoustic waves, and chemistry all happening at once. With so much going on, it’s a challenge to sort out the physics that makes one ignition attempt work while another fails. The animations here show a numerical simulation of combustion in a turbulent mixing layer. The grayscale indicates density contours of a hydrogen-air mixture. The top layer is moving left to right, and the lower layer moves right to left. This sets up some very turbulent mixing, visible in middle as multi-scale eddies turning over on one another.
Ignition starts near the center in each simulation, sending out a blast wave due to the sudden energy release. Flames are shown in yellow and red. As the flow catches fire, more blast waves appear and reflect. But while the combustion is sustained in the upper simulation, the flame is extinguished by turbulence in the lower one. This illustrates another challenge engineers face: turbulence is necessary to mix the fuel and oxidizer, but turbulence in the wrong place at the wrong time can put out an engine. (Image, research, and submission credit: J. Capecelatro, sources 1, 2)


Convection
Blue paint in alcohol forms an array of polygonal convection cells. We’re accustomed to associating convection with temperature differences; patterns like the one above are seen in hot cooking oil, cocoa, and even on Pluto. In all of those cases, temperature differences are a defining feature, but they are not the fundamental driver of the fluid behavior. The most important factors – both in those cases and the present one – are density and surface tension variations. Changing temperature affects both of these factors, which is why its so often seen in Benard-Marangoni convection.
For the paint-in-alcohol, density and surface tension differences are inherent to the two fluids. Because alcohol is volatile and evaporates quickly, its concentration is constantly changing, which in turn changes the local surface tension. Areas of higher surface tension pull on those of lower surface tension; this draws fluid from the center of each cell toward the perimeter. At the same time, alcohol evaporating at the surface changes the density of the fluid. As it loses alcohol and becomes denser, it sinks at the edges of the cell. Below the surface, it will absorb more alcohol, become lighter, and eventually rise at the cell center, continuing the convective process. (Image credit: Beauty of Science, source)

The Winds of Mars
The Martian atmosphere is scant compared to Earth’s, but its winds still sculpt and change the surface regularly. The average atmospheric pressure on Mars is only 0.6% of Earth’s, and the density is similarly low at 1.7% of Earth’s. Despite this thinness, Martian winds are still substantial enough to shift sands on a daily basis, as shown above. These two images were taken one Martian day apart, showing how sand ripples moved and how the Curiosity rover’s tracks can be quickly obscured. Part of the reason Mars’ scant atmosphere is still so good at moving sand is that Martian gravity is roughly one-third of ours; if the sand is lighter, it doesn’t take as much force to move! (Image credit: NASA/JPL-CALTECH/MSSS)

Shadows of Flow
In the latest Veritasium video, Derek demonstrates how to see gas motions that are normally invisible using a schlieren photography set-up. Schlieren techniques have been important in fluid dynamics for well over a century, and Derek’s set-up is one of the two most common ways to set up the technique. (The other method uses two collimating mirrors instead of a single spherical or parabolic one.) As explained in the video, the schlieren optical set-up is sensitive to small changes in the refractive index, making density changes or differences in a gas visible. This makes it possible to distinguish gases of different temperatures or compositions and even lets you see shock waves in supersonic flows. (Video and image credit: Veritasium; submitted by Paul)


Quad Copter Schlieren
Schlieren photography is a classic method of flow visualization that utilizes small variations in density (or temperature) to make otherwise unseen air motion visible. Because changing air’s density or temperature changes its index of refraction, variations in either quantity show up as dark and light regions. Here researchers use it to reveal some of the airflow around a small quadcopter, including the vortices that spiral off each propeller and help generate the lift necessary for take-off. The full video includes a couple of neat demos, including what happens when the blades are wet (shown below). In that case, the wingtip vortices are somewhat disrupted by strings of water droplets being flung off the blades by centrifugal force. Beautiful! (Video and image credit: K. Nolan et al., source; submitted by J. Stafford)


