Antlion larvae catch prey by digging conical pits in sand. The steep walls of the trap are near the angle of repose, the largest angle a granular material can maintain before grains slide down. When a hapless ant wanders into the trap, the antlion throws sand from the center of the pit, triggering a sandslide that carries the ant downward. The act of flinging sand also helps the antlion maintain the pit, correcting any disruptions to the pit’s steep sides caused by its flailing prey. (Image and research credit: S. Büsse et al.; via Science)
Category: Research

Uncovering Erosion Patterns
Gypsum and limestone cliffs sometimes form patterns of long, parallel grooves known as rillenkarren. Recent research shows that these patterns form when a thin layer of water flows over a dissolvable surface. As the running water picks up solute, its concentration increases, causing changes in the local hydrodynamics. What begins as a small perturbation in an otherwise flat surface grows into a groove with walls that eventually rise out of the water layer. At that point, the growth mechanism shifts because the flow is restricted to channels in the rock. (Image credit: Ymaup/Wikimedia Commons; research credit: A. Guérin et al.; via APS Physics; submitted by Kam-Yung Soh)

Hedgehogs Atop Waves
Since Michael Faraday, scientists have watched the curious patterns that form in a vibrating liquid. By adding floating particles to such a system, researchers have discovered spiky, hedgehog-like shapes that form near the surface. At low amplitudes, the surface patterns resemble the typical smooth rounded lobes one would expect, but as the wave amplitude increases, spikes form in the tracers, driven by the motion of the waves. (Image and research credit: H. Alarcón et al.; via APS Physics)

Stabilizing Foams
Bubbles in a pure liquid don’t last long, but with added surfactants or multiple miscible liquids, bubbles can form long-lasting foams. In soapy foams, surfactants provide the surface tension gradients necessary to keep the thin liquid layers between bubbles from popping. But what stabilizes a surfactant-free foam?
New work finds that foams in mixtures of two miscible fluids only form when the surface tension depends nonlinearly on the concentration of the component liquids. When this is true, thinning the wall between bubbles creates changes in surface tension that stabilize the barrier and keep it from popping.
In mixtures without this nonlinearity, foams just won’t form. The new results are valuable for manufacturing, where companies can avoid unintentional foams simply by careful selection of their fluids. (Image credit: G. Trovato; research credit: H. Tran et al.; via APS Physics; see also Ars Technica, submitted by Kam-Yung Soh)

Oil in Water
In the decade since the Deepwater Horizons oil spill, scientists have been working hard to understand the intricacies of how liquid and gaseous hydrocarbons behave underwater. The high pressures, low temperatures, and varying density of the surrounding ocean water all complicate the situation.
Released hydrocarbons form a plume made up of oil drops and gas bubbles of many sizes. Large drops and bubbles rise relatively quickly due to their buoyancy, so they remain confined to a relatively small area around the leak. Smaller drops are slower to rise and can instead get picked up by ocean currents, allowing them to spread. The smallest micro-droplets of oil hardly rise at all; instead they remained trapped in the water column, where currents can move them tens to hundreds of kilometers from their point of release. (Image and research credit: M. Boufadel et al.; via AGU Eos; submitted by Kam-Yung Soh)

Adjusting for Gusts
In flight, birds must adjust quickly to wind gusts or risk crashing. Research shows that the structure of birds’ wings enables them to respond faster than their brains can. The wings essentially act like a suspension system, with the shoulder joint allowing them to lift rapidly in response to vertical gusts. This motion keeps the bird’s head and torso steady, so they can focus on more complex tasks like landing, obstacle avoidance, and prey capture. (Image and research credit: J. Cheney et al.; submitted by Kam-Yung Soh)

Lake Stars
As snow-covered frozen lakes melt, stars appear on their surface. These lake stars form around holes in the ice where (relatively) warm water seeps up into the slush layer. The stars form through a competition between thermal effects and flow through the porous snow. Researchers have built mathematical models that capture the first-order effects, like predicting the number of arms a star will form. (Image and research credit: V. Tsai and J. Wettlaufer; submitted by keeonn)

Dual Structure of Water
Water is so ubiquitous in our lives that we rarely recognize just how strange it is. For example, when pure liquid water is supercooled well below its freezing temperature, it takes on not one but two molecular arrangements, one of which is high-density and one of which is low-density. Theory had posited this configuration for some time, but only recently has experimental evidence supported it.
The experimental challenge was water’s rapid crystallization in the temperature region of interest. Any time water was held at those temperatures in order to study it, it would crystallize before researchers could make their observations. To get around this, a team studied extremely thin layers of water which they heated with a laser before rapidly cooling. By repeating this heating-and-cooling cycle many times, they were able to measure water properties that only make sense if it conforms to the two-density theory. (Image credit: T. Holland/Pacific Northwest National Laboratory; research credit: L. Kringle et al.; via Science News; submitted by Kam-Yung Soh)

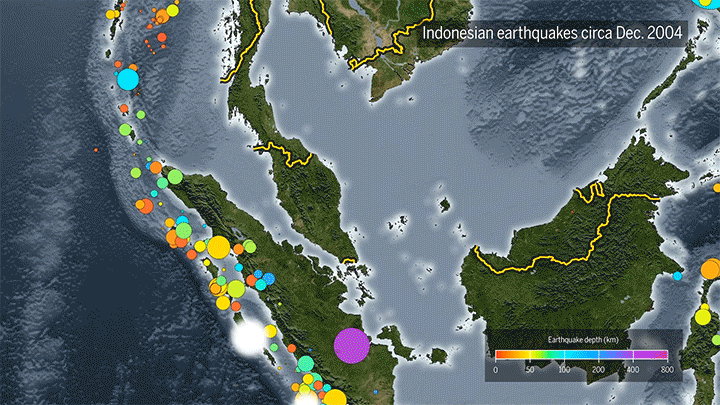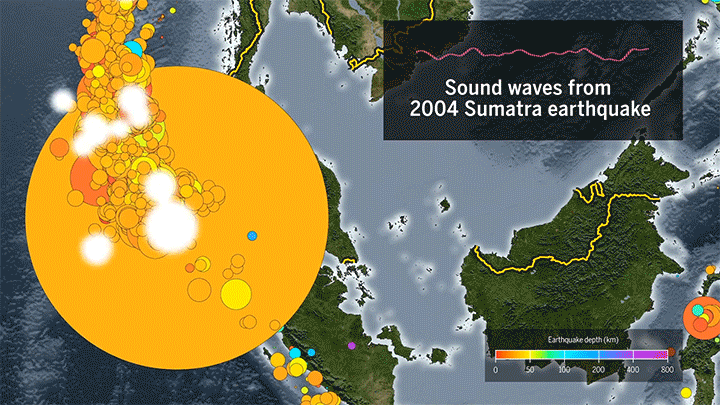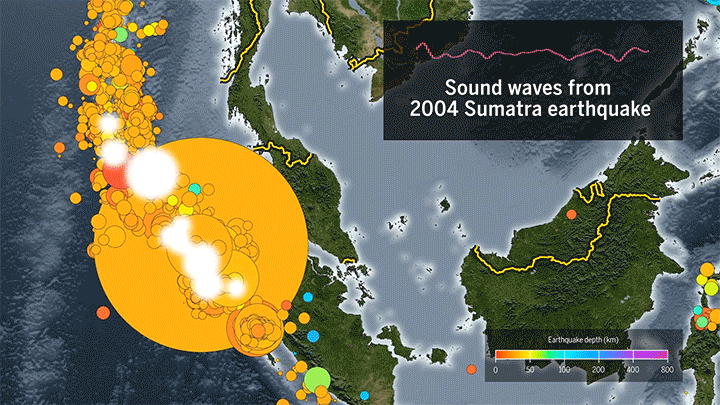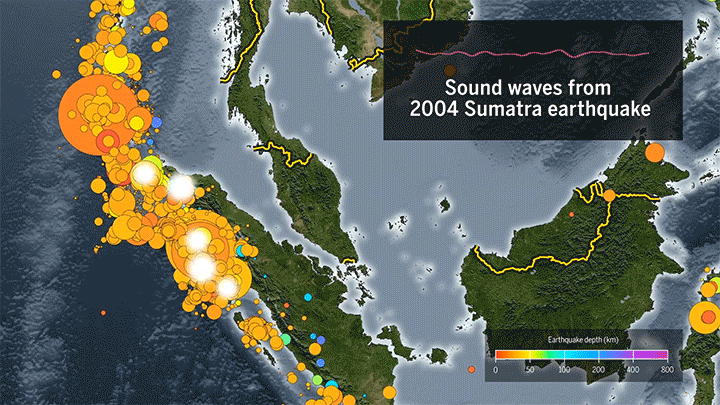
Seismic Events Reveal Ocean Temperatures
Decades ago, researchers proposed sending sound waves through the ocean to measure its temperature. Although the technique worked, it ran into noise pollution issues, but now it’s back, using naturally-occurring seismic events as the sound source.
When fault lines shift, they generate seismic waves that travel through the ocean as sound. When they reach a land mass, the waves get converted back into seismic energy that’s then picked up by a receiver. Knowing the distance from the source to the receiver and the time necessary for the wave to travel, scientists can then determine the average temperature of the water based on the speed of sound.
The technique can track temperature changes down to thousandths of a degree. Based on more than a decade of seismic data from the Indian Ocean, researchers found almost double the temperature increase measured by a different sensor network. (Image and video credit: Science; research credit: W. Wu et al.; submitted by Kam-Yung Soh)

Rocket Yeast
Usually, microbial colonies are grown on a solid substrate, but what happens when they grow on a liquid surface? That’s the question explored in this Gallery of Fluid Motion video featuring colonies of brewer’s yeast on various liquid substrates. When the viscosity of the liquid is low enough, the colony actually gets pulled apart (Image 2). This behavior is driven by a convective flow in the liquid caused by the colony’s own growth. As the yeast grow, they deplete nearby sugar, creating a density gradient that triggers convection beneath the colony. (Image, video, and research credit: S. Atis et al.)