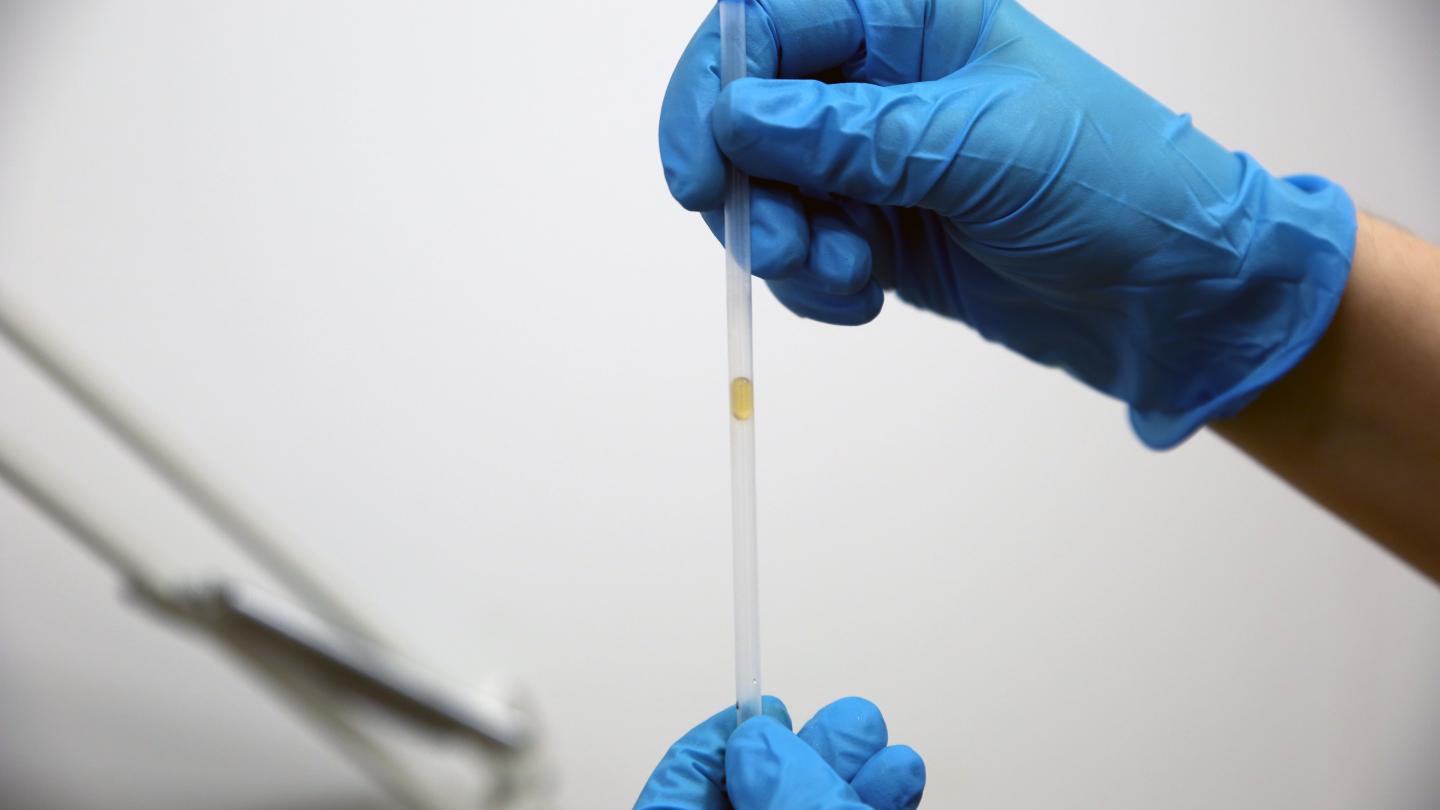
With its high viscosity, no one would ever pick honey to beat water in a race. But a new study shows there’s at least one circumstance where honey wins: inside a narrow, superhydrophobic tube with one or both ends closed. Inside these specially coated tubes a narrow cushion of air stays between the drop and the wall, reducing friction and increasing flow speed for both fluids.
But when one or both ends of the tube are blocked, the drops can only move when air squeezes past. In less viscous fluids, like water, the researchers found rapid internal flows inside the drop. These flows pressed the surface of the drop outward, reducing the air cushion and making it harder for air to squeeze past so that the drop could flow. In contrast, honey showed very little internal flow and so was able to flow through the tubes ten times faster than water! (Image and research credit: M. Vuckovac et al.; via Physics World; submitted by Kam-Yung Soh)