Here a ferrofluid climbs a spiral steel structure sitting on an electromagnet. Magnetic field lines emanating from the sculpture’s edges tend to push the ferrofluid out into long spikes–part of the normal field instability–but surface tension resists. The short, somewhat squat spikes we see are the balance struck between these opposing forces. Though known for their wild appearance, ferrofluids appear many in common applications, including hard drives, speakers, and MRI contrast agents. Researchers have also recently suggested they might help understand the behavior of the multiverse. (Photo credit: P. Davis et al.)
Tag: surface tension

Surface Tension in Action
Surface tension creates a glassy, smooth layer of water over U.S. swimmer Tyler Clary the instant before he surfaces as he competes in the backstroke. Surface tension arises from intermolecular forces between water molecules. In the bulk of the liquid, any given water molecule is being pulled on in every direction by the surrounding molecules, which results in zero net force. At the surface, however, molecules only experience forces from those to the side and below them. As a result, these molecules are pulled inwards, forcing the liquid to take on a form with minimal area. (Photo credit: Getty Images; submitted by drhawkins)

Droplet Springs
Prior to reaching terminal velocity, a falling droplet typically oscillates between a prolate shape (like an American football about to be kicked) and an oblate one (like that same football when thrown or carried). As explained by Minute Laboratory, this oscillation behaves very similarly to a mass on a spring. For a spring/mass system, the frequency of oscillation is related to the spring’s stiffness; for the falling droplet, it is instead governed by surface tension. If only high schools had high-speed cameras, this would make a fantastic fluids lab experiment! (Video credit: Minute Laboratory; submitted by Pascal W.)
FYFD is sporting a new look and new about and resources pages for those who want more fluidsy goodness.

The Evolution of Icicles
The time-lapse video above shows the growth of icicles of various compositions under laboratory conditions. Many icicles in nature exhibit a rippling effect in their shape, which some theories attribute to an effect of lower surface tension in some liquids. Here researchers show the icicle growth of three liquids: pure distilled water, and water with two concentrations of dissolved salt. They found that lowering the surface tension of the freezing liquid with non-ionic surfactants (i.e. not salt) did not produce ripples, but that dissolved ionic impurities like salt strongly affected the growth of ripples. They posit that this may be due to constitutional supercooling, in which growth of the solid-liquid interface is destabilized by the preferential concentration of impurities near the interface. (Video credit: A. S. Chen and S. Morris)

Inksplosion
Artist Pery Burge utilizes surface tension driven flows created with inks and water for much of her work. As mesmerizing as this is in still-life, it is more lovely still to see it develop and evolve in motion. The explosive outward motion of the ink is driven by the addition of a liquid with a lower surface tension than the ink/water mixtures. This is known as the Marangoni effect. You can observe it yourself using a plate of milk and food coloring into which you drop a tiny bit of dish soap. (The experiment works best with milk with some fat content.) Or, like the artist herself, you can experiment with other fluids you have on-hand! For more of Bruge’s work, see her website. (Video credit: Pery Bruge)

Bouncing and Break-Up
In the collage above, successive frames showing the bouncing and break-up of liquid droplets impacting a solid inclined surface coated with a thin layer of high-viscosity fluid have been superposed. This allows one to see the trajectory and deformation of the original droplet as well as its daughter droplets. The impacts vary by Weber number, a dimensionless parameter used to compare the effects of a droplet’s inertia to its surface tension. A larger Weber number indicates inertial dominance, and the Weber number increases from 1.7 in (a) to 15.3 in (d). In the case of (a), the impact of the droplet is such that the droplet does not merge with the layer of fluid on the surface, so the complete droplet rebounds. In cases (b)-(d), there is partial merger between the initial droplet and the fluid layer. The impact flattens the original droplet into a pancake-like layer, which rebounds in a Worthington jet before ejecting several smaller droplets. For more, see Gilet and Bush 2012. (Photo credit: T. Gilet and J. W. M. Bush)
Reader Question: Snow from Boiling Water?
[original media no longer available]
Reader kylewpppd asks:
Have you seen the post of a man in Siberia throwing boiling water off of his balcony? Can you provide a better explanation of what’s going on?
As you can see in the video (and in many similar examples on YouTube), tossing near boiling water into extremely cold air results in an instant snowstorm. Several effects are going on here. The first thing to understand is how heat is transferred between objects or fluids of differing temperatures. The rate at which heat is transferred depends on the temperature difference between the air and the water; the larger that temperature difference is the faster heat is transferred. However, as that temperature difference decreases, so does the rate of heat transfer. So even though hot water will initially lose heat very quickly to its surroundings, water that is initially cold will still reach equilibrium with the cold air faster. Therefore, all things being equal, hot water does not freeze faster than cold water, as one might suspect from the video.
The key to the hot water’s fast-freeze here is not just the large temperature difference, though. It’s the fact that the water is being tossed. When the water leaves the pot, it tends to break up into droplets, which quickly increases the surface area exposed to the cold air, and the rate of heat transfer depends on surface area as well! A smaller droplet will also freeze much more quickly than a larger droplet.
What would happen if room temperature water were used instead of boiling water? In all likelihood, a big cold bunch of water would hit the ground. Why? It turns out that both the viscosity and the surface tension of water decrease with increasing temperature. This means that a pot of hot water will tend to break into smaller droplets when tossed than the cold water would. Smaller droplets means less mass to freeze per droplet and a larger surface area (adding up all the surface area of all the droplets) exposed. Hence, faster freezing!

Tears of Wine
Physicist Richard Feynman once famously ended a lecture by describing how the whole universe can be found in a glass of wine. And there is certainly plenty of fluid dynamics in one. In the photo above, we see in the shadows how a film of wine drips down into the main pool below. This effect is known by many names, including tears of wine and wine legs; it can also be found in other high alcohol content beverages. Several effects are at play. Capillary action, the same effect that allows plants to draw water up from their roots, helps the wine flow up the wall of the glass. At the same time, the alcohol in this wine film evaporates faster than the water, raising the surface tension of the wine film relative to the main pool of wine below. Because of this gradient in surface tension, the wine will tend to flow up the walls of the glass away from the area of lower surface tension. This Marangoni effect also helps draw the wine upward. When the weight of the wine film is too great for capillary action and surface tension to hold it in place, droplets of wine–the legs themselves–flow back downward. (Photo credit: Greg Emel)

Bouncing Jet
For the right flow speeds and incidence angles, a jet of Newtonian fluid can bounce off the surface of a bath of the same fluid. This is shown in the photo above with a laser incorporated in the jet to show its integrity throughout the bounce. The walls of the jet direct the laser much the way an optical fiber does. The jet stays separated from the bath by a thin layer of air, which is constantly replenished by the air being entrained by the flowing jet. The rebound is a result of the surface tension of the bath providing force for the bounce. (Photo credit: T. Lockhart et al.)

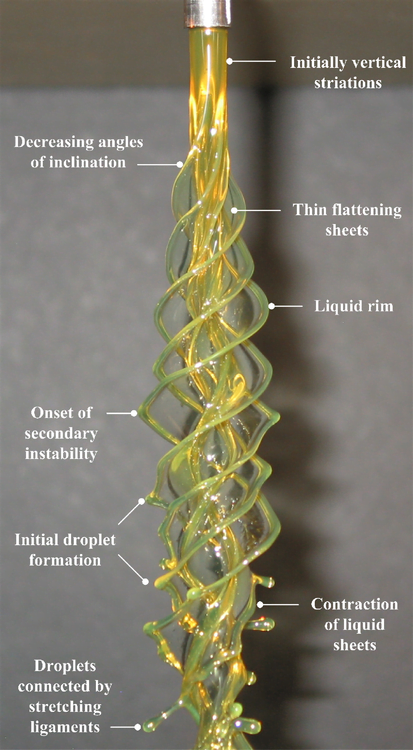
Swirling Jets
In fluid dynamics, we like to classify flows as laminar–smooth and orderly–or turbulent–chaotic and seemingly random–but rarely is any given flow one or the other. Many flows start out laminar and then transition to turbulence. Often this is due to the introduction of a tiny perturbation which grows due to the flow’s instability and ultimately provokes transition. An instability can typically take more than one form in a given flow, based on the characteristic lengths, velocities, etc. of the flow, and we classify these as instability modes. In the case of the vertical rotating viscous liquid jet shown above, the rotation rate separates one mode (n) from another. As the mode and rotation rate increase, the shape assumed by the rotating liquid becomes more complicated. Within each of these columns, though, we can also observe the transition process. Key features are labeled in the still photograph of the n=4 mode shown below. Initially, the column is smooth and uniform, then small vertical striations appear, developing into sheets that wrap around the jet. But this shape is also unstable and a secondary instability forms on the liquid rim, which causes the formation of droplets that stretch outward on ligaments. Ultimately, these droplets will overcome the surface tension holding them to the jet and the flow will atomize. (Video and photo credits: J. P. Kubitschek and P. D. Weidman)