A drop of silicone oil falling through a liquid with lower surface tension distorts into multiple vortex rings connected by thin films. This behavior is caused by the interaction between viscous and capillary forces and is observable for only a narrow range of oil viscosities. (Photo credit: A. Felce and T. Cubaud)
Tag: surface tension

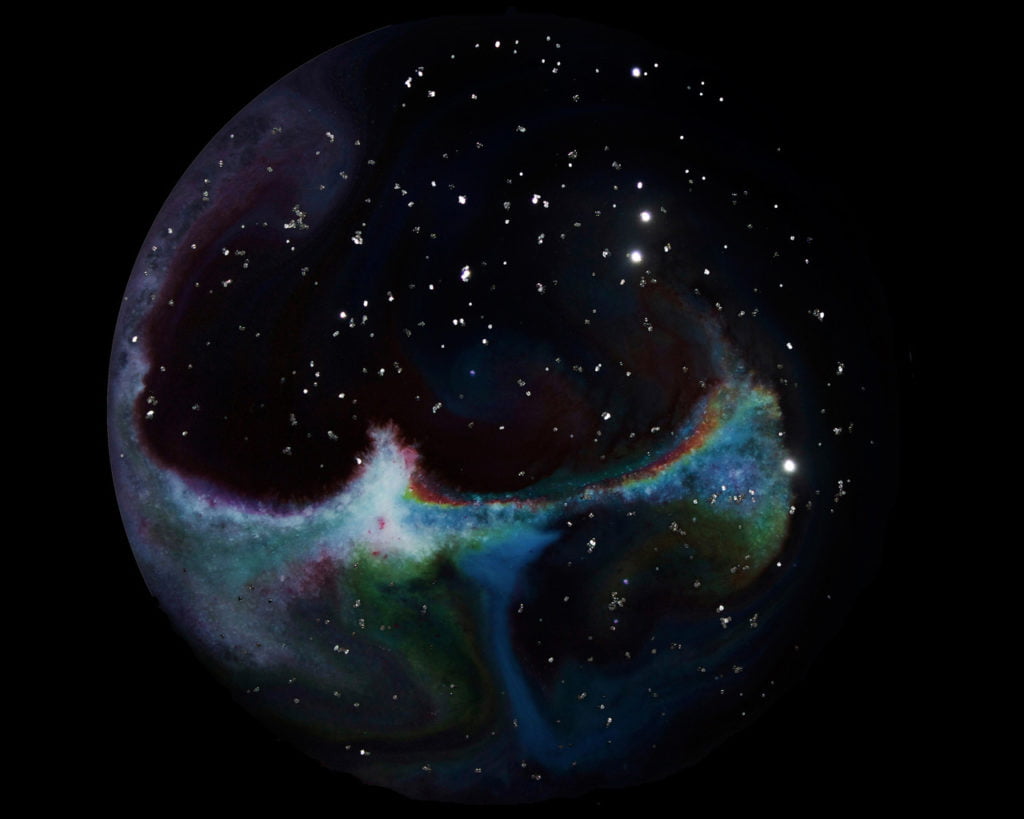
Homemade Astronomy
Artist Julia Cuddy uses liquids, soaps, and glitter to create photographs that replicate the look of deep space astronomy. By adding soap to the dyes, she uses Marangoni effects to drive surface tension instabilities that cause swirling colors and motions reminiscent of galaxies and nebulae. Although I’ve seen fluid dynamics used in art before, this may be one of the cleverest usages I’ve seen! (Photo credits: Julia Cuddy)

Astro Puffs
Microgravity continues to be a fascinating playground for observing surface tension effects on the macroscale without pesky gravity getting in the way. Here astronaut Don Pettit has created a sphere of water, which he then strikes with a jet of air from a syringe. Initially, the momentum from the jet of air creates a sharp cavity in the water, which rebounds into a jet of water that ejects one or more satellite drops. Surface waves and inertial waves (inside the water sphere) reflect back and forth until the fluid comes to rest as a sphere once more. Note how similar the behavior is to the pinch-off of a water column. Both effects are dominated by surface tension, but on Earth we can only see this behavior with extremely small droplets and high-speed cameras! (Video credit: Don Pettit, Science Off the Sphere)

Pinch-Off
This high-speed video reveals a fascinating bit of kitchen sink physics. When a water droplet pinches off from the nozzle, the thin filament of fluid that connected the droplet to the water on the nozzle often breaks off as well. Surface tension snaps the filament together into a sphere, causing wild oscillations and even ejection of microjets in the tiny satellite droplet. (Video from S. Thoroddsen et al. 2008’s Annual Review)

Soap Film Loops
Here’s a fun demonstration of the effects of surface tension. If a loop of thread is dropped onto a soap film, as shown above, popping the soap film inside the thread will pull the thread into a circle. This is because the surface tension of the soap film outside the thread is reacting to the sudden loss of the balancing force exerted by surface tension inside the thread loop. Surface tension arises from intermolecular forces in a fluid. Because those forces are in balance except along the interface of a fluid–where the fluid molecules are not completely surrounded by identical molecules–there is a net force exerted at the surface.

Labyrinth
A labyrinthine pattern forms in this timelapse video of a multiphase flow in a Hele-Shaw cell. Initially glass beads are suspended in a glycerol-water solution between parallel glass plates with a central hole. Then the fluid is slowly drained over the course of 3 days at a rate so slow that viscous forces in the fluid are negligible. As the fluid drains, fingers of air invade the disk, pushing the beads together. The system is governed by competition between two main forces: surface tension and friction. Narrow fingers gather fewer grains and therefore encounter less friction, but the higher curvature at their tips produces larger capillary forces. The opposite is true of broader fingers. Also interesting to note is the similarity of the final pattern to those seen in confined ferrofluids. (Video credit and submission: B. Sandnes et al. For more, see B. Sandes et al.)

Soap Bubbles Bursting
To the human eye, the burst of a soap bubble appears complete and instantaneous, but high-speed video reveals the directionality of the process. Surface tension is responsible for the spherical shape of the bubble, and, when the bubble is pierced, surface tension is broken, causing the soap film that was the bubble to contract like elastic that’s been stretched and released. Droplets of liquid fly out from the edges of the sheet until it atomizes completely.

Breaking Water with Sound
Previously we saw how vibration could atomize a water droplet, breaking it into a spray of finer droplets. Here astronaut Don Pettit shows us what the process looks like in microgravity using some speakers and large water droplets. At low frequencies the water displays large wavelength capillary waves and vertical vibrations. Higher frequencies–like the earthbound experiment on much smaller droplets–cause fine droplets to eject from the main drop when surface tension can no longer overcome their kinetic energy. (submitted by aggieastronaut, jshoer and Jason C)
(Source: /)
Surface Tension Floats Coins
Surface tension arises from intermolecular forces along the interface of a fluid, but despite its molecular origins, it can have some substantial macroscopic effects. Here researchers demonstrate how surface tension can hold up metal coins that would otherwise sink. Moreover, when multiple coins are set on the surface of the water, surface tension draws them together into a closely packed array because it reduces the surface energy by creating a single large well instead of many small ones. This is the same reason that your Cheerios tend to clump together on the surface of your milk when you’re eating breakfast! (Video credit: Lawrence Berkeley National Lab)

Bursting Bubbles
Sometimes bursting one bubble just leads to more bubbles. This high-speed video shows how popping a bubble sitting on a fluid surface can lead to a ring of daughter bubbles. When the surface of the bubble is ruptured, filaments of the liquid that made up the surface are drawn back toward the pool by surface tension, trapping small pockets of the air that had been inside the bubble. A dimple forms on the surface and rebounds as a jet that lacks the kinetic energy to eject droplets. Watch as the jet returns to the interface, and you will notice the tiny bubbles around it. At 56 ms, one of the daughter bubbles on the left bursts. See Nature for more. (Video credit: J. Bird et al)