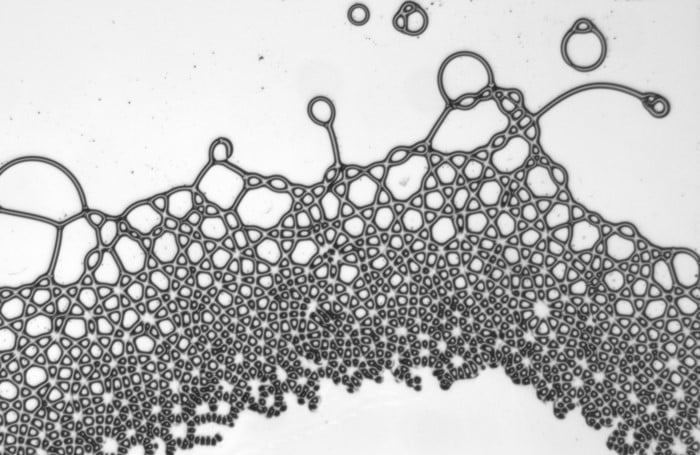
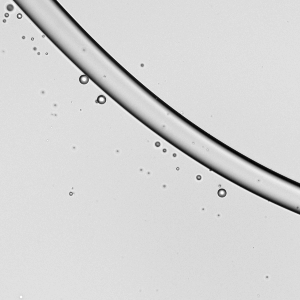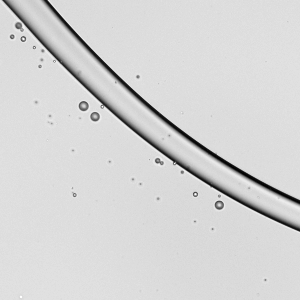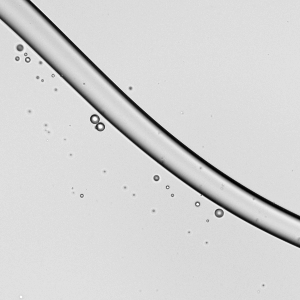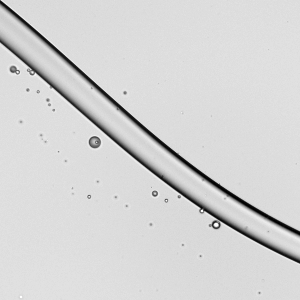
As wearing face masks for long periods has become more typical, you may have wondered whether a soggy mask offers less protection. All masks — cloth, surgical, and N-95s — get moist from their wearer’s breath. A recent study indicates this isn’t a cause for alarm, though.
Researchers looked at how relatively high-speed droplets (like those from a cough or sneeze) impact dry and wet masks. These high-speed droplets can break into smaller droplets upon impact with a mask layer. The more layers a mask has, the fewer droplets make it through. But even for single-layer masks*, a moistened mask layer lets fewer droplets through. So you don’t have to worry if it’s a little humid in there. Your mask is still working! (Image credit: top – V. Davidova, other – S. Bagchi et al.; research credit: S. Bagchi et al.; via APS Physics)
* To be clear, you should be wearing masks that are more than a single layer thick. Personally, I’m still only going into indoor public spaces in an N-95 at this point.