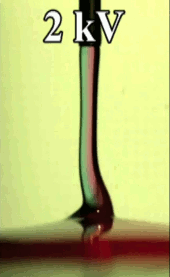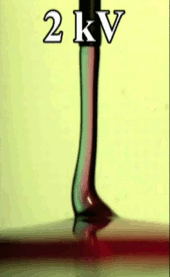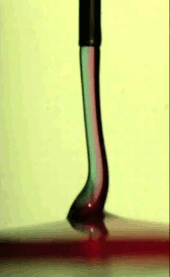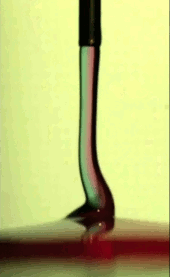Every year Chicago dyes part of its river green to celebrate St. Patrick’s Day. This timelapse video gives a great view of the 2016 dyeing. If you watch closely, you’ll see that what’s being put in the river isn’t originally green. It’s actually an orange powder being distributed through flour sifters by the men on the boat. The exact formula is secret, but the dye is considered environmentally safe. To mix up the dye, a chase boat follows the dye boat, using its motor and wake structure to help add some turbulence to the river. It takes several passes to get the water uniformly green, but it requires a remarkably small amount of dye to do so, only about a paint can’s worth. So enjoy a little fluid dynamics today with your festivities! (Or, if you prefer to celebrate a different sort of fluid dynamics today, allow me to offer you the physics of Guinness.) (Video credit: Chris B Photo)
Tag: mixing

Electric Coiling
A falling jet of viscous fluid–like honey or syrup–will often coil. This happens when the jet falls quickly enough that it gets skinnier and buckles near the impact point. Triggering this coiling typically requires a jet to drop many centimeters before it will buckle. In many manufacturing situations, though, one might want a fluid to coil after a shorter drop, and that’s possible if one applies an electric field! Charging the fluid and applying an electric field accelerates the falling jet and induces coiling in a controllable manner.
An especially neat application for this technique is mixing two viscous fluids. If you’ve ever tried to mix, say, food coloring into corn syrup, you’ve probably discovered how tough it is to mix viscous substances. But by feeding two viscous fluids through a nozzle and coiling the resulting jet, researchers found that they could create a pool with concentric rings of the two liquids (see Figure C above). If you make the jet coil a lot, the space between rings becomes very small, meaning that very little molecular motion is necessary to finish mixing the fluids. (Image credits: T. Kong et al., source; via KeSimpulan)

A Toast!
When you lift a glass of champagne or sparkling wine at midnight tonight, your nose and mouth will be greeted by a plethora of aromas, flavors, and sensations propagated by the tiny bubbles in the drink. Carbon dioxide dissolved in the wine gathers in a stream of tiny bubbles that rise at the center of the glass. (The bubbles form at the center because champagne glasses are often etched in a ring there to provide nucleation points where the bubbles can grow.) This stream of rising bubbles generates vortical motion in the glass that helps carry the carbon dioxide to the surface, where it is released when the bubbles burst. In the tall, thin champagne flute these vortices mix the entire contents of the glass, but, in a wider coupe, the vortices are confined to the center, leaving a stiller region along the glass’s edges. For those who find that a freshly poured flute of champagne stings their noses–a side effect of the high gaseous carbon dioxide concentration just after decanting–the wider coupe lowers the concentration at the glass’s lip and may provide a more pleasant experience for toasting the new year. (Image credit: F. Beaumont et al.)

Viscous Droplet Impacts
Viscosity can have a notable effect on droplet impacts. This poster demonstrates with snapshots from three droplet impacts. The blue drops are dyed water, and the red ones are a more viscous water-glycerol mixture. When the two water droplets impact, a skirt forms between them, then spreads outward into a sheet with a thicker, uneven rim before retracting. The second row shows a water droplet impacting a water-glycerol droplet. The less viscous water droplet deforms faster, wrapping around and mixing into the other drop before rebounding in a jet. The last row switches the impacts, with the more viscous drop falling onto the water. As in the previous case, the water deforms faster than the water-glycerol. The two mix during spreading and rebound slower. In the last timestep shown, the droplet is still contracting, but it does rebound as a jet thereafter. (Image credit: T. Fanning et al.)

Phytoplankton Flow Viz
Nutrient-rich waters off Patagonia in South America blossom with phytoplankton in this satellite image. When present in large quantities, these microscopic photosynthesizers lend a green hue to the water. They act as seed particles in the flow, highlighting the currents and flow that carry them. If you check out the full resolution version of the photo, you can admire the rich detail in the whorls of ocean mixing. There even seem to be Kelvin-Helmholtz-like instabilities creating trains of vortices along the interface between separate bands. (Photo credit: NASA/ASU; via SpaceRef; submitted by jshoer)

The Rayleigh-Taylor Instability
What’s this? An FYFD video?! Yes, at long last, I’ve begun filming some videos of my own. This first one takes a look at the Rayleigh-Taylor instability and all that action that goes on in your coffee cup. I hope to bring you more FYFD-produced videos in the future, including some videos from the American Physical Society Division of Fluid Dynamics conference in San Francisco next week. What kind of topics would you guys be interested in for the future? (Video credit: N. Sharp)

“Milky WaY”
Photographer Paulo Stagnaro uses milk and food coloring in his series “Milky WaY”. Despite the simple ingredients, the photos illustrate the enormous variety of shape and form in fluid dynamics. Surface tension, diffusion, and intentional mixing create abstract and ephemeral portraits of fluid motion. For similar work, see Pery Bruge’s art or just try browsing through FYFD’s “fluids as art” tag for more examples of science and art intersecting. (Photo credit: P. Stagnaro; submitted by Stephanie M.)

Phytoplankton Bloom
In satellite imagery the blue and green whorls of massive phytoplankton blooms stand out against the ocean backdrop. These microscopic organisms are part of a delicate predator-prey balance and can be very sensitive to nutrient concentrations and other environmental conditions. Their individual size is negligible, but in a bloom phytoplankton are numerous enough that they act as seed particles for the flow. As a result, differing concentrations of phytoplankton reveal the swirling, turbulent mixing of ocean waters. (Image credit: NASA/USGS; via SpaceRef; submitted by jshoer)

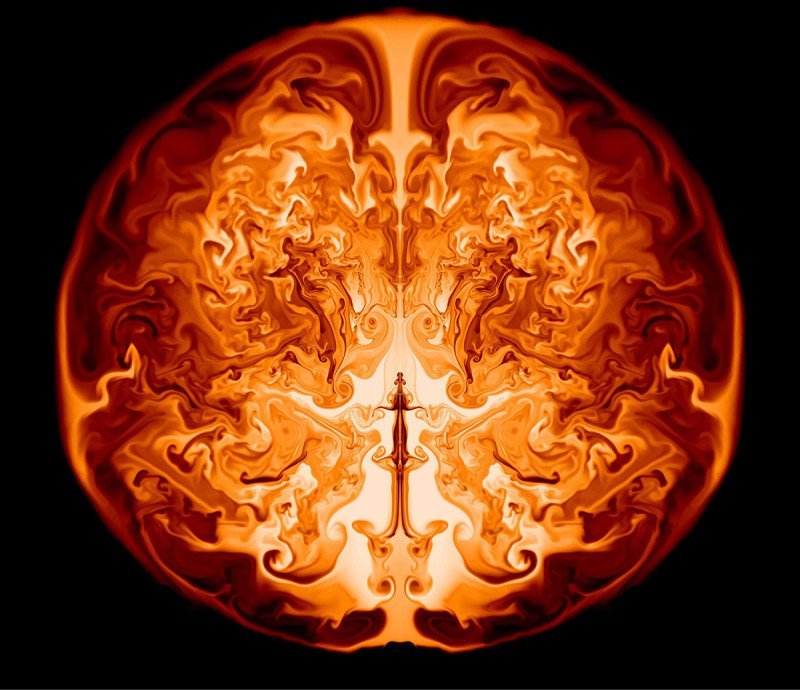
Supernova Simulation
New research shows that supermassive first-generation stars may explode in supernovae without leaving behind remnants like black holes. The work is a result of modeling the life and death of stars 55,000 to 56,000 times more massive than our sun. When such stars reach the end of their lives, they become unstable due to relativistic effects and begin to collapse inward. The collapse reinvigorates fusion inside the star and it begins to rapidly fuse heavier elements like oxygen, magnesium, or even iron from the helium in its core. Eventually, the energy released overcomes the binding energy of the star and it explodes outward as a supernova. The image above is a slice through such a star approximately one day after its collapse is reversed. Hydrodynamic instabilities like the Rayleigh-Taylor instability produce mixing of the heavy elements throughout the expanding interior of the star. The mixing should produce a signature that can be observed in the aftermath as these stars seed their galaxies with the heavy elements needed to form planets. For more, see Science Daily and Chen et al. (Image credit: K. Chen et al., via Science Daily; submitted by mechanicoolest)

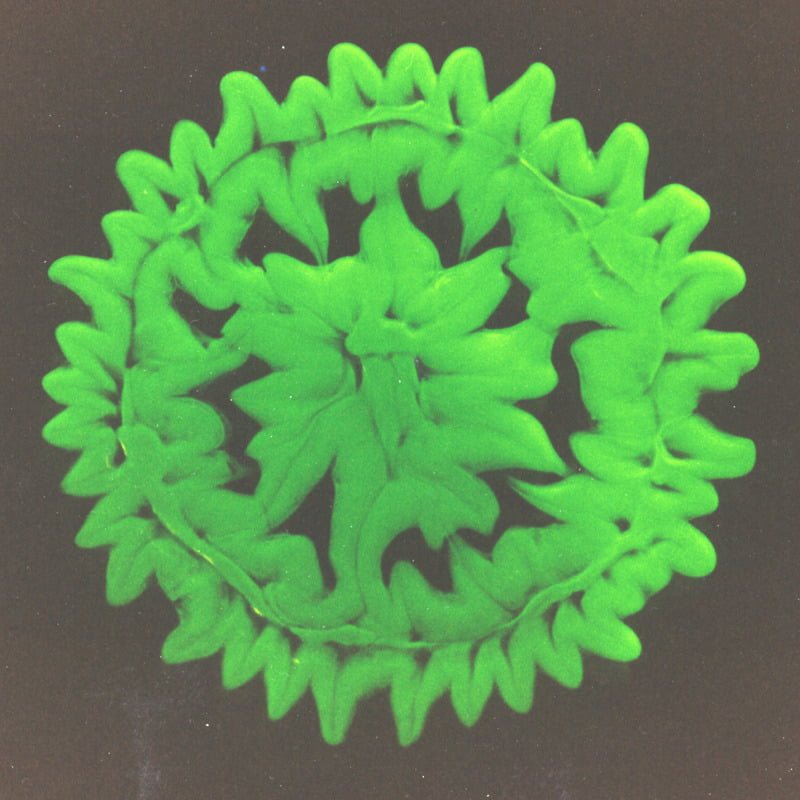
Distorted Rings
The Marangoni effect is generated by variations in surface tension at an interface. Such variations can be temperature-driven, concentration-driven, or simply due to the mixing between fluids of differing surface tensions as is the case here. The pattern in the image above formed after a dyed water droplet impacted a layer of glycerin. The initial impact of the drop formed an inner circle and outer ring. This image is from 30 seconds or so after impact, after the Marangoni instability has taken over. The higher surface tension of the water pulls the glycerin toward it, resulting in a flower-like pattern. (Photo credit: E. Tan and S. Thoroddsen)