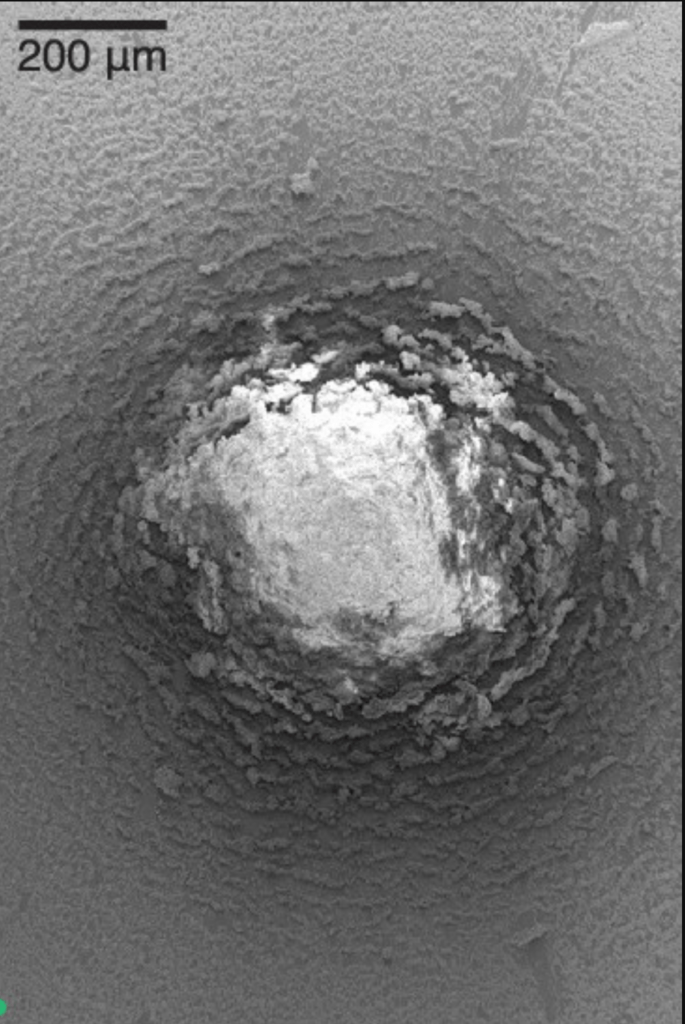
When they evaporate, drops of liquids like coffee and red wine leave behind stains with a darker ring along the edges, thanks to capillary action and surface tension pulling particles to that outer edge. In contrast, sublimating a frozen droplet leaves a stain pattern that concentrates at the center (top). When droplets freeze from the surface upward, particles within the droplet are driven toward the center as the freeze front pushes toward the drop apex. The final shape of the stain depends on the initial geometry of the droplet, and the concentration of particles toward the center occurs because of the way that the particle freezes, not how it sublimates (bottom).
Since many industrial processes rely on droplet evaporation to spread coatings, this work offers a new way to control the final outcome. (Image and research credit: E. Jambon-Puillet, source)