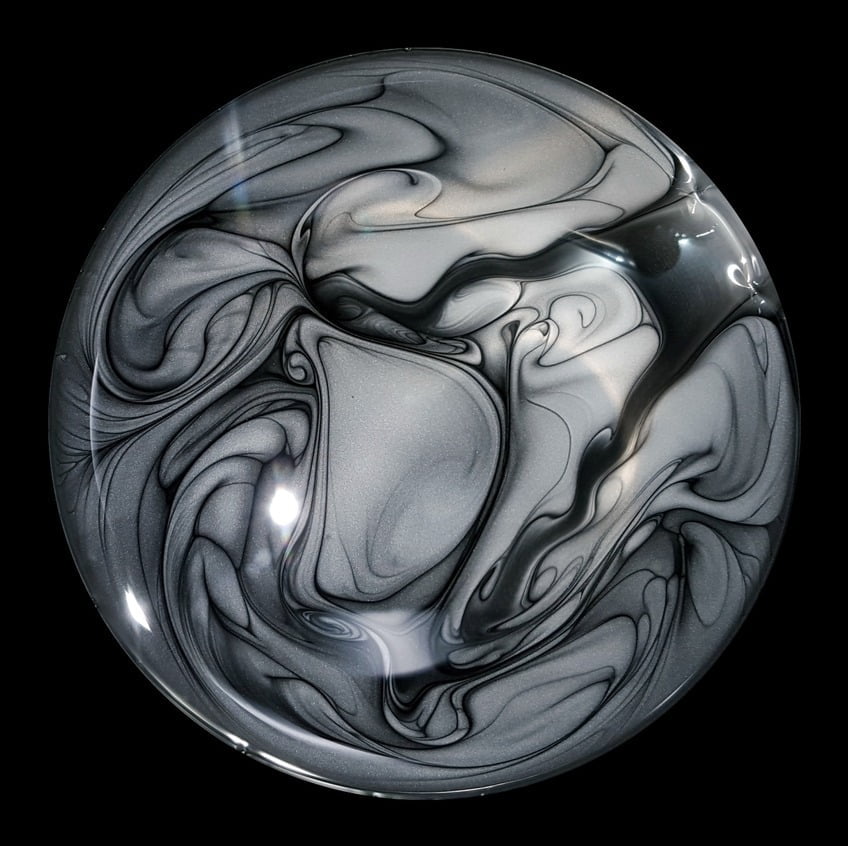
At first glance, the drinking bird is a simple desk toy, but the physics and engineering behind the device is clever enough to have challenged many great minds. In this video, Bill Hammack dissects the drinking bird, revealing the heat engine beneath the felt and feathers. The bird’s drinking is driven by thermodynamics and the relative pressures of fluids in its head and body. When the beak is wetted, fluid wicks up the felted head and slowly evaporates, thereby cooling the vapor inside the head. Some of that vapor condenses, lowering the vapor pressure in the head and allowing liquid to rise from the body. When enough fluid reaches the head, the bird tips forward. This allows vapor to rise up the liquid column into the head, equalizing the pressure between the two ends. The bird sits up with a freshly wetted head and starts the cycle over. Check out the full video for more detail, including a look at what other methods can drive the bird, including bourbon and light bulbs. (Video and image credit: B. Hammack; via J. Ouellette)