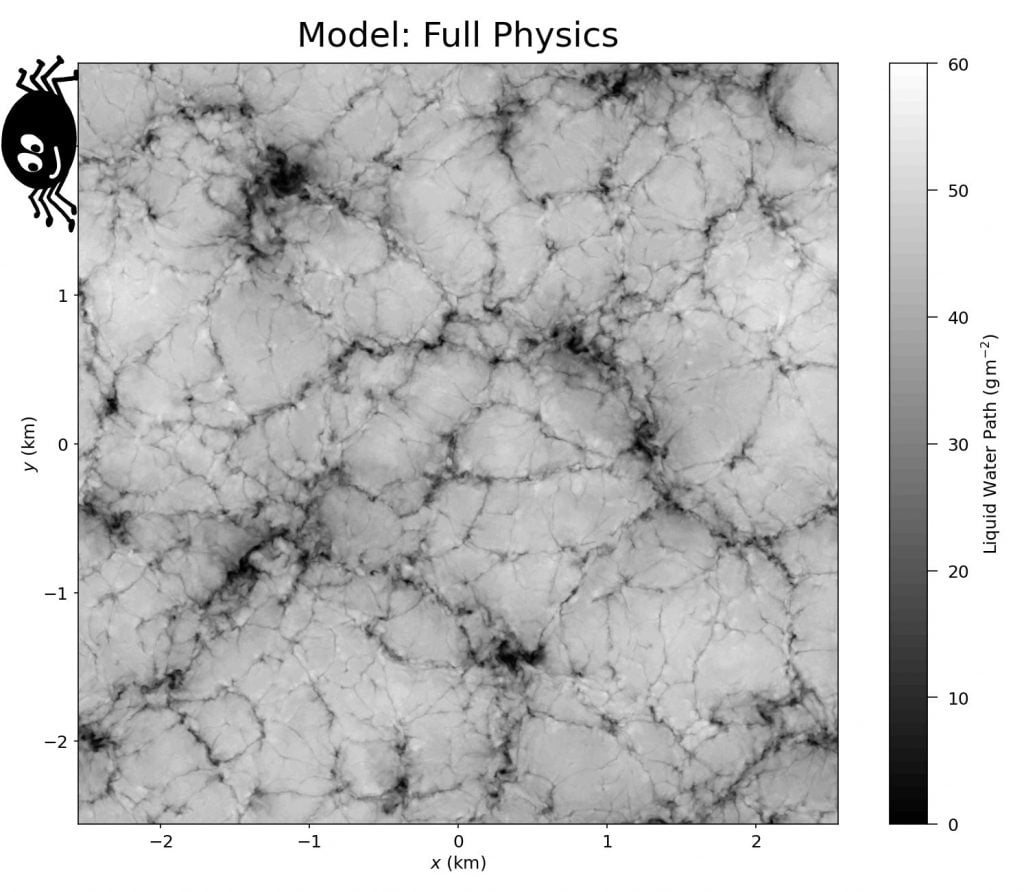
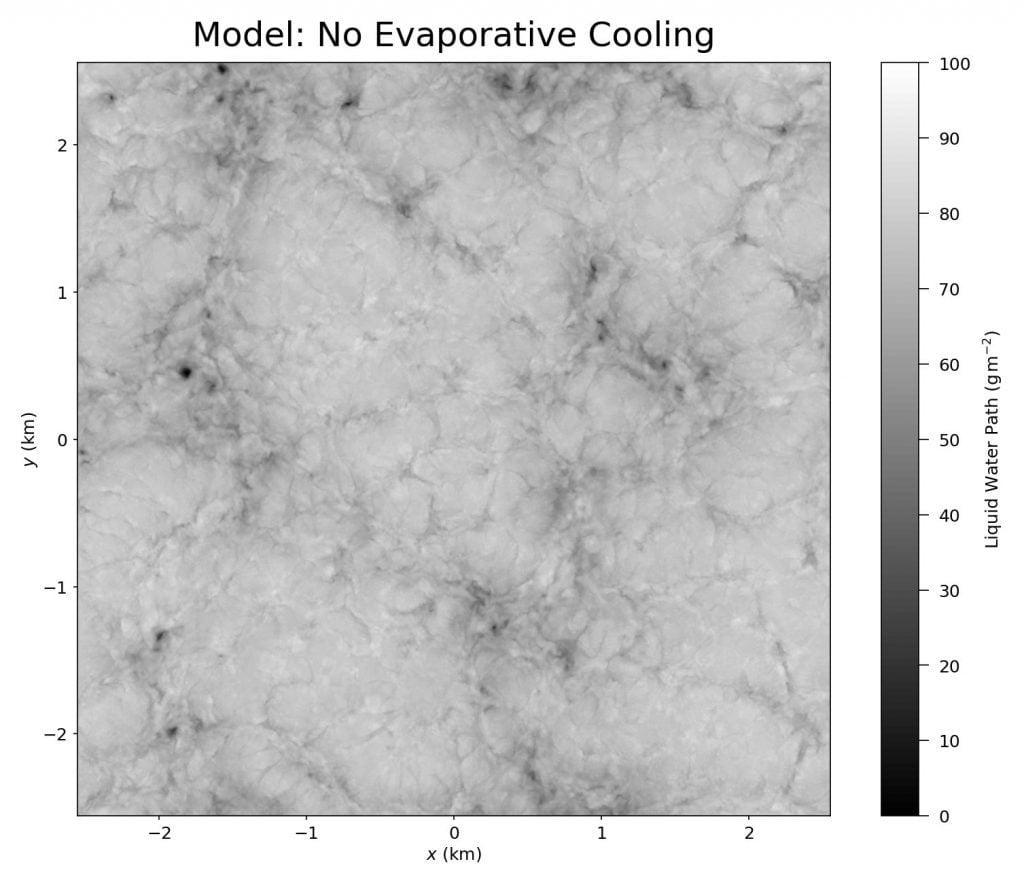
Stratocumulus clouds cover about 20% of Earth’s surface at any given time, and they form distinctive patterns of lumpy cells separated by thin slits. Because of their interconnectedness, researchers nicknamed these narrow regions spiderwebs. New simulations show that evaporative cooling along the cloud tops drives the formation of these spiderwebs (Image 2). Without it (Image 3), the cloud pattern looks very different. (Image credits: featured image – L. Dauphin/MODIS, others – UConn ME 3250; research credit: G. Matheou et al.)
Tag: evaporation

Inside Drying Wood
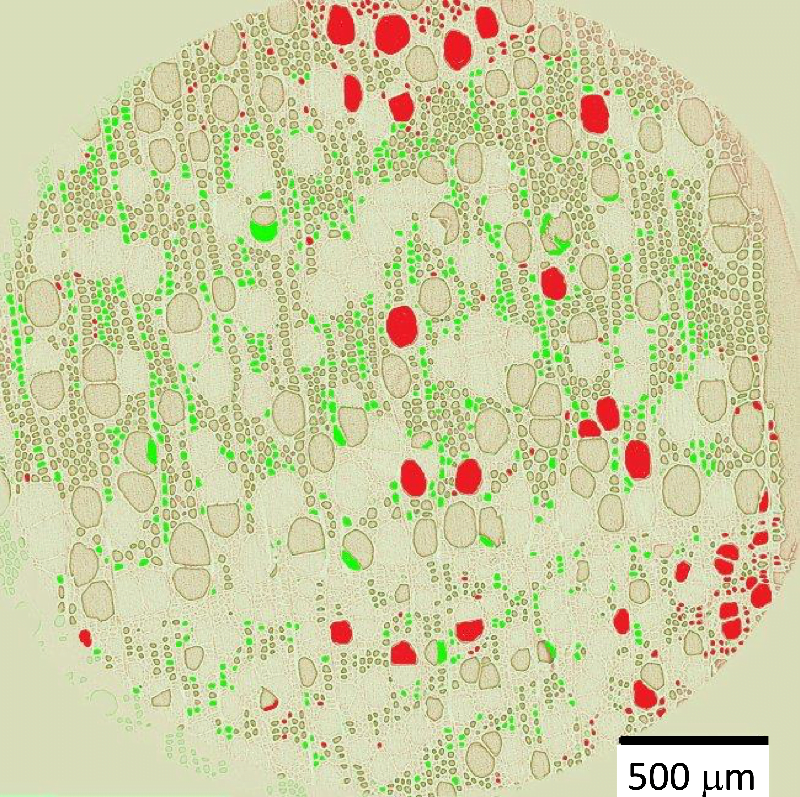
Wood must dry before it can be used in most applications, but with its complex internal structure exactly how wood dries out has been unclear. New experiments combining MRI and x-ray imaging reveal a process quite different than expected.
Inside hardwoods like poplar — the species studied here — wood contains both solid structures and pores where water can gather. The pores do not form a fully interconnected network, so capillary action alone is unable to carry water through the pores and out to a surface where it can evaporate.
Instead, researchers found that water evaporating at the surface came from so-called “bound water” in the wood’s solid structures. As the bound water evaporated, it caused water in the wood pores to diffuse into the solid walls, becoming bound and continuing to feed the evaporation. (Image and research credit: H. Penvern et al.; via APS Physics)

Vanishing Spirits: Aging
The necessary ingredients for scotch whisky’s evaporation patterns are alcohol, surfactants, and polymers; some of those components are absorbed during the spirit’s aging in oak casks. Photographer Ernie Button explored how long it takes for whisky to absorb enough of these chemicals by photographing the stains left by samples aged between 1 and 5 weeks in an oak cask. He found that it takes about 5 weeks for the scotch patterns to begin emerging.
The aging process for scotch and other cask-aged spirits depends on the fluid’s flow through the porous grain of the oak. Evaporation plays a significant role in the process, so the aging process differs based on environmental conditions. For example, distillers in the dry, high-altitude climate of Colorado must use climate-controlled storage, whereas Scottish distillers use a more humid natural climate to their advantage.
Another major factor in the aging process is the charred oak cask itself. Some whiskys, like American bourbon, always use a brand new barrel, whereas scotch is often aged in a previously-used cask. With older casks, absorption of molecules from the wood takes longer, which is why scotch is typically aged for much longer than some other types of whisky. (Image, research, and submission credit: E. Button; see also)

Vanishing Spirits: Rice-Based Whisky
In yesterday’s post, photographer Ernie Button showed us that barrel-aged gin can leave behind an evaporation pattern remarkably similar to scotch whisky. But even among whiskys not every spirit uses the same grains.
Here we see patterns left behind by a 10-year-aged, rice-based whisky. The stains are entirely different than those of (barley-based) scotch. The rice leaves behind stains with distinct regions, including a radially uniform rim and an interior reminiscent of satellite photos. Presumably the interaction of rice and the cask leaves the whisky with surfactants and polymers that behave rather differently than those of scotch.
It takes time for spirits to take on character from the casks they’re aged in. Tomorrow we’ll take a look at just how much aging is necessary for scotch’s patterns to emerge. (Image, research, and submission credit: E. Button; see also)

Vanishing Spirits: Gin
Photographer Ernie Button has spent years exploring the patterns left by evaporating scotch. A team of researchers found that the uniformity of scotch whisky’s stain requires three ingredients: alcohol to drive concentration gradients, surfactants to pull particulates away from the drop’s edge, and polymers to help stick particles to the glass.
Button wondered whether other spirits might produce similar patterns, and, indeed, some do. The photos above are stains left behind by evaporated gin that’s been aged for a year in oak casks. The patterns are extremely similar in appearance to those from aged scotch whiskies, suggesting that the same fluid dynamical effects are at play here, despite the difference in liquor. But do all grain spirits make these patterns? Check back tomorrow to find out. (Image, research, and submission credit: E. Button; see also)

Storm Eyes and Mushrooms in a Drop
In industry, drying droplets often have many components: a liquid solvent, solid nanoparticles, and dissolved polymers. The concentration of that last component — the polymers — can have a big effect on the way the droplet dries, as seen in the video above.
Without polymers, the droplet dries similarly to a coffee ring stain. But at moderate concentration, we see something very different. The droplet forms an eye in the middle, similar to a hurricane’s, and the edges of the droplet sprout mushroom-shaped plumes that grow and merge with one another along the edge. With even larger polymer concentrations, the mushrooms sweep their way inward, leaving a feathery stain behind. (Video, image, and research credit: J. Zhao et al.)

Why Aren’t Trees Taller?
Trees are incredible organisms, with some species capable of growing more than 100 meters in height. But how do trees get so big and why don’t they grow even taller? The limit, it turns out, is how far fluid forces can win over gravity.
To live and grow, trees must be able to transport nutrients between their roots and their highest branches. As explained in the video, there are three forces that enable this transport inside trees: transpiration, capillary action, and root pressure. Of these, you are probably most familiar with capillary action, where intermolecular forces help liquids climb up the inside of narrow spaces, like the straw in your drink. Capillary action can’t lift liquids more than a few centimeters against gravity, though.
Similarly, root pressure is limited in how far it can raise liquids. Functionally, it’s pretty similar to the way a column of water or mercury can be held up by atmospheric pressure acting at the base of a barometer. But atmospheric pressure can only hold up 10.3 meters of water, so what’s a tree to do?
This is where transpiration — the most important force for sap transport in the tree — comes in. As water evaporates out of the tree’s leaves, it creates negative pressure that — along with water’s natural cohesion — literally drags sap up from the roots. It’s this massive pull that drives the flow and enables most of a tree’s height. (Image and video credit: TED-Ed)

Crystalline Critters
In 5th grade, I grew crystals by evaporating solutions of salt water from miniature pie tins. The results were white, boxy crystals whose size depended on how much salt I’d managed to dissolve into the water. But it turns out I could have gotten much cooler results if I’d evaporated my salt water a drop at a time on a hot superhydrophobic surface. That’s how these researchers formed the “crystal critters” shown in the video above.
Initially, the evaporating salt water drop is what we would expect, but once enough water is gone to leave a shell of salt, the drop grows legs and lifts off the surface. From that point, all growth occurs from the surface up. Because the surface is heated, evaporation happens quickest at that point of contact, and the water that remains is drawn down the legs, providing more fluid for evaporation as well as additional salt to grow the crystal. (Video, image, and research credit: S. McBride et al.)

The Vortex Beneath a Drop
While we’re most used to seeing levitating Leidenfrost droplets on a solid surface, such drops can also form above a liquid bath. In fact, the smoothness of the bath’s surface, combined with mechanisms discussed in a new study, means that drops will levitate at a cooler temperature over a liquid than they will over a solid surface.
Researchers found that a donut-shaped vortex forms in the bath beneath a levitating droplet, but the direction of the vortex’s circulation is not always the same. For some liquids, the flow moves radially outward from beneath the drop. In this case, researchers found that the dominant force was shear stress caused by the vapor escaping from under the droplet.
With other droplet liquids, the flow direction instead moved inward, forming a sinking plume beneath the center of the drop. In this situation, researchers found that evaporative cooling dominated. As the liquid beneath the droplet cooled, it became denser and sank. At the same time, the lower temperature changed the bath’s local surface tension, creating the inward surface flow through the Marangoni effect. (Image credit: F. Cavagnon; research credit: B. Sobac et al.)

Mossy Vortex Rings
Many plants have evolved an ability to move remarkably quickly. Often, this capability is driven by water. Here we see the moss Sphagnum affine, which disperses its spores explosively. The process is triggered by the spore capsule gradually drying out; its shape changes from round to cylindrical, pressurizing the capsule. Once the internal pressure is high enough to overcome the strength of the capsule’s upper membrane, the capsule bursts, sending a plume of spores aloft. The sudden release of spore-laden air forms a vortex ring, which lifts the spores higher far more efficiently than they would be otherwise. (Image credit: capsule dry-out – J. Edwards et al., spore dispersal – J. Edwards et al. 2010; research credit: J. Edwards et al.)