Here’s a simple fluids experiment you can try at home using acrylic paints, ink, isopropyl alcohol and a few other ingredients. When dropped onto diluted acrylic paint, a mixture of black ink and alcohol spreads in a fractal fingering pattern. The radial (outward) flow is driven by the alcohol’s evaporation, which increases the local surface tension and draws fluid outward. The shape and density of the fingers depends, at least in part, on the viscosity of the underlying paint layer; more viscous paint layers grow smaller and denser fractal patterns. (Image and video credit: S. Chan et al.)
Tag: evaporation

The Yarning Droplet
Marangoni bursting takes place in alcohol-water droplets; as the alcohol evaporates, surface tension changes across the liquid surface, generating a flow that tears the original drop into smaller droplets. Here researchers add a twist to the experiment using PMMA, an additive that dissolves well in alcohol but poorly in water. As the alcohol evaporates, the PMMA precipitates back out of the water-rich droplet, forming yarn-like strands. (Image and video credit: C. Seyfert and A. Marin)

Cracking Droplets
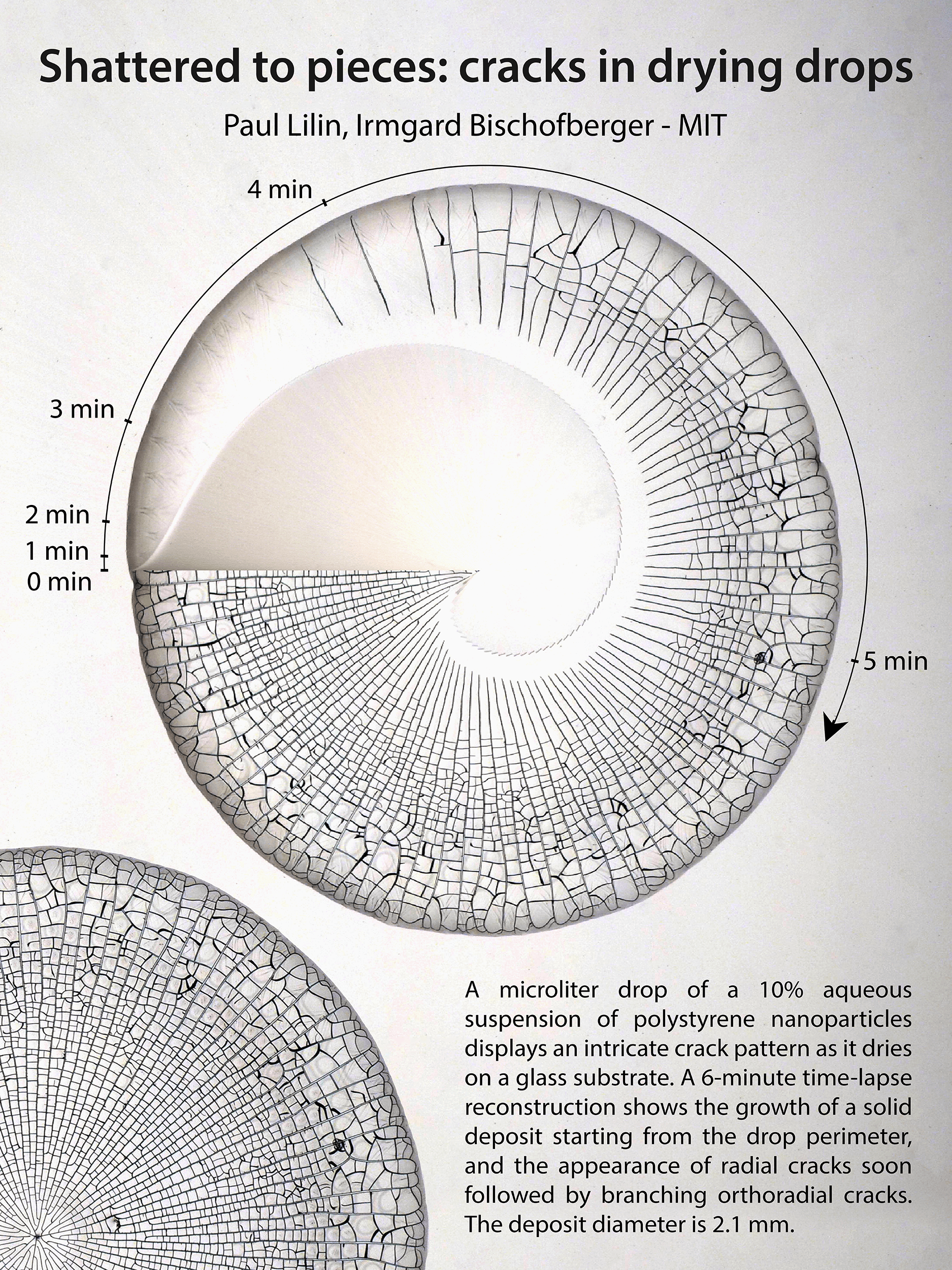
Droplets infused with particles — like coffee — can leave complex stains once they evaporate. Here researchers show the complex cracking pattern that develops as a droplet with nanoparticles evaporates. The central image in the poster actually shows the drop’s pattern changing in time. The initial drop is shown at 9 o’clock, and as you move clockwise around the drop, time passes and the crack structure becomes more complex. What a neat way to visualize the changes! (Image and research credit: P. Lilin and I. Bischofberger)

Triple Leidenfrost Effect
Droplets can skitter across a hot surface on a layer of their own vapor, thanks to the Leidenfrost effect. If two Leidenfrost droplets of the same liquid collide, they merge immediately. But that doesn’t always happen with two dissimilar liquids. A new study looks at how dissimilar Leidenfrost droplets collide. The researchers found that these drops can bounce off one another repeatedly before their eventual merger (Image 1).
Just as a vapor layer prevents the drops from touching the hot plate, a vapor layer forms between them when they collide, preventing contact (Image 2). Because of these three distinct areas of Leidenfrost vapor (one beneath each drop and one between the drops), the researchers call this the triple Leidenfrost effect.
Eventually, the more volatile (in other words, easily evaporated) drop shrinks to a size similar to its capillary length, at which point the drops merge. If the boiling points of the two liquids are vastly different, the merger can be explosive (Image 3). (Image and research credit: F. Pacheco-Vázquez et al.; via APS Physics)

Modelling Volcanic Bombs
When magma meets water on its journey to the surface, the two form a large, partially molten chunk known as a volcanic bomb. As you would expect from their name, these bombs can often be explosive, either in the air or upon impact. But a surprising number of these bombs never explode. Since catching volcanic bombs in action is far too dangerous, researchers modeled them instead to determine what makes a dud.

The type of volcanic bomb they were most interested in comes from Surtseyan eruptions, where the bombs travel through shallow sea or lake water, collecting moisture along the way. When the water reaches the molten interior of the volcanic bomb, it flashes into steam. That’s where the pressure to explode the bombs comes from. But the team found that the bombs are also extremely porous, thanks to bubbles created as the magma depressurizes on its trip to the surface. If the bomb is porous enough, steam escapes the rock before it can build to explosive pressures. (Image credit: top – NASA, others – E. Greenbank et al.; research credit: E. Greenbank et al.; via NYTimes; submitted by Kam-Yung Soh)

Fractal Frost
As nightly temperatures drop in the northern latitudes, many of us are beginning to wake up to frosty patterns on leaves, windows, and cars. Frost‘s spread is a complex dance between evaporation and nucleation, as seen in this recent study.
Here, researchers watched frost grow on a surface covered in 30-micrometer-wide micropillars. The pillars serve as anchor points for droplets, making frosting easier to observe. At low humidity levels (Image 1), droplets evaporate so quickly that frost regions remain isolated and do not interact. At high humidity levels (Image 3), on the other hand, the droplets evaporate so slowly that they’re able to poach water vapor from their neighbors to form frost spikes. When a spike touches another droplet, it freezes the region almost instantly. As a result, the frost spreads quickly and covers nearly every part of the surface. At intermediate humidity levels (Image 2), though, this frost chain reaction and evaporation compete, causing the frost to grow in fractals. (Image and research credit: L. Hauer et al.; via APS Physics)

Sliding Along
Robust, self-cleaning surfaces are a holy grail for many engineers, but they’re tough to achieve. One necessary ingredient for a self-cleaning surface is the ability to shed water, which is why superhydrophobic coatings and surface treatments are popular. Here, researchers prompt their droplets to move at speeds up to 16 cm/s by dropping them onto a thin layer of heated oil.
Longtime readers will no doubt be reminded of self-propelling Leidenfrost drops, but this situation is not quite the same. In general, the oil layer suppresses the Leidenfrost effect. Instead, the oil heats the drop, evaporating its vapor. A bubble of vapor will nucleate at a random location in the droplet and eject itself, pushing the drop in the opposite direction. Because of the disruption caused by that ejection, new bubbles will preferentially form at the same spot, providing an ongoing supply of vapor that keeps the drop sliding in the same direction. It’s like a miniature rocket zooming along the oil film! (Image and research credit: V. Leon and K. Varanasi; via APS Physics)

Programmable Capillary Action
Capillary action combines the cohesive forces within a liquid and the adhesive forces between a liquid and solid to enable a liquid to fill narrow spaces, even against the force of gravity. To control capillary action, researchers are 3D-printing what they call “unit cells,” tiny structures that water and other liquids can climb. There’s no pump raising the liquid through these structures, just capillary action.
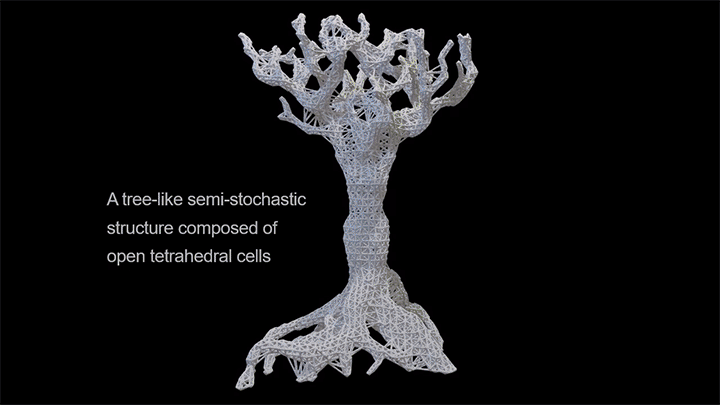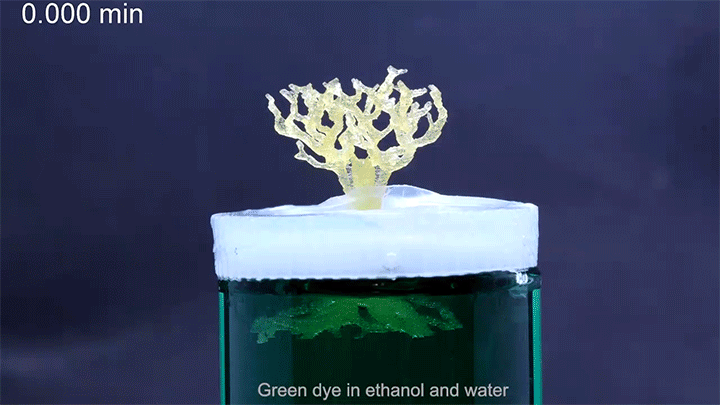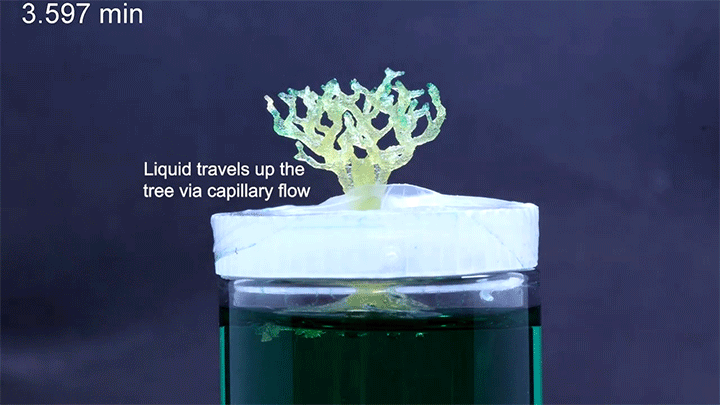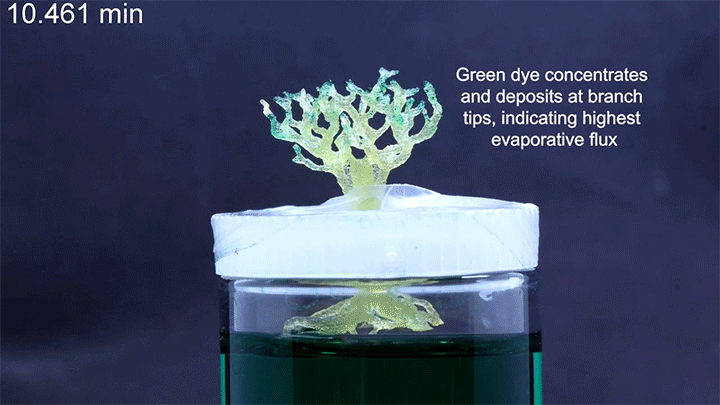
In a particularly neat demonstration of the technology, the researchers built a tree-like structure out of many open-walled unit cells and placed the “root” system in a closed reservoir. Capillary action drew liquid up the structure to the tips of its branches, where the dyed water evaporated. The process is similar to transpiration in trees, though in trees, capillary action provides much less of the lift. (Image and research credit: N. Dudukovic et al.; via Nature; submitted by Kam-Yung Soh)

Snapping When Swollen
The Venus flytrap snaps shut on its hapless prey by swelling cells in its leaves with water. Under the added pressure of a fly’s footstep, the leaves’ snapping instability triggers, trapping the insect. Researchers are using similar physics to create jumping and snapping polymer gels, like the one seen below.

To trigger the behavior, researchers soaked their polymer-based gel strips and shells in a solvent of n-hexane, which easily permeated the material and made it swell up. As the solvent evaporates from the swollen gel, the polymer material changes shape, sometimes in smooth bends and sometimes in abrupt snaps. The group was able to harness those snaps to have their materials descend slopes and climb ladders — all without motors, batteries, or external sources of energy. (Image credit: plant – A. Dénes, shell – Y. Kim et al.; research credit: Y. Kim et al.; via Physics World)

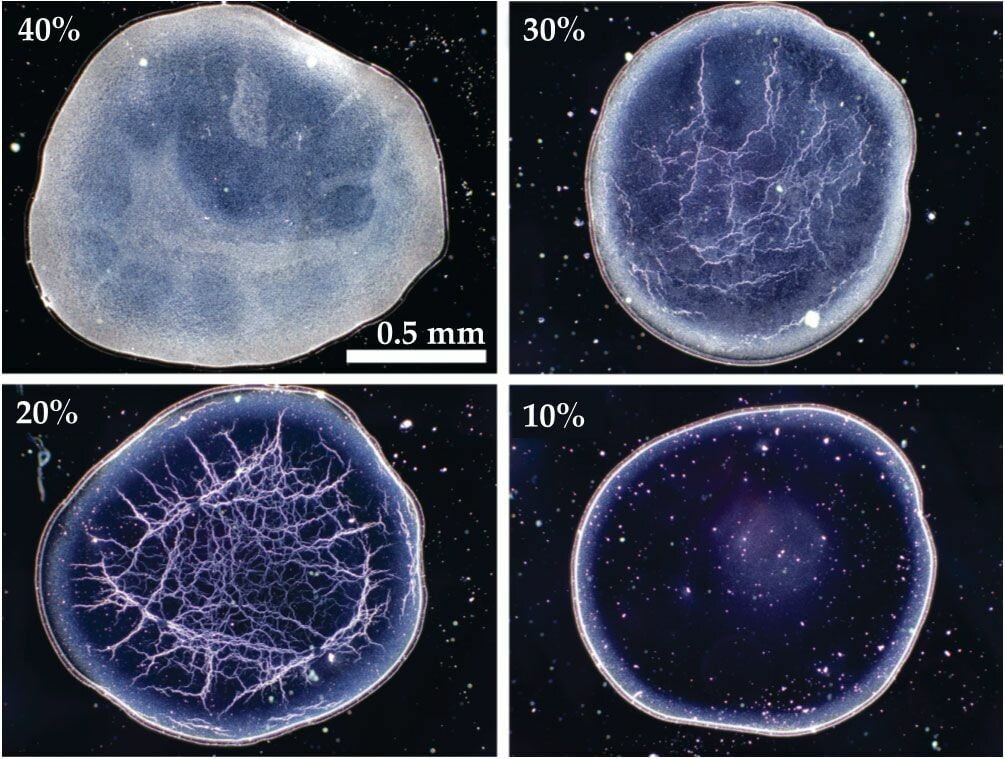
Whiskey Webs
Unlike scotch whisky, when American bourbon whiskeys are diluted, they form unique web-like evaporation patterns. These differences arise in part from the way the liquors are aged: scotch is aged in re-used barrels, whereas bourbons require aging in a new, charred American white oak barrel*.
During aging, the whiskey picks up water-insoluble chemicals from the barrel. When water is added to the bourbon, it helps transport those insoluble components to the surface of a droplet, where they form a monolayer of fatty acid chains (Image 2; in green). As evaporation continues and the droplet gets smaller, the molecules at the shrinking surface collapse inward, forming the rigid web structure we see left behind. The patterns that form act as a kind of fingerprint for the bourbon. Check out some of the brand-to-brand variations over at the researchers’ Whiskey Webs site. (Image and research credit: S. Williams et al.; via Physics Today)
* In case you were wondering, this is actually a legal requirement in order to be considered bourbon. Bourbons must also be made from a grain mixture that is >50% corn.