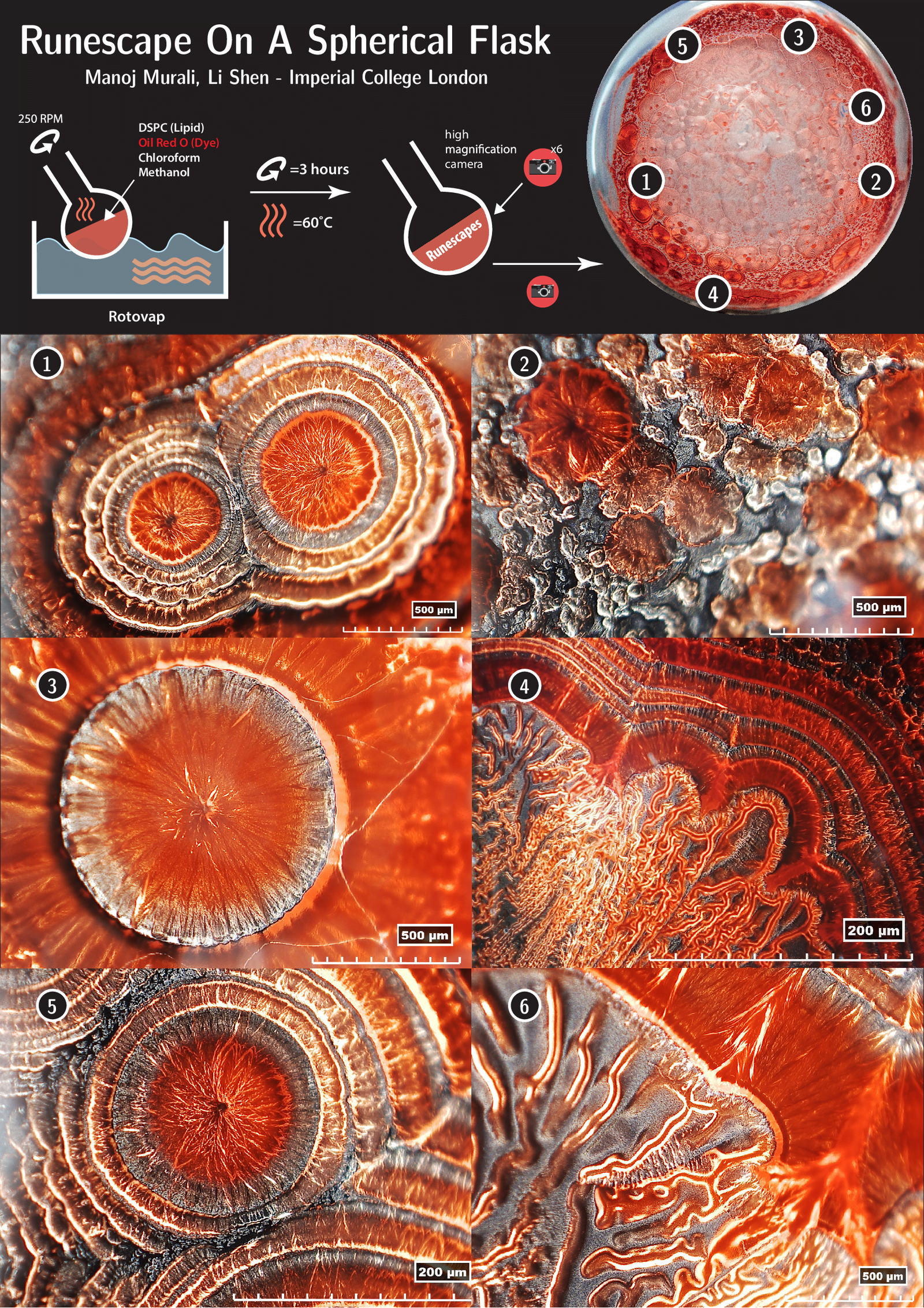
Drying fluids can leave behind all kinds of fascinating patterns, as we’ve seen before with whiskey, coffee, and even blood. Here researchers study patterns left behind by lipids, dyes, and other fluids. They place their mixture in a rotating flask kept in a warm bath. For a few hours, the fluids mix, chemically react, and evaporate. The complex interactions that take place in that time leave behind fascinating, rune-like patterns, seen here under a microscope. It’s a bit like looking at photos of Martian landscapes! (Image credit: M. Murali and L. Shen)
Tag: evaporation

Drying Cracks
Droplets with particles in them can leave complex stains when they dry — just look at coffee rings and whiskey marks! Here, researchers look at the patterns left on glass by small droplets that evaporated and left behind their nanoparticles. As evaporation takes place, the droplet’s shape changes, adding stress to the growing layer of nanoparticle residue. Cracking is one way to relieve that stress. Another method is delamination — peeling up from the surface. On the leftmost drop, the outer rim of nanoparticles delaminated — as seen from the circular fringes — which released stress without cracking. The rightmost drop, which had a smaller contact angle with the surface, couldn’t delaminate and instead cracked throughout. (Image credit: M. Ibrahim et al.)

Ominous Mammatus
Mammatus clouds are fairly unusual and often look quite dramatic. Most clouds have flat bottoms, caused by the specific height and temperature at which their droplets condense. But mammatus clouds have bubble-like bottoms that are thought to form when large droplets of water or ice sink as they evaporate. Although they can occur in the turbulence caused by a thunderstorm, mammatus clouds themselves are not a storm cloud. They appear in non-stormy skies, too. The clouds are particularly striking when they’re lit from the side, as in the image above. (Image credit: J. Olson; via APOD)

Explaining Salt Polygons
Around the world, salt playas are criss-crossed with meter-sized polygons formed by ridges of salt. The origins of these structures — and the reason for their consistency across different regions of the world — have been unclear, but a new study shows that salt polygons form due to convection happening in the soil underground.
Through a combination of numerical modeling, simulation, lab-scale experiment, and field work, the team revealed the mechanism underlying salt polygons. Areas that form polygons have much greater rates of evaporation than precipitation, and, as water evaporates, these areas draw groundwater from nearby. Salt gets carried with this groundwater.
With strong evaporation, the lake bed forms a highly-concentrated layer of salty water near the surface. Convection cells form, with some regions drawing less saline water upward, while denser, saltier water sinks in other areas. The subsurface convection lines up exactly with the surface structures. The interior regions of polygons are areas where less salty water rises, and salt instead concentrates along the edges of polygons, where saltier water sinks below the surface while evaporation draws solid salt to the surface.

This snapshot shows a numerical simulation of the subsurface convection and surface evaporation that lead to salt polygon formation. Low salinity areas are yellow, while high salinity ones are black. At the surface, blue regions have the maximum upward flow and red regions have the maximum downward flow. The dark, highly saline fingers under the surface align to the red areas on the surface, indicating areas where salty water is sinking. It’s a beautiful result that matches the size, shape, and development time observed for salt polygons in the real world. The team even excavated below salt polygons in Death Valley to confirm that the salt content below ground matched their model’s patterns. Since salt playas are a major source for dust and aerosols that affect climate, their work will be an important factor in future climate modelling. (Image credit: feature – T. Nevidoma, simulation – J. Lasser et al.; research credit: J. Lasser et al.; via APS Physics; submitted by Kam-Yung Soh)

Rocket-Like Supercooled Drops
Many droplets can self-propel, often through the Leidenfrost effect and evaporation. But now researchers have observed freezing droplets that self-propel, too. The discovery came when observing the freezing of supercooled water drops inside a vacuum chamber. The researchers kept losing track of drops that seemingly disappeared. Upon closer inspection, though, they found that the drops weren’t shattering; they were flying away as they froze.
Inside a drop, freezing starts at a point, the nucleation point, and spreads from there. But the nucleation point isn’t always at the center of the drop. This asymmetry, the researchers found, is at the heart of the drop’s propulsion. When ice nucleates, the phase change releases heat that increases the drop’s evaporation rate, which can impart momentum to the drop. For an off-center nucleation, that momentum is enough to send the drop shooting off at nearly 1 meter per second. (Image credit: SpaceX; research credit: C. Stan et al.; via APS Physics)

The Optical Atom
Researchers applied a quantum mechanical technique to study an evaporating drop in extreme detail. The team trapped a spherical water drop and collected the light scattered off it as it evaporated. Using an analytic technique originally developed for an atom, they were able to study changes in the drop down to the nanometric level without relying on numerical simulations to interpret the results. The authors suggest that their method is well-suited to studying the concentration of chemical or biological contaminants on the surface of a drop as it evaporates. (Image credit: droplet – Z. Kaiyv, Fano combs – J. Marmolejo et al.; research credit: J. Marmolejo et al.; via APS Physics)

Illustration of the Fano combs seen by analyzing light scattered from an evaporating drop. 
Chilly Soap Films
Evaporation is a well-known effect in soap films and bubbles. It’s responsible for the ever-changing thickness reflected in the film’s many colors. But evaporation does more than change the bubble’s thickness: it affects its temperature, too. Just as sweat evaporating off our skin cools us, the soap film’s evaporation makes it cooler than the surrounding air.
Researchers found that their soap films could be as much as 8 degrees Celsius cooler than the surrounding air! They also found that the film’s glycerol content affect how much cooler the soap film is; films with more glycerol had higher temperatures, which could impact their overall stability. (Image credit: E. Škof; research credit: F. Boulogne et al.; via APS Physics)

Leidenfrost On Ice
We’ve seen many forms of Leidenfrost effect — that wild, near-frictionless glide that liquid droplets make on a very hot surface — over the years, but here’s a new one: the three-phase Leidenfrost effect. Researchers found that they could generate a Leidenfrost effect using an ice disk placed on an extremely hot surface. During the effect, the ice and its melting layer of water glide on vapor, hence the name.
The team found that getting a three-phase Leidenfrost effect requires a much, much higher temperature than the regular Leidenfrost effect. Water will get its glide on at 150 degrees Celsius. Getting ice to glide on the same surface required a stunning 550 degrees Celsius! Why the big difference? The challenge is that water layer, which, by definition, has a 100-degree difference between its boiling side and its frozen boundary. It takes so much heat to maintain that layer that there’s little energy left over for evaporation; that’s why it takes so much more energy to get the three-phase Leidenfrost effect. (Image and research credit: M. Edalatpour et al.; via Ars Technica; submitted by Kam-Yung Soh)

Everlasting Bubbles
Soap bubbles are delicate and ephemeral, always a breath away from collapse due to thinning driven by gravity or evaporation. But that frailty can be countered. Adding microparticles to the bubble’s shell in place of surfactants counters drainage and makes bubbles last for tens of minutes (left). Adding glycerol to the mix takes things a step further (right). The glycerol, which absorbs water from the surrounding air, counteracts the evaporation, allowing bubbles to remain intact — with no discernible change to their radius — almost indefinitely. So far the researchers have made such a bubble last for 465 days! (Image and research credit: A. Roux et al.; via APS Physics)