Power plants (and other industrial settings) often need to cool water to control plant temperatures. This usually requires cooling towers like the iconic curved towers seen at nuclear power plants. Towers like these use little to no moving parts — instead relying cleverly on heat transfer, buoyancy, and thermodynamics — to move and cool massive amounts of water. Grady breaks them down in terms of operation, structural engineering, and fluid/thermal dynamics in this Practical Engineering video. Grady’s videos are always great, but I especially love how this one tackles a highly visible piece of infrastructure from multiple engineering perspectives. (Video and image credit: Practical Engineering)
Tag: evaporation

Active Cheerios Self-Propel
The interface where air and water meet is a special world of surface-tension-mediated interactions. Cereal lovers are well-aware of the Cheerios effect, where lightweight O’s tend to attract one another, courtesy of their matching menisci. And those who have played with soap boats know that a gradient in surface tension causes flow. Today’s pre-print study combines these two effects to create self-propelling particle assemblies.
The team 3D-printed particles that are a couple centimeters across and resemble a cone stuck atop a hockey puck. The lower disk area is hollow, trapping air to make the particle buoyant. The cone serves as a fuel tank, which the researchers filled with ethanol (and, in some cases, some fluorescent dye to visualize the flow). Like soap, ethanol’s lower surface tension disrupts the water’s interface and triggers a flow that pulls the particle toward areas with higher surface tension. But, unlike soap, ethanol evaporates, effectively restoring the interface’s higher surface tension over time.
With multiple self-propelling particles on the interface, the researchers observed a rich series of interactions. Without their fuel, the Cheerios effect attracted particles to each other. But with ethanol slowly leaking out their sides, the particles repelled each other. As the ethanol ran out and evaporated, the particles would again attract. By tweaking the number and position of fuel outlets on a particle, the researchers found they could tune the particles’ attractions and motility. In addition to helping robots move and organize, their findings also make for a fun educational project. There’s a lot of room for students to play with different 3D-printed designs and fuel concentrations to make their own self-propelled particles. (Research and image credit: J. Wilt et al.; via Ars Technica)

“Last Breath of Autumn”
On a rainy autumn day, Agorastos Papatsanis headed to the forest in search of fungi. There he captured this fairytale-like scene with falling rain and drifting spores. Near the forest floor, any breeze is slight, so mushrooms use their own humidity to move air and spread their spores. As water evaporates from the mushroom’s cap, it cools the air nearby, causing it to spread outward. Since that water vapor is lighter than air, it rises, too, carrying the mushroom’s spores along with it. (Image credit: A. Papatsanis; via Wildlife PotY)

Self-Cleaning With Salt Critters
Even freshwater contains trace salts and minerals that cause scaly buildups as they evaporate. Getting rid of the scale usually requires toxic chemicals and/or lots of scrubbing, neither of which are desirable at the industrial level. At the same time, we’re extremely limited in the amount of freshwater that we have available; only about 1% of Earth’s water is liquid and fresh. If we could use salt water in more industrial processes, that would preserve freshwater for drinking and agriculture. But how do we tackle the scaly buildup?

(A) On microtextured surfaces, salt from evaporating drops can work its way into the gaps, destroying the superhydrophobicity of the surface. (B) In contrast, nanotextured surfaces give the salt nowhere to adhere, resulting in “salt critters” that grow upward and detach. Enter “salt critters.” Researchers found that when salt water evaporated from microtextured surfaces designed to shed water, salt would eventually build up in the gaps, breaking the hydrophobic effect and allowing scale to build up. In contrast, a nanotextured surface left nowhere for the salt to adhere. On these surfaces, evaporating salt water built jellyfish-like salt critters that rose from the surface and, eventually, broke off and rolled away, leaving the surface pristine. (Image credit: S. McBride; research credit: S. McBride et al.; via Physics Today)

How Ferns Spread Themselves



Ferns don’t rely on pollen and pollinators to spread. Instead, they use a little water and a lot of ingenuity, as shown in this video from Deep Look. Peer underneath a fern and you’ll find leaves dotted with spores. As they mature, water evaporates from the sporangium, eventually triggering a catapult that launches the spores. Those spores grow little gametophytes that produce the fern’s sperm and eggs; given a little rain or a nice puddle, the sperm and eggs can find each other and trigger the birth of a new baby fern. (Video and image credit: Deep Look)

Skittering Drops
Drip some ethanol on a hot surface, and you’d expect it to spread into a thin layer and evaporate. But that doesn’t always happen, and a recent study looks at why.
Ethanol is what’s known as a volatile liquid, meaning that it evaporates easily at room temperatures, well below its boiling point. When dropped on a uniformly heated surface above 45 degrees Celsius, the drop contracted into a hemisphere and then began to wander randomly across the surface. Researchers trained an infrared camera on the drop from below (above image), and found an unsteady, roiling motion inside the drop. These asymmetric flows, they concluded, drive the drop’s erratic self-propulsion. They suspect the mechanism may explain why some ink droplets wind up in the wrong place on a page during ink-jet printing. (Image and research credit: P. Kant et al.; via APS Physics)

Drying Unaffected by Humidity
Water evaporates faster in dry conditions than in humid ones, but the same isn’t true of paint. Instead, paint’s drying time is largely independent of the day’s humidity. That’s because of paint’s long chains of polymers. As water in the paint evaporates, these polymers are drawn to the surface, forming a viscoelastic layer that hinders evaporation and keeps the drying rate independent up to about 80 percent humidity.

Illustration depicting evaporation of water (left) and evaporation of a polymer solution (right). As water evaporates from the polymer solution, it draws polymers to the surface, where they form a layer that hinders evaporation and makes its rate independent of humidity. The polymer layer explains why evaporation isn’t affected by humidity at longer times, but researchers also saw humidity-independent evaporation early in their experiments. Under a microscope, they discovered a thin gel layer (top image) covering the air-polymer interface. They propose that this fast-forming layer further hinders evaporation. Their findings may be significant for virus-laden respiratory droplets, which also contain polymers. (Image and research credit: M. Huisman et al.; see also J. Salmon et al.; via APS Physics)

Gravity Changes Droplet Shapes
With small droplets, gravity usually has little effect compared to surface tension. An evaporating water droplet holds its spherical shape as it evaporates. But the story is different when you add proteins to the droplet, as seen in this recent study.

The protein-filled sessile drop starts out largely spherical, but as the drop evaporates, the concentration of proteins reaches a critical point and an elastic skin forms over the drop. From this point onward, the drop flattens. As a protein-doped droplet sitting on a surface evaporates, it starts out spherical, like its protein-free cousin. But, as the water evaporates, it leaves proteins behind, gradually increasing their concentration. Eventually, they form an elastic skin covering the drop. As water continues to evaporate, the droplet flattens.

For a hanging droplet, the shape again starts out spherical. But as the drop’s water evaporates and the proteins concentrate, it also forms an elastic skin. As the drop evaporates further, the skin wrinkles. In contrast, a hanging droplet with proteins takes on a wrinkled appearance once its elastic skin forms. The key difference, according to the model constructed by the authors, is the direction that gravity points. Despite these droplets’ small size, gravity makes a difference! (Image, video, and research credit: D. Riccobelli et al.; via APS Physics)

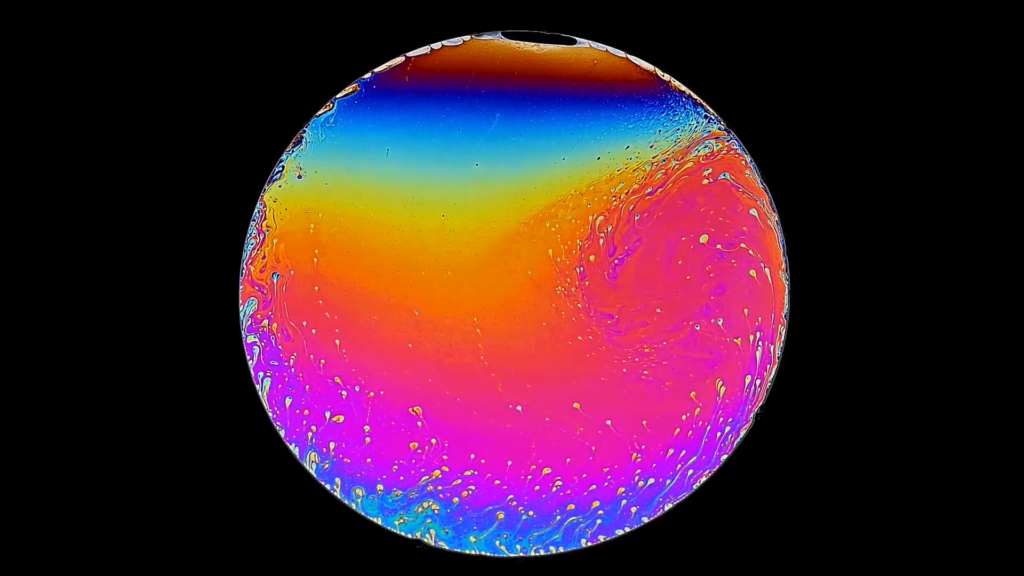
Colorful Drainage
Bright colors mark this slowly draining soap film. The film sits slightly off-horizontal, so flow shifts over time from the top of the frame to the bottom. The fluid is also evaporating. All the faster shifts are caused by ambient air currents from the room. The colors of the film are directly related to the local thickness; as the film thins and evaporates, the bright colors shift to darker ones. Eventually, that black region at the top will expand and the film will break up. (Video credit: B. Sandnes/Complex Flow Lab)