Beads of condensation on a cooling, oil-slicked surface have a dance all their own in this video. Large droplets gobble up their fellows as they follow serpentine paths; each new droplet donates its interfacial energy to feed the larger drop’s kinetic energy. Eventually, the big drops switch to a circular path, like an ouroboros, the tail-eating serpent of mythology. This transition happens due to the oil shifted by the dancing droplets. You can recreate the effect at home by rubbing a thin layer of oil over glass and setting it atop a hot mug of your favorite beverage. (Video and image credit: M. Lin et al.; research credit: M. Lin et al.)
Tag: droplets

Skittering Drops
Drip some ethanol on a hot surface, and you’d expect it to spread into a thin layer and evaporate. But that doesn’t always happen, and a recent study looks at why.
Ethanol is what’s known as a volatile liquid, meaning that it evaporates easily at room temperatures, well below its boiling point. When dropped on a uniformly heated surface above 45 degrees Celsius, the drop contracted into a hemisphere and then began to wander randomly across the surface. Researchers trained an infrared camera on the drop from below (above image), and found an unsteady, roiling motion inside the drop. These asymmetric flows, they concluded, drive the drop’s erratic self-propulsion. They suspect the mechanism may explain why some ink droplets wind up in the wrong place on a page during ink-jet printing. (Image and research credit: P. Kant et al.; via APS Physics)

Spreading Frost
Condensation forms beads of water on a surface. When suddenly cooled, those drops begin to freeze into frost. This video looks at the process in optical and in infrared, revealing the patterns of spreading frost and the tiny ice bridges that link one freezing drop to the next. (Video and image credit: D. Paulovics et al.)

Miniature Ice Stupas
Ice stupas are conical artificial glaciers built with snow cannons; they’re used to store water for spring irrigation. Here, researchers explore a miniaturized lab-grown version made from atomized water droplets. The growing drop breaks and spills, forming frozen fingers in all directions. Further drops flow and freeze as rivulets atop the stupa — or they destabilize and rotate toward another finger, leaving behind a wrinkling shape. Although the formation works very differently (and the scales are completely different) these tiny ice stupas remind me of volcanic flows. (Image credit: D. Papa et al.)

Sliding on Fibers
Water drops slide down spiderwebs, along the spines of desert plants, and across the armored exterior of horned lizards. Thin, grooved surfaces like these pop up frequently in nature when organisms need to direct water. A recent study of droplets sliding on fibers suggests why.
A drop sliding down a fiber is constantly shrinking, leaving a little of itself behind as a thin film that coats the fiber. The thicker a fiber is, the slower the drop moves along it. Similarly, if you bundle multiple fibers together, a drop will travel slower along the thicker bundle. But, to the researchers’ surprise, droplets actually travel faster on bundles than they do along single fibers of the same overall diameter. The key to this result seems to be the tiny grooves between fibers in a bundle. Water fills these areas, creating a “rail” along which the droplets slide more efficiently.
The team hope to put their new insights to use on a water harvester that could help capture precious moisture in arid environments, much like those desert-dwelling plants and lizards do. (Image and research credit: M. Leonard et al.; via Physics World)

Droplet Medusa
Vibration is one method for breaking a drop into smaller droplets, a process known as atomization. Here, researchers simulate this break-up process for a drop in microgravity. Waves crisscrossing the surface create localized craters and jets, making the drop resemble the Greek mythological figure of Medusa. With enough vibrational amplitude, the jets stretch to point of breaking, releasing daughter droplets. (Image and research credit: D. Panda et al.)

Dancing to Chopin
Droplets of paint whirl to Chopin’s “Nocturne Op. 9 No. 2” in this short film from artist Thomas Blanchard. The glitter particles in the paints act as seed particles that highlight the flow within and around each drop. It’s a beautiful dance of surface tension, advection, and buoyancy. (Image and video credits: T. Blanchard; via Colossal)

Linking Size and Origin in Droplets
Respiratory diseases like measles, flu, tuberculosis, and COVID-19 are all transmitted by droplets. Some are tiny and airborne, capable of traveling long distances. Other drops are larger and only capable of traveling short distances. A new review paper consolidates what we know about these droplets and categorizes them by size and origin.
It turns out that a droplet’s size can tell us where it originated in the body. The largest type of droplets come from our mouths, lips, and tongues. Some form from filaments of saliva that stretch across our mouths and burst during exhalation. Others originate in our nasal passages where a sneeze can destabilize the mucus film there. These types of droplets are best suited to transmitting diseases that reside in the upper respiratory tract. Coughing, sneezing, singing, and speaking all produce these droplets, but breathing does not.
In contrast, the smallest classes of droplets come from the bronchial passages of the lungs, where films form after exhalation closes a passage. When we inhale again, the passage reopens, the film breaks up, and tiny droplets flow further into the lungs before getting exhaled. Breathing alone is enough to create and spread these tiny droplets, which are well-suited to spreading diseases that reside deep in the lungs, like tuberculosis.
In between these extremes are medium-sized droplets created from movement around our vocal cords. The formation mechanism for these droplets is least understood, but they are connected to breathing, coughing, speaking, singing, and so on.
Ultimately, understanding the mechanics of disease transmission is about knowing how to best prevent transmission. Knowing the size of droplets responsible for transmission lets us prioritize responses that work. For example, if large droplets are the primary transmission mechanism, loose-fitting masks and face masks will stop the spread. But for smaller droplets, ventilation measures and well-fitted N-95 respirators are the better choice. (Image credit: Anton; research credit: M. Pöhlker et al.; via APS Physics)

Imitating a Cough
Coughing and sneezing create violent air flows in and around our bodies. As that fast air rushes over mucus layers in our lungs, throat, and sinuses, the resulting flow breaks up the mucus into droplets. To explore the details of that process, researchers built a “cough machine” that sends a rush of air over a thin film of water mixed with glycerol. The setup allows them to observe the physics in a way that’s nearly impossible in a human cough or sneeze.

Imitating a cough: high-speed video shows how a thin film made of water and glycerol breaks down in a strong airflow. Parts of the film inflate into hollow bags that form thinner weak spots. When the film breaks in those places, it forms rims and ligaments that create a spray of droplets. As seen above, air flowing past shears the viscous fluid, stretching it out. The leading edge of the film destabilizes and breaks into large drops, but it’s what comes next that really gets things going. Areas of the film inflate to form hollow bags. When sections of the bag thin to about 1 micron, the film ruptures and the bags burst. This triggers a cascade of instabilities in the film’s rim that ultimately rip the film into a spray of tiny aerosol droplets. The researchers found that, despite their tiny size, these droplets collectively carry a large volume of liquid, making them all the more important for understanding transmission of respiratory illnesses. (Image credit: top – A. Piacquadio, experiment – P. Kant et al.; research credit: P. Kant et al.)

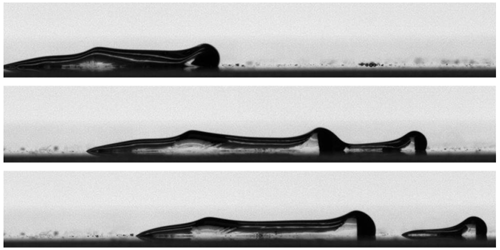
Scooting Droplets
As a child, I always loved watching rain on the windows as I rode in the car. Hemispherical droplets got stretched by the wind flowing over them. But they never stretched smoothly; instead they seemed to shiver and shake unevenly. A recent study looks at a similar situation: drops of glycerin forced to slide along a horizontal surface under the force of the wind. Like the drops on my parents’ car, the glycerin gets stretched out into an elongated oval. Surface waves develop atop the drop and move downstream. The drops, the authors observe, move a bit like a crawling caterpillar, pilling up and smoothing out as they move. (Image credit: rain – A. Alves, experiment – A. Chahine et al.; research credit: A. Chahine et al.; via APS Physics)

This series of images shows an elongated droplet subjected to airflow moving from left to right. Waves form on the drop and move downstream in a fashion similar to a caterpillar crawling.