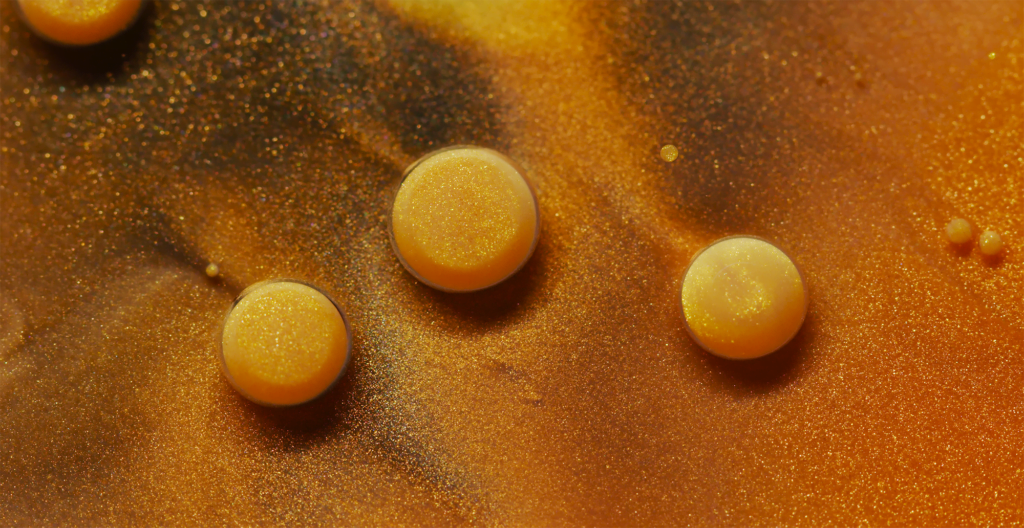
“Emerald and Stone” is filmmaker Thomas Blanchard’s tribute to the music of Brian Eno. The short film is made, as Blanchard puts it, with “inks and painting,” but I suspect there’s some oil in there, too, to coat the droplets we see. Much of the movement is likely driven by surface tension variations in the background fluid. I love the effect this has on the droplets. If you watch closely, some of them appear to rotate like a miniature planet; others have counter-rotating sections within the drop. The difference, I suspect, is one of scale: I think the smaller drops rotate altogether while larger ones develop more complex internal flows. (Video and image credit: T. Blanchard)
Tag: droplets

Gravity Changes Droplet Shapes
With small droplets, gravity usually has little effect compared to surface tension. An evaporating water droplet holds its spherical shape as it evaporates. But the story is different when you add proteins to the droplet, as seen in this recent study.

The protein-filled sessile drop starts out largely spherical, but as the drop evaporates, the concentration of proteins reaches a critical point and an elastic skin forms over the drop. From this point onward, the drop flattens. As a protein-doped droplet sitting on a surface evaporates, it starts out spherical, like its protein-free cousin. But, as the water evaporates, it leaves proteins behind, gradually increasing their concentration. Eventually, they form an elastic skin covering the drop. As water continues to evaporate, the droplet flattens.

For a hanging droplet, the shape again starts out spherical. But as the drop’s water evaporates and the proteins concentrate, it also forms an elastic skin. As the drop evaporates further, the skin wrinkles. In contrast, a hanging droplet with proteins takes on a wrinkled appearance once its elastic skin forms. The key difference, according to the model constructed by the authors, is the direction that gravity points. Despite these droplets’ small size, gravity makes a difference! (Image, video, and research credit: D. Riccobelli et al.; via APS Physics)

How a Leak Can Stop Itself
Some leaks can actually stop themselves, and a new analysis shows how. When a vertical pipe has a small hole, water initially spouts out of it, then dribbles, and, finally, drips as the water level in the pipe falls, decreasing the driving pressure of the flow. But the pipe doesn’t have to empty to a level below the hole for the leak to stop. Instead, a final droplet can form a cap over the hole, with its shape providing enough pressure to balance the remaining pressure from fluid in the pipe.

Water leaking from a vertical pipe transitions from continuous flow to discrete drops (left). Dripping continues until the final droplet forms at t = 0 seconds. The researchers found that the final drop’s kinetic energy (as well as its potential energy) was critical to determining which drop would stop the flow. The last drop behaves like a lightly-damped harmonic oscillator; it needs enough potential energy to counter the flow and a small enough inertia that it doesn’t slip away down the pipe. (Image credit: top – G. Crofte, experiment – C. Tally et al.; research credit: C. Tally et al.; via APS Physics)

Twisted Fibers
A drop sliding down a fiber can do so asymmetrically or symmetrically. The asymmetric configuration is unstable and will spontaneously shift to a symmetric one. Adding a second, parallel fiber stabilizes an asymmetric drop, letting it slide without shifting. And twisting the two fibers together gives even more control, allowing researchers to tweak drop shape, speed, and orientation independent of properties like the drop’s volume or viscosity. (Image and video credit: V. Kern and A. Carlson)

Oil-Covered Bubbles Popping
When bubbles burst, they release smaller droplets from the jet that rebounds upward. Depending on their size, these droplets can fall back down or get lofted upward on air currents that spread them far and wide. Thus, knowing what kind of bubbles produce small, fast droplets is important for understanding air pollution, climate, and even disease transmission.

The jet from a bubble of clean water is broad and slow, releasing fewer and larger drops. In a recent study, researchers compared droplets made by clean, water-only bubbles, and the ones generated from water bubbles with a thin layer of oil coating them. The clean bubbles created jets that were broad and relatively slow moving; this motion produced a few large drops that quickly fell back down.

The jet from an oil-covered bubble is skinny and fast-moving. It produces many small droplets. In contrast, the oil-slicked bubbles made a narrow, fast-moving jet that broke into many small droplets. These droplets could stay aloft for longer periods, indicating that contaminated water can produce more aerosols than clean. (Image credit: top – J. Graj, bursting – Z. Yang et al.; research credit: Z. Yang et al.; submitted by Jie F.)

Giant Droplet Splashes
When droplets get larger than 0.27 cm, they no longer stay spherical as they fall. Here, researchers look at very large droplets (equivalent to 3.06 cm in diameter) falling into water. On their way to the pool, the droplets oscillate — some lengthening, some flattening, and some bulging into a bag. The droplet’s shape at impact (and its speed) determine what shape of splash and cavity form. Wider drops make wider and shallower cavities. (Image credit: S. Dighe et al.)

A 2D Splash
We see plenty of droplets splash when they fall into a pool, but what happens when the drop and pool are two-dimensional? Here researchers captured the familiar process of a splash in an unfamiliar way by looking at a falling drop contained within a soap film. As the drop reached the thicker lower boundary of the soap film (which acts like a pool), its impact sent up ejecta that stretch and curl, much like the three-dimensional splashes we’re accustomed to. (Image credit: A. Alhareth et al.)

A Game of Toss
Over the past few years, we’ve seen lots of droplets bouncing and walking on waves. But today’s example is a little different. In this set-up, the wave is a large standing wave that sloshes from side-to-side in a narrow container. As it does, the wave catches and tosses a large ~3mm water droplet. The system is surprisingly stable, with this game of catch lasting for tens of thousands of cycles and up to 90 minutes before the droplet coalesces. The researchers found that, if the droplet tries to wander from its spot, the oscillating surface wave corrects it, guiding the droplet back to the optimal position. (Image and research credit: C. Sandivari et al.; via APS Physics; submitted by Kam-Yung Soh)

Rocket-Like Supercooled Drops
Many droplets can self-propel, often through the Leidenfrost effect and evaporation. But now researchers have observed freezing droplets that self-propel, too. The discovery came when observing the freezing of supercooled water drops inside a vacuum chamber. The researchers kept losing track of drops that seemingly disappeared. Upon closer inspection, though, they found that the drops weren’t shattering; they were flying away as they froze.
Inside a drop, freezing starts at a point, the nucleation point, and spreads from there. But the nucleation point isn’t always at the center of the drop. This asymmetry, the researchers found, is at the heart of the drop’s propulsion. When ice nucleates, the phase change releases heat that increases the drop’s evaporation rate, which can impart momentum to the drop. For an off-center nucleation, that momentum is enough to send the drop shooting off at nearly 1 meter per second. (Image credit: SpaceX; research credit: C. Stan et al.; via APS Physics)

How Large Particles Get in Sea Spray
When bubbles burst at the ocean’s surface, they eject droplets that can carry high concentrations of contaminants like pollutants, viruses, and microplastics. Previous theories posited that only particles smaller than the microlayer surrounding the bubble could make their way into these drops, but new work shows otherwise.
As bubbles rise to the surface, they carry particles on their surface, collecting them to a concentration that’s even higher than the surrounding seawater. But which particles make it into the air depend on the details of what happens when the bubble pops. Previously, researchers assumed that the thin microlayer of fluid surrounding the bubble was uniform, but that turns out not to be the case. As the bubble pops, some regions of the microlayer stretch and thin, while others grow thicker. The thicker the microlayer, the larger the particles it can pull along. In their single-bubble experiments, the researchers found that 15- and 30-micrometer plastic beads — representing oceanic microplastics — appeared in high concentrations in ejected droplets.

This animated simulation shows how fluid along the edge of a bubble makes its way into ejected droplets. Green particles indicate fluid from the left half of the bubble; blue shows fluid from the right side. Environmental scientists are keen to understand these mechanisms because they link our oceans and atmosphere, potentially affecting rainfall, pollution spread, and epidemiology. (Image, video, and research credit: L. Dubitsky et al.; via APS Physics)