This short film from Macro Room shows how pills dissolve in timelapse. Dissolution is a complex process driven both by flow and chemical concentration. Any small motion in the water helps erode the surface, and as the chemicals dissolve, the subsequent variations in the concentration drive additional flow. This is why we often see a turning point in how quickly the pills come apart. The initial breakdown is slow, but once enough of the pill dissolves, it enhances the surrounding flow, which increases erosion. Notice how many of the pills – liquid-filled capsules, especially – have a point where fluid begins streaming away from them. Unlike the capsules, the solid pills seem to get an extra boost from bubbles that form and then pull away material. (Image and video credit: Macro Room; submitted by clogwog)
Tag: dissolution

The Shaky Life of a Droplet
An evaporating drop of ouzo goes through several stages due to the interactions of oil, alcohol and water. If you turn the situation around by placing a drop of (blue-dyed) water in a mixture of alcohol and anise oil (top image), you get some similarly odd behavior. The drop of water shimmies and grows as alcohol dissolves into it, carrying the occasional oil droplet with it. Eventually, the droplet grows large enough and buoyant enough that part of it detaches and floats to the surface (middle image). If you increase the alcohol ratio in the surrounding fluid, you speed up this process, causing droplets to stream up to the surface (bottom image). (Image and video credit: O. Enriquez et al., source)

Carbonation in Microgravity
Bubbly beverages are popular among humans, but there’s surprising complexity underlying their seemingly simply carbonation, as explored in a new Physics Today article. Most drinks get their bubbles from carbon dioxide, which at higher than atmospheric pressures, can stay dissolved inside water and other liquids. When that pressure gets released, any carbon-dioxide-filled gas cavity in the liquid adopts the allowable saturation concentration for the ambient pressure, which sets up a concentration gradient of carbon dioxide between the liquid and the bubble. That causes carbon dioxide gas to diffuse into the bubbles, making them grow.
Here on Earth, those growing bubbles are buoyant, and they form rising plumes of bubbles. They continue gathering carbon dioxide as they rise, making them grow ever larger (lower left). In microgravity, on the other hand, the bubbles congregate where they form and continue growing through diffusion (lower right). This is one reason carbonated beverages are unpopular in space – instead of rising to the surface and escaping, all the carbon dioxide in a drink gets consumed, leaving astronauts with no way to expel it aside from burping!
For lots more fascinating facts about bubbly drinks – including how they relate to geology! – check out the full Physics Today article. (Image credits: beer – rawpixel; bubbles – P. Vega-Martínez et al.; see also: R. Zenit and J. Rodríguez-Rodríguez)

Dissolving Candy
In nature, solid surfaces often evolve over time in conjunction with the flows around them. This is how stalactites, canyons, and hoodoos all form and change over time. Here researchers examine a surface formed from hard candy that is dissolving from below. Over time, the initially flat surface develops a pitted appearance (top image, scale bar is 1 cm) with roughness that is approximately 1 mm in scale. Flow visualization (bottom row) suggests that these pits result from local flow where narrow, millimeter-sized dense plumes fall away from the surface.
As material dissolves from the candy, it forms a dense layer of sugar-water mixture near the solid surface. Once that layer grows to a critical thickness, it will be too unstable for viscosity to counter. At that point, the Rayleigh-Taylor instability takes over, causing the dense sugar-water layer to break up into narrow, sinking plumes. Although each area is evolving independently, the rate at which material dissolves is uniform everywhere, so the dissolving body retains the same shape over time. (Image and research credit: M. Davies Wykes et al., source)

Scallops and Erosion
Although we often think of solids as immovable in the face of flow, the motion of air and water sculpts many parts of our world. One common pattern, seen both on surfaces that melt and those that dissolve into a flow, is called scalloping. Mathematical analysis shows that flat surfaces exposed to a flow that melts or dissolves them unavoidably develop these scallops. The surface becomes rougher as the scallops form, but the instability that drives them only works up to a specific level of roughness. Instead of the scallops becoming deeper and deeper, the flow shifts as the surface changes. Peaks in the surface erode faster than the valleys, which tends to keep the scallops relatively uniform in depth after they’ve formed. Scallops like these are often seen in soluble rocks like limestone or marble as well as in snow and ice. (Image credit: Seattle Times, G. Smith; research credit: P. Claudin et al., L. Ristroph)

Eroding Candy
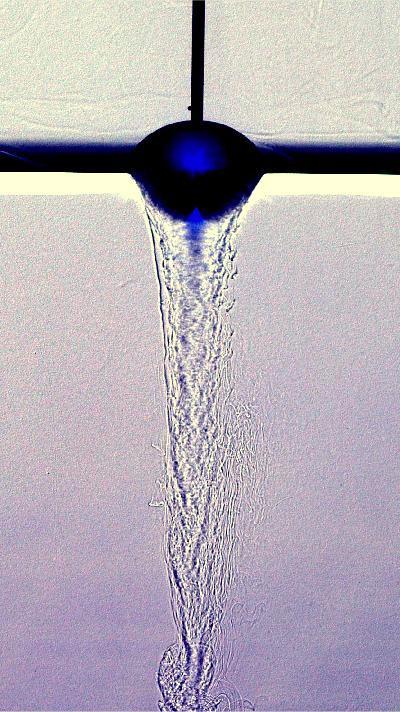
When you pop a hard candy in your mouth, you probably don’t give much thought to the fluid dynamics involved in dissolving it. The series above shows a hard candy suspended in water being slowly eaten away. As sugars in the candy dissolve into the water, the fluid becomes denser and falls away. This creates the downward flow visible in the center of the image. As sugar-laden water sinks, fresher water is pulled in alongside the walls of the candy. That flow helps erode the candy, creating a rougher surface. Since rough surfaces have a greater surface area exposed (than a smooth surface), they prompt further and faster dissolution. That strengthens the downward flow, pulls in more ambient water, and keeps the whole process going. (Image credit: M. Wykes)

The Colorful Dissolution of Candies
Many solids can dissolve in liquids like water, and while this is often treated as a matter of chemistry, fluid dynamics can play a role as well. As seen in this video by Beauty of Science, the dissolving candy coating of an M&M spreads outward from the candy. This is likely surface-tension-driven; as the coating dissolves, it changes the surface tension near the candy and flow starts moving away thanks to the Marangoni effect. With multiple candies dissolving near one another, these outward flows interfere and create more complex flow patterns.
These flows directly affect the dissolving process by altering flow near the candy surface, which may increase the rate of dissolution by scouring away loose coating. They can also change the concentration of dissolved coating in different areas, which then feeds back to the flow by changing the surface tension gradient. (Video and image credit: Beauty of Science)

Dissolving
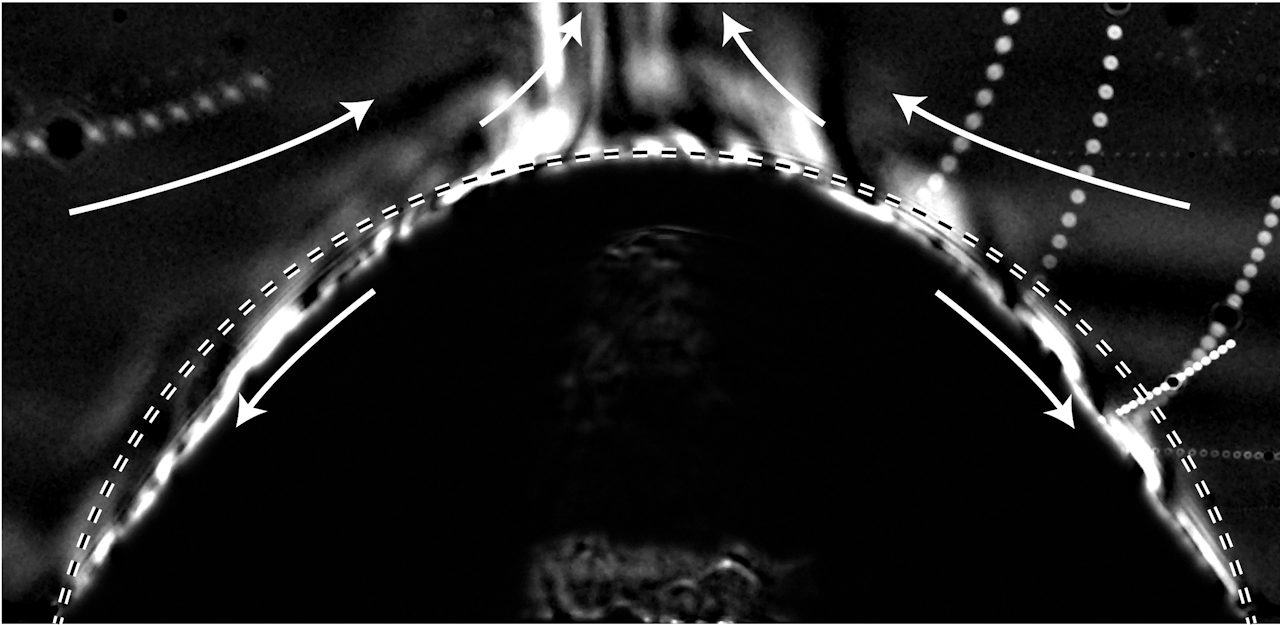
It looks like the fiery edge of a star’s corona, but this photo actually shows a dissolving droplet. The droplet, shown as the lower dark region in this shadowgraph image, is a mixture of pentanol and decanol sitting in a bath of water. Pentanol is a type of alcohol that is fully miscible with decanol and is water soluble, so that it will dissolve into the surrounding water over time. Decanol, on the other hand, is immiscible with water, so that part of the droplet won’t mix with the surrounding water.
The bright swirls along the droplet’s edge show areas with more pentanol. As the alcohol dissolves into the water, it forms a buoyant plume at the top of the droplet that rises due to pentanol’s lower density. That rising plume draws fresh water in from the sides, shown by the upper white arrows. Inside the droplet, flow moves in the opposite direction, from the top toward the outer edges. This is a result of uneven surface tension within the droplet. Scientists are interested in understanding the dynamics of these multiple component drops for applications like printing, where it’s desirable for pigments in an ink drop to be distributed evenly as the drop dries. (Image credit: E. Dietrich et al.)

How Many Licks Does It Take to Get to the Center of a Lollipop?
Many a child has wondered how many licks it takes to get to the center of a lollipop. Physically, this is a problem of a solid body dissolving in a flow, and it’s one scientists are interested in for its geological, industrial, and pharmacological applications.
The animation above shows flow around a dissolving (candy!) body that was originally spherical. With both spheres and cylinders, the final shape the body takes is consistent – it has a front boundary with a curvature of nearly constant radius and a back face that is approximately flat. This creates a boundary layer of uniform thickness across the front face, and that uniform flow makes the surface dissolve steadily and evenly so that it maintains the same overall shape.
With their model and experiments, researchers have even been able to tackle the classic question of how many licks it takes:
“For candy of size 1 cm licked at a speed of 1 cm/s, we estimate a total of 1000 licks, a prediction that is notoriously difficult to test experimentally.”
(Image credit: J. Huang et al., source, pdf)

Pinning a Drop
The shape of a droplet sitting on a surface depends, in part, on its surface tension properties but also on the nanoscale roughness of the surface. Small variations in the height and shape of the surface will change the area a drop contacts as well as the contact angle the edge of the drop makes with the surface. If the contact line between the drop and surface stays the same as a droplet evaporates into the surrounding gas or dissolves into the surrounding liquid, then we say the drop is pinned. A pinned drop’s contact angle will decrease as the drop’s volume decreases. This strains the ability of the nanoscale roughness to keep the drop’s edge pinned. As individual points of contact fail, the drop’s edge may jump inward to a new contact point. This set of discrete jumps between pinned states is called a stick-jump or stick-slip mode. (Image credit: E. Dietrich et al., source; see also: E. Dietrich et al. 2015)