Unusually high rainfall in Bolivia’s Salar de Uyuni turned the world’s largest salt flat into a shallow salt lake. These natural-color satellite images show the area in late January 2022. If you zoom in on the full resolution image, there are incredible detailed swirls in the water. It’s like peering at an abstract or Impressionist painting. The many colors are attributable to several sources, including volcanic sediments, runoff, and a variety of microbes and algae thriving in the mineral-filled waters. (Image credit: L. Dauphin; via NASA Earth Observatory)
Tag: dissolution

Dissolving Pinnacles
Limestone and other water-soluble rocks sometimes form sharp stone pinnacles like the ones seen here in Borneo. Scientists have recreated these structures in the laboratory simply by immersing water-soluble substances (essentially blocks of candy) into water. Without any background flow, the blocks will slowly form these pinnacle forests as material dissolves into the nearby water, creating a heavy solute-rich fluid that sinks down the exterior of the block. The convection generated by this dissolution drives the material into these sharp shapes, as shown mathematically in this recent study. (Image credit: N. Naim; research credit: J. Huang and N. Moore; via APS Physics)

Conserving the Hill House
In 1904, Scottish architect Charles Rennie Mackintosh created the Hill House, a masterpiece of modern design decades ahead of its time. Unfortunately, the Portland cement used for the house’s exterior has not held up well to a century of Scottish rains. As water saturated the cement, it began to dissolve and crumble. So to save the property, conservators commissioned the giant chainmail Box that currently surrounds the house. It protects the house from rain while allowing ventilation that dries the house out slowly. As an added bonus, the superstructure allows visitors to appreciate the house from new angles. (Video credit: T. Scott; via Colossal)

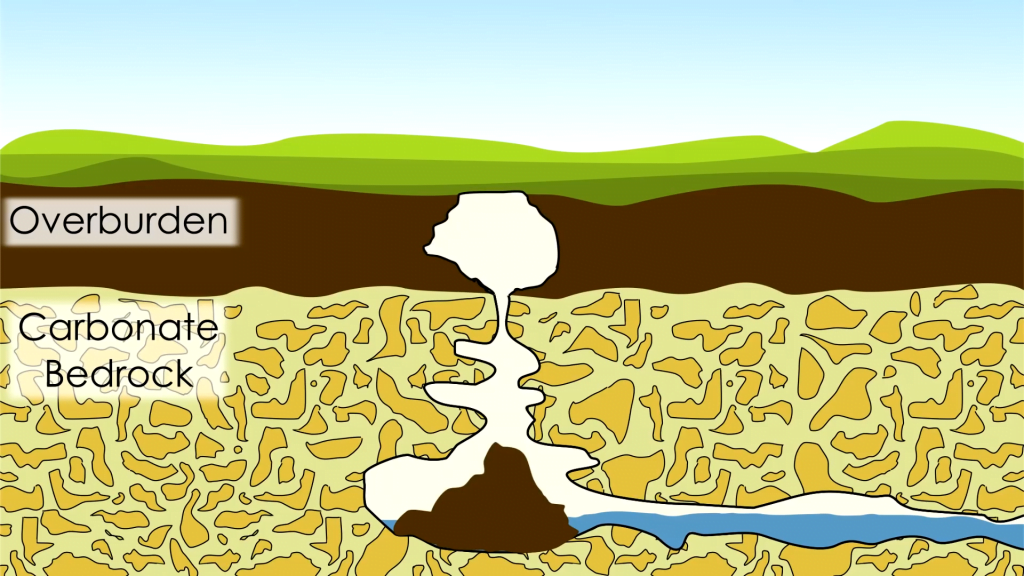
How Sinkholes Form
Growing up in the Ozarks, I explored my fair share of caves and sinkholes. These geological features form when flowing groundwater erodes soil, sand, and even rock underground. The Ozark Plateau consists largely of limestone, which is water soluble, making it very prone to this internal erosion. As bedrock dissolves away, it is eventually unable to hold up the weight of ground above it, causing a catastrophic collapse into a sinkhole. Although my childhood sinkholes were naturally occurring, they can also form in spots where leaking pipes and infrastructure help wash underlying soil away. Unfortunately for engineers, this internal erosion can take place for years without any visible sign above ground. (Image and video credit: Practical Engineering)

Uncovering Erosion Patterns
Gypsum and limestone cliffs sometimes form patterns of long, parallel grooves known as rillenkarren. Recent research shows that these patterns form when a thin layer of water flows over a dissolvable surface. As the running water picks up solute, its concentration increases, causing changes in the local hydrodynamics. What begins as a small perturbation in an otherwise flat surface grows into a groove with walls that eventually rise out of the water layer. At that point, the growth mechanism shifts because the flow is restricted to channels in the rock. (Image credit: Ymaup/Wikimedia Commons; research credit: A. Guérin et al.; via APS Physics; submitted by Kam-Yung Soh)

Dissolving Caramel
In nature, erosion patterns are driven by the interactions of flow and topography. Here, researchers study that process in the lab by placing an inclined block of caramel in quiescent syrup and watching as it dissolves. Initially, the bottom surface of the block develops regularly-spaced plumes — the dark lines seen in the first image. But because the caramel-laden plumes are heavier than the surrounding fluid, the flow quickly becomes unstable. The plumes cross one another and begin to carve chevrons into the caramel.
The chevrons appear to march their way upward in the video. They slowly grow and change into a distinctly scalloped pattern. Scallops like these are often seen by geologists in caves and icebergs, and the authors argue that their results and modeling indicate the importance of buoyant flow effects on such natural formations. (Image and research credit: C. Cohen et al.)

Replacing Injections With Pills
In medicine, many medications contain molecules too large to be easily absorbed through the intestinal wall, so these so-called biologics — like the insulin administered to diabetics — are injected into the body. Researchers are studying ways that such injections could eventually be replaced with pills, but there are plenty of challenges involved.
Some substances, known as transient permeability enhancers, allow the intestines to absorb larger molecules, but they work for only tens of minutes, which means researchers must understand how and when to administer them relative to the medication they help patients absorb. To do so, researchers are building computational fluid dynamics models of the human digestive system so that they can better understand how and when different kinds of pills break down in the body. (Image credit: Macro Room, source; via CU Engineering; submitted by Jenny B.)

Paint Versus Hydrogel
In this bizarre short film, we get to see a battle between dissolution and absorption. I think the Chemical Bouillon team has coated hydrogel beads in a layer of paint and then immersed them in water. As the beads absorb water, they expand and grow, tearing their fragile outer layer of paint to smithereens.
One thing that struck me when watching several of the sequences is just how regular the hole spacing in the paint is for the round hydrogels. That hints at an orderly breakdown in the solid paint layer while the interior hydrogel polymer symmetrically expands. It’s a little like watching holes grow in a splash curtain. (Video and image credit: Chemical Bouillon)

Turning a Corner in Microfluidics
Over the past couple decades, microfluidic devices have become a staple of medical and biological diagnostics and analysis. Tests that once required large and specialized equipment can now be completed closer to a patient, using only a few drops of sample fluid. Running multiple tests on a single chip can become difficult, though, since flow through the device tends to dissolve and mix the dried reagents used for tests. But a new method cleverly uses fluidic forces to keep reagents separated without the need for complicated microfluidic structures.
The basic concept is outlined in the illustration above. You’re looking down on a microfluidic channel that’s long and very thin. A shallow groove down the middle serves as a barrier by pinning the contact line of the incoming fluid. So when the sample fluid flows in through the inlet on the left, it will only fill the top half of the cell. When it reaches the far right side, it turns the corner and flows to the left, encountering the first of the dried reagents it must dissolve for the device’s tests. The fluid will fill the lower channel quickly and then come to rest while the reagents dissolve.
With both sides of the channel full of liquid, the shallow barrier can no longer hold, and the fluid will take up the full width of the channel, with two well-dispersed – but separated – regions of reagents. Once that’s happened, a valve – represented by the pale blue line near the right side of the illustration – releases the fluid into the next section of the chip, allowing the analysis to proceed. (Image credit: Nature; research credit: O. Gökçe et al.; submitted by Kam-Yung Soh)

The Disappearing Cotton Candy
Moisture is cotton candy’s natural enemy. The spun sugar dissolves incredibly quickly under the influence of even a couple drops of water. Why that’s so is clearer when looking at a single fiber. Inside the droplet there’s a gradient in the sugar concentration. The more sugary water sinks, and the sugar fiber dissolves more quickly in the upper part of the droplet, where the less sugary water can more easily take up new sugar.
Once the fiber breaks, capillary forces draw the droplet upward, giving it a fresh section of fiber to dissolve. In a web of fibers, this process can pull droplets apart and together as they quickly eat through the spun sugar. (Image and video credit: S. Dorbolo et al.; submitted by Alexis D.)