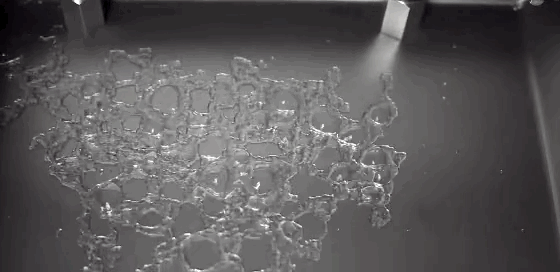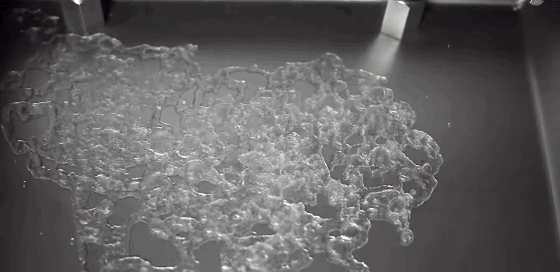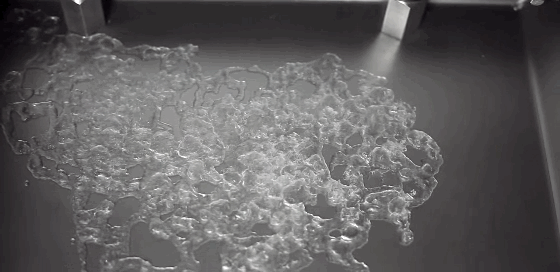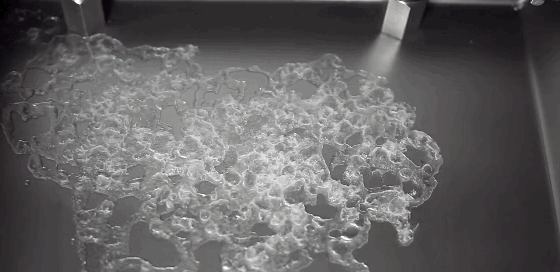Boiling is a process most of us don’t pay much attention to. But it can be remarkably entertaining and beautiful. This award-winning video shows boiling on and around a heated wire immersed in oil. Depending on the diameter of the wire and the power used to heat it, the researchers observe several different regimes of behavior. In one, vapor bubbles form on the wire and interact with one another: bouncing, merging, and dancing back and forth. When the bubbles become large enough, their buoyancy lifts them upward. In another regime, the wire is hot enough for film boiling. Like the Leidenfrost effect, film boiling occurs when a surface is so hot that it instantly vaporizes any liquid near it. The vapor layer then acts like coating, insulating the remaining liquid from the hot surface. The bubbles formed on the wire in this regime are mesmerizing, rising in periodic patterns or shifting back and forth gobbling up lesser bubbles. (Video credit: A. Duchesne et al.)
Tag: boiling

Boiling Water in Oil
Most people know that throwing water into hot oil is a bad idea. But, as dramatic as the results can be, the boiling of a water droplet submerged in oil is remarkably beautiful, as seen in the animations above. The initial water droplet expands as it shifts from liquid to vapor (top). At a critical volume, the expansion occurs explosively (middle), causing the bubble to overexpand relative to the pressure of the surrounding fluid. The higher pressure of the oil around it collapses the drop, which then re-expands, creating the cycle we see in the final two animations. This oscillation triggers a Rayleigh-Taylor type instability along the bubble’s interface, causing the surface corrugations observed. The vapor bubble will continue to rise through the oil, eventually breaking the surface and scattering hot oil droplets. (Image credits: R. Zenit, source)

Reader Question: When Mercury Meets Lava
Reader lucondri asks:
What happens when mercury touches lava?
That’s an interesting thought experiment, but hopefully no one tries it any time soon given mercury’s toxicity. So, what might happen? Mercury has a boiling point just under 630 Kelvin, and, although the temperature of molten lava varies, it’s between 970 and 1470 Kelvin when it first erupts. So mercury would definitely vaporize (i.e. boil) on contact with lava. (Again, this is very bad for anyone nearby.) If you’re curious what boiling liquid mercury looks like, wonder no further.
Molten lava is much, much hotter than the boiling point of mercury, though, so there’s a possibility that the mercury won’t boil away instantly. This is because of the Leidenfrost effect, where a thin layer of vapor forms between a liquid and an extremely hot surface. The vapor has such low friction that the liquid can essentially skate across a surface, and it doesn’t boil away instantly because the vapor insulates it from the extreme heat. After some digging, I found a paper that placed the Leidenfrost temperature of mercury between about 850 and 950 Kelvin, meaning that fresh lava is probably hot enough to generate mercury Leidenfrost drops.
So pouring a lot of mercury on lava will probably result in some boiling, but there’s also a good chance that it will form a bunch of skittering mercury droplets that will stick around awhile before they evaporate into toxic mercury gas. That said, it’s a lot easier and safer to watch awesome Leidenfrost drop videos with other liquids. (Collage credit: N.Sharp; images sources: Z. T. Jackson, and A.Biance)

Does Liquid in a Vacuum Boil or Freeze?
What happens to a liquid in a cold vacuum? Does it boil or freeze? These animations of liquid nitrogen (LN2) in a vacuum chamber demonstrate the answer: first one, then the other! The top image shows an overview of the process. At standard conditions, liquid nitrogen has a boiling point of 77 Kelvin, about 200 degrees C below room temperature; as a result, LN2 boils at room temperature. As pressure is lowered in the vacuum chamber, LN2’s boiling point also decreases. In response, the boiling becomes more vigorous, as seen in the second row of images. This increased boiling hastens the evaporation of the nitrogen, causing the temperature of the remaining LN2 to drop, the same way sweat evaporating cools our bodies. When the temperature drops low enough, the nitrogen freezes, as seen in the third row of images. This freezing happens so quickly that the nitrogen molecules do not form a crystalline lattice. Instead they are an amorphous solid, like glass. As the residual heat of the metal surface warms the solid nitrogen, the molecules realign into a crystalline lattice, causing the snow-like flakes and transition seen in the last image. Water can also form an amorphous ice if frozen quickly enough. In fact, scientists suspect this to be the most common form of water ice in the interstellar medium. (GIF credit: scientificvisuals; original source: Chef Steps, video; h/t to freshphotons)

Explosive Boiling
A superheated liquid can reach temperatures higher than its boiling point without actually boiling – similar to how liquids can be supercooled below their freezing point without solidifying. The photo sequence above shows how explosive the boiling of a superheated water droplet submersed in sunflower oil can be. Image (a) in the lower left shows the superheated droplet resting on the bottom of its container. Then droplet vaporizes explosively in (b), expanding dramatically. The bubble overexpands and and begins to oscillate around its equilibrium radius. This triggers a Rayleigh-Taylor instability in the bubble’s interface, creating the large lobes in © and enlarged in the upper image. Finally, the bubble fragments in (d). See the original paper for more on superheated droplet boiling. (Image credit: M. A. J. van Limbeek et al.; via @AIP_Publishing)

Dynamic Leidenfrost Impact
The Leidenfrost effect occurs when a liquid encounters a solid object much hotter than the liquid’s boiling point, like when water skitters on a hot griddle or someone plunges a hand in liquid nitrogen. A thin layer of vapor forms between the liquid and the solid, thereby (briefly) insulating the remaining liquid. The Leidenfrost effect can be static–like a droplet sitting on a pan–or dynamic, like the video above in which a droplet impacts the hot object. The video shows both a top and a side view of a droplet striking a plate that is over five times hotter than the liquid’s boiling point. On impact, the droplet spreads and flattens, and a spray of even tinier droplets is ejected before rebound. (Video credit: T. Tran and D. Lohse, from a review by D. Quere)

Peering Inside the Kettle
Here natural convection is explored experimentally in a quasi-2D environment. The researchers demonstrate how this phenomenon, which is much like that seen in a boiling pot, can be investigated by measuring the refractive distortions caused by the thin heated fluid layer. They also demonstrate types of boiling that can occur. Typically, bubbles nucleate at the heated surface and then rise to pull hot fluid with them. At high enough temperatures above the liquid’s boiling point, however, an unstable layer of vapor can form over the heated surface. This “boiling crisis” or critical heat flux produces a marked reduction in heat transfer due to the insulation provided by the vapor layer. (Video credit: S. Wildeman et al.)

Boiling Without Bubbles
Water droplets sprinkled on a sufficiently hot frying pan will skitter and skate across the surface on a thin layer of vapor due to the Leidenfrost effect. When a solid object is much warmer than a liquid’s boiling temperature, the surface is surrounded by a vapor cloud until the solid cools to the point that the vapor can no longer be sustained. Then the vapor breaks down in an explosive boiling full of bubbles. Unless, as researchers have just published in Nature, the solid is treated with a superhydrophobic coating. The water-repellent surface prevents the bubbling, even as the sphere cools. The technique could be used to reduce drag in applications like the channels of a microfluidic device. (Video credit: I. Vakarelski et al.; see also Nature News; submitted by Bobby E)

Creating Lava
In Syracuse, NY, artists and scientists work together to study volcanic flows by melting crushed basalt in a special furnace before releasing the lava into the parking lot. This particular flow is very prone to boiling behavior, likely because of the cold air and ground temperatures (less than 0 C). The outer layers of rock cool quickly, leaving bubble-shaped chambers which hotter lava can fill before melting out. (via It’s Okay To Be Smart; submitted by @jpshoer)

Boiling in Microgravity
This week’s edition of the ISS research blog focuses on the Boiling Experiment Facility (BXF) and the goals of unlocking the secrets of boiling in microgravity. Without gravity to provide buoyant convection, boiling in space tends to produce one giant bubble instead of the hundreds of tiny ones we’re accustomed to seeing on our stoves. According to Dr. Tara Ruttley:
TheBoiling Experiment Facility or BXF, which launched on STS-133 in February 2010, will enable scientists to perform in-depth studies of the complexities involved in bubble formation as a result of heat transfer. For instance, what roles do surface tension and evaporation play during nucleate boiling when buoyancy and convection are not in the equation? What about the variations in the properties of the heating surface? By controlling for gravity while on the International Space Station, scientists can investigate the various elements of boiling, thus potentially driving improved cooling system designs. Improved efficiency in cooling technology can lead to positive impacts on the global economy and environment; two hot topics that have much to gain from boiling in space.