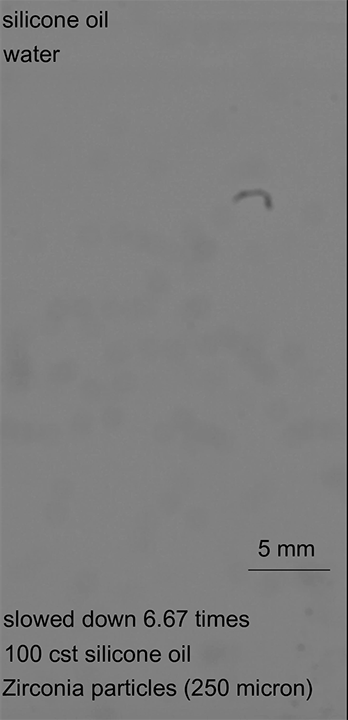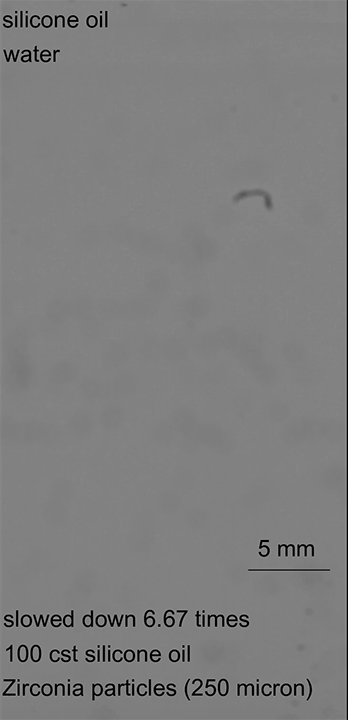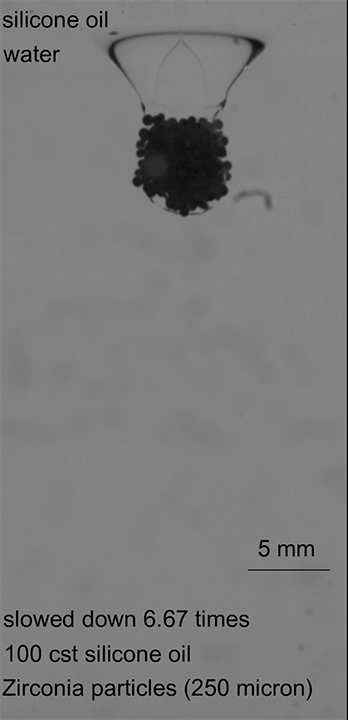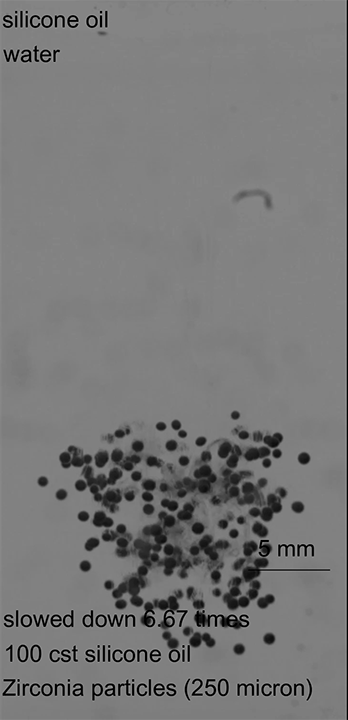
When a drop of soap falls into a pool of water, its surface-loving molecules spread out on the water’s surface. Exactly how the soap spreads depends on the local concentration of its surfactant molecules, which create areas with different surface tensions that cause flow. All in all, it’s a tough process to predict because it varies in time at every point on the pool. But a recent paper offers a new class of exact solutions for the problem.
The paper considers a surfactant-laden droplet spreading over a (relatively speaking) deep pool. Other researchers showed recently that this situation can be described with a complex version of the Burgers’s equation, which was originally developed to describe turbulent flows. The authors solved the equation for a variety of initial conditions and found that the time-dependent spread of the surfactants was sensitive to the initial surface distribution. The higher the initial surface concentration, the faster the surfactants spread. (Image credit: T. Despeyroux; research credit: T. Bickel and F. Detcheverry; via APS Physics; submitted by Kam-Yung Soh)