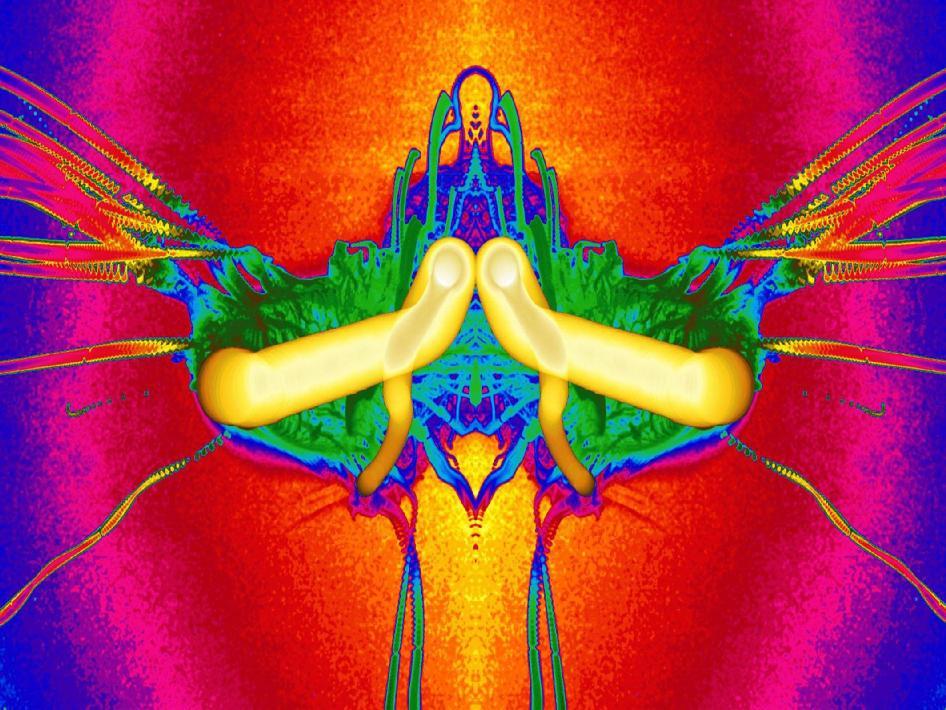
Combustion in microgravity is markedly different than that on earth, due to a lack of buoyant convection. The combustion of a droplet of heptane is shown here as a composite image. The bright yellow structure shows the path of the droplet, which gets smaller as it burns. The green structures show the initial development of soot, which eventually streams outward as long streaks. # (submitted by jshoer)
Search results for: “droplet”

Stereo Liquid Sculpture
This stereo 3D photo shows the Worthington jet ejected when a droplet impacts a pool. The flat crowning drop is formed from an ejected droplet colliding with a falling droplet.

Giant Water Balloon Physics
Playing with a giant water balloon and high-speed cameras is like a giant experiment in surface tension, right up until the tensile strength of the balloon comes into play. The rippling in the balloon is reminiscent of the motion of droplet breakup or impact on superhydrophobic surfaces. (submitted by Daniel B)

Jet Breakup
A non-cylindrical stream falling through a slit nozzle exhibits the Plateau-Rayleigh instability, which drives a falling jet of fluid to break into droplets due to surface tension. The fingers formed off the falling stream may be a form of Rayleigh-Taylor instability. #

Happy Anniversary
ESA astronaut Pedro Duque shown refracted through a water droplet in microgravity. Today marks the 50th anniversary of human space flight. #

Liquids Lens Breakup
A decane liquid lens floating on water (think drops of fat in chicken soup) displays different breakup and pinch-off than seen in three-dimensional droplet breakup. The pinch-off process in two dimensions relies on line tension rather than surface tension, and the quasi-2D liquid lens system is somewhere between these. The video above is a magnification of the filament connecting one liquid lens as it is broken into two smaller liquid lenses (the dark areas on the left and right of the screen). # (via scienceisbeauty)

Liquid Acrobatics
Imagine blowing through a straw into a nearly empty glass–we probably all did this as children and sent water, milk, and soda flying everywhere! In essence, this video shows that same act, but filmed by a high-speed camera. The “straw” blows a steady stream of helium into a shallow pool of silicone oil and slowly moves so that the angle the straw makes with the pool changes. As the angle changes, different regimes are visible. First waves appear on the surface of the pool, then a bulge forms, which develops into a droplet stream, then on into the chaos of bubbles and jets. It’s good I couldn’t see this in slow motion as a child or I would have never used my straw for drinking!

High-Speed Cooking
I suspect demonstrating fluid mechanics was not what this cookbook had in mind when they filmed creamer poured into coffee at 2000 fps, but there’s some awesome droplet breakup, crowning, roiling turbulent mixing, and even some deformed Worthington jets here. It’s a reminder that, even though we may not notice it, fluid dynamics are all around.

High-Speed Leidenfrost Levitation
The Leidenfrost effect occurs when a liquid encounters a surface with a temperature much higher than its boiling point. Some of the liquid is instantly vaporized and then a droplet will skate across the surface on that vapor. This video shows the process at 3000 frames per second.

Reader Question: Surface Tension vs. Viscosity
lazenby asks:
How can superfluid liquid Helium have zero viscosity while retaining surface tension? (assuming something like surface tension is required for a liquid to form drops)
The short answer is that surface tension and viscosity are two totally separate properties for a fluid. To illustrate how one can exist without the other in a superfluid, we’ll imagine two different scenarios. For the first, imagine that you have a narrow vertical pipe. Any fluid you put in the pipe will flow downward due to the force of gravity. If you put water through the pipe, you’ll get some rate of outflow. Now imagine putting something like molasses through the pipe. Even with the same external forces on it, the molasses will never move through the pipe as quickly as the water does. This is because the molasses has higher viscosity and resists flowing. In a force balance, viscosity would act like friction, opposing the downward motion of the fluid.
Surface tension arises from a different balance of forces. Now imagine that you have a stationary droplet of one fluid (A) floating in a different fluid (B). Deep inside the droplet, each molecule of Fluid A is being pulled on all sides by other identical molecules of Fluid A. A molecule at the surface of the droplet, though, doesn’t experience that neighborly pull on all sides; it experiences different intermolecular forces from Fluid B. Our imaginary droplet is stationary, though, so all the forces on it and all the forces on its individual molecules have to balance, otherwise there’d be acceleration. Surface tension acts along the interface by pulling molecules of Fluid A in toward one another–much like the elastic of a balloon–thereby balancing the forces in the droplet and equalizing the force across the interface between Fluid A and Fluid B. (Illustration credit: Wikipedia)
In the superfluid, this balance of forces across the interface between air and helium-3 must still exist, despite the superfluid’s lack of viscosity.