When still, water drops sitting on a surface are roughly hemispherical, drawn into that shape by surface tension. But on a vibrating surface, the same water drop displays many different shapes, like those in the video above. Researchers have observed more than 30 different mode shapes by varying the driving frequency. The metal mesh placed beneath the glass on which the drops sit helps the researchers determine the drop’s shape. As the drop deforms, the mesh appears to distort due to the refraction of light through the changing shape of the drop’s water-air interface. The distortion allows observers to visualize (and in some experiments even reconstruct) the shape of the drop’s surface. Understanding this kind of droplet behavior is valuable for many applications, including ink-jet printing and microfluidic devices. (Video credit: C. Chang et al.; via Science)
Search results for: “droplet”

Drop-Tower Droplets
A microgravity environment can cause some nonintuitive behaviors in fluids. Many of the effects that dominate fluid dynamics in space are masked by gravity’s effects here on Earth. As a result, it can be very difficult to predict how seemingly straightforward technologies like heat exchangers, refrigeration units, and fuel tanks will behave. The photos above show two bubble jets–created by injecting a liquid-gas mixture into a liquid–colliding in microgravity. This particular experiment was conducted in a drop tower rather than on-orbit, which produced some side effects like the large bubbles seen in the images. These were created by the coalescence of smaller bubbles that congregated near the top of the tank shortly before the experiment attained free-fall. (Photo credit: F. Sunol and R. Gonzalez-Cinca)

Reader Question: Non-Coalescing Droplets
Reader ancientavian asks:
I’ve often noticed that, when water splashes (especially as with raindrops or other forms of spray), often it appears that small droplets of water skitter off on top of the larger surface before rejoining the main body. Is this an actual phenomenon, or an optical illusion? What causes it?
That’s a great observation, and it’s a real-world example of some of the physics we’ve talked about before. When a drop hits a pool, it rebounds in a little pillar called a Worthington jet and often ejects a smaller droplet. This droplet, thanks to its lower inertia, can bounce off the surface. If we slow things way down and look closely at that drop, we’ll see that it can even sit briefly on the surface before all the air beneath it drains away and it coalesces with the pool below. But that kind of coalescence cascade typically happens in microseconds, far too fast for the human eye.
But it is possible outside the lab to find instances where this effect lasts long enough for the eye to catch. Take a look at this video. Here Destin of Smarter Every Day captures some great footage of water droplets skittering across a pool. They last long enough to be visible to the naked eye. What’s happening here is the same as the situation we described before, except that the water surface is essentially vibrating! The impacts of all the multitude of droplets create ripples that undulate the water’s surface continuously. As a result, air gets injected beneath the droplets and they skate along above the surface for longer than they would if the water were still. (Video credit: SuperSloMoVideos)

Droplets Within Droplets
This video shows a multi-layered droplet, in which several droplets are formed one inside the other as an initial drop falls through a layer of oil sitting atop another liquid. When the drop falls, its potential energy gets transformed into interface energy, creating a fascinating interplay of surface tension, deformation, and miscibility between the fluids. Such self-contained multi-layered droplets, similar to multiple emulsions, could be helpful in pharmaceutical development. (Video credit: E. Lorenceau and S. Dorbolo 2004)

Dye Droplet
A drop of fluorescent dye falling into quiescent water forms fantastical structures that are a mixture of vorticity, turbulence, and molecular diffusion. The horseshoe-like shape near the front of the drop is a typical shape for two fluids strained by moving past one another. The main section of the drop billows outward like a parachute, but the turbulence of its wake stretches the dye into fine threads that quickly disperse in the water. (Photo credit: D. Quinn et al.)

Droplet Bounce
This high-speed video shows the remarkable resilience of a water droplet upon impact against as a solid surface. The droplet deforms into a pancake-shape, with its center depressing almost flat before rebounding upward. The rest of the drop follows, splitting into several droplets as capillary waves dance across its surface. When one satellite drop almost escapes, the main droplet just barely comes in contact with it, the coalescence enough to tip surface tension into pulling them together instead of breaking them apart. (Video credit: K. Suh/ChemistryWorldUK)

Breaking into Droplets
A falling column of liquid, like the water from your faucet, will tend to break up into a series of droplets due to the Plateau-Rayleigh instability. This instability is driven by surface tension. Small variations in the radius of the column occur naturally. Where the radius shrinks, the pressure due to surface tension increases, causing liquid to flow away, which shrinks the column’s radius even further. Eventually the column pinches off and breaks into droplets. What’s especially neat is that the size of the final droplets can be predicted based on the column’s initial radius and the wavelength of its disturbances. (Video credit: BYU Splash Lab)
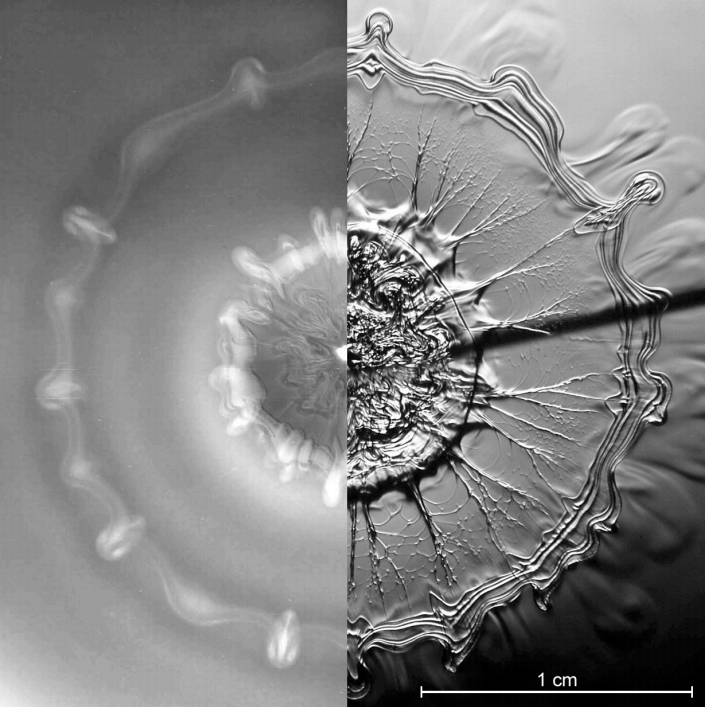
Droplet Impact Visualized
When a drop falls from a moderate height into a shallow pool, its impact creates a complicated pattern. The photo above is a composite image showing a top-down view 100 ms after such an impact. On the left side, the flow is visualized using dye whereas the right shows a schlieren photograph, in which contrast indicates variations in density. Both methods show the same general structure – an inner vortex ring generated at the edge of the impact crater and formed mostly of drop fluid and an outer vortex ring, consisting primarily of pool fluid, formed by the spreading wave. Both regions show signs of instability and breakdown. (Photo credit: A. Wilkens et al.)

Encapsulating Droplets
In applications like drug delivery, it’s often desirable to encapsulate one or more liquid droplets in an additional immiscible fluid. These drops-within-drops, called double emulsions, are typically a multi-step process, created from the innermost drop outward. In this new microfluidic technique, though, researchers are able to create multi-component emulsions in a single step. A double-bored capillary tube creates the two inner droplets (both water, dyed different colors) while oil flows down the outside of the injection tube to encapsulate the droplets. The multi-component double emulsions then flow as one to the right in the outer carrier fluid. The spacing of the capillary tubes is critical to prevent the inner droplets from coalescing with one another. (Video credit: L. L. A. Adams et al.)

Shocking Droplets
Typical liquid drops will break apart into long, stretched ligaments and a spray of tiny droplets when deformed. But with just a small addition of polymers, these same liquids become viscoelastic and capable of some pretty incredible behaviors. This video shows a viscoelastic drop being struck by a shock wave that passes from right to left. The droplet is smashed and deformed, then stretches into jellyfish-like sheet of liquid. But incredibly, the elastic forces in the droplet are enough to hold it together. Researchers are interested in understanding these behaviors for many applications, including preventing accidental explosions caused by explosive fuels atomizing in air. (Video credit: T. Theofanous et al.)