Over the past few years, we’ve seen lots of droplets bouncing and walking on waves. But today’s example is a little different. In this set-up, the wave is a large standing wave that sloshes from side-to-side in a narrow container. As it does, the wave catches and tosses a large ~3mm water droplet. The system is surprisingly stable, with this game of catch lasting for tens of thousands of cycles and up to 90 minutes before the droplet coalesces. The researchers found that, if the droplet tries to wander from its spot, the oscillating surface wave corrects it, guiding the droplet back to the optimal position. (Image and research credit: C. Sandivari et al.; via APS Physics; submitted by Kam-Yung Soh)
Search results for: “art”

Cellular Clouds
Though tough to make out from the surface, our oceans are often covered by cell-shaped clouds stretching thousands of kilometers. This satellite image shows off two such types of marine stratocumulus cloud. Open-celled clouds appear as thin wisps of vapor around an empty middle; in these clouds, cool air sinks through the center while warm air rises along the edges. Open-celled clouds are good rain producers.
On the flip side, closed-cell clouds have a vapor-filled center and breaks in the cloud cover along each cell’s edge. These clouds don’t produce much rain, but they do lift warm, moist air through their middles and let cool air sink along their edges. Closed-cell clouds tend to last much longer than their open-celled counterparts; they can stick around for half a day, whereas open-celled clouds break up in only a couple hours. (Image credit: J. Stevens; via NASA Earth Observatory)

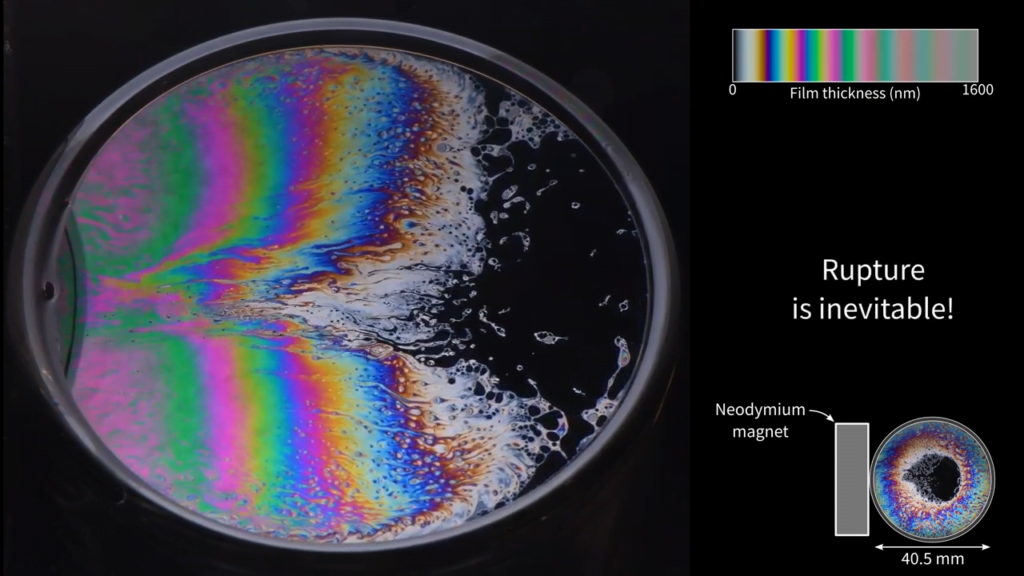
Magnetic Soap Films
Soap films naturally thin over time as fluid evaporates and differences in film thickness cause surface-tension-driven flows. In this video, researchers experiment with adding magnetic nanoparticles to the soap film. In the second image, the white structures near the center of the film contain nanoparticles, and they’re drawn toward the magnet that sits (out-of-frame) to the left of the film. With more nanoparticles and a stronger magnetic field (Image 3), the entire soap film takes on a distinctive profile that thins from left to right. The effect is so strong that the film quickly thins to the point of rupture. (Image and video credit: N. Lalli et al.)

Wreathed
A woman hides in silt and sediment in this award-winning underwater photo by Lee Jongkee. The motion of her plunge sends water spinning downward, where it picks up particles from the ground. Slow to settle, the sediment forms an ethereal mask for the swimmer. See more of the 2023 Sony World Photography winners here. (Image credit: Lee Jongkee)

Rocket-Like Supercooled Drops
Many droplets can self-propel, often through the Leidenfrost effect and evaporation. But now researchers have observed freezing droplets that self-propel, too. The discovery came when observing the freezing of supercooled water drops inside a vacuum chamber. The researchers kept losing track of drops that seemingly disappeared. Upon closer inspection, though, they found that the drops weren’t shattering; they were flying away as they froze.
Inside a drop, freezing starts at a point, the nucleation point, and spreads from there. But the nucleation point isn’t always at the center of the drop. This asymmetry, the researchers found, is at the heart of the drop’s propulsion. When ice nucleates, the phase change releases heat that increases the drop’s evaporation rate, which can impart momentum to the drop. For an off-center nucleation, that momentum is enough to send the drop shooting off at nearly 1 meter per second. (Image credit: SpaceX; research credit: C. Stan et al.; via APS Physics)

Submarine Volcano
This pale green plume signals the activities of Kaitoku, an underwater seamount near Japan. Periodic activity picked up there in August 2022 and continued into the new year. The rising plume likely consists of superheated acidic seawater mixed with particulates, sulfur, and rock fragments. Underwater volcanoes like this one are thought to account for up to 80 percent of our planet’s volcanic activity. (Image credit: L. Dauphin; via NASA Earth Observatory)

Sedimentation After Flooding
The new year brought California a series of atmospheric rivers that poured record amounts of water onto drought-stricken lands. While the precipitation refreshed snowpacks and reservoirs, much of it washed away as soils oversaturated. Those flows carried sediment with them, creating swirls of brown and green along the coastline.
Compare the two satellite images above to see how different January 2022 looked from January 2023, post-deluge. The snow levels in January 2023 were about 248 percent of their average level for that part of the season. But the sediment levels in the ocean are also drastically increased, indicating high levels of erosion. (Image credit: J. Stevens; via NASA Earth Observatory)

“FLOW”
We live in a world of fluids. We breathe them, move through them, and have them move in us. “FLOW” is a celebration of that pervasive motion, animated from hand-drawn artwork. It features fluid dynamics from our daily lives — a candle’s flame, breaking waves, pedestrian traffic — all the way to astronomical scales far beyond typical human experience — the rotation and collision of galaxies. It’s a beautiful reminder that flows are always surrounding us, linking our lives from the small to the unbelievably large. (Video credit: MIT LineStorm Animation Consortium; submitted by Pell O.)

A Bubble’s Path
Centuries ago, Leonardo da Vinci noticed something peculiar about bubbles rising through water. Small bubbles followed a straight path, but slightly larger ones swung back and forth or corkscrewed upward. The mechanism behind this behavior has been a matter of debate ever since, but the authors of a recent study believe they’ve nailed down the answer.
The forces determining a bubble’s path are remarkably complex, which is why it’s taken so long to figure this out. Viscosity acts as a source of drag on the rising bubble, acting across a thin boundary region surrounding the bubble. That boundary isn’t constant, though; the bubble’s shape changes as the flow pushes on it, and the changing shape of the bubble pushes on the flow, in turn. Capturing those subtle interactions numerically and comparing them to careful experiments was necessary to unravel the mystery.
The team found that bubbles above a critical radius (0.926 millimeters) begin to tilt. That tilt causes a change in the bubble’s shape, which increases the flow along one side. This kicks off the wobbling motion, which carries on because of the continuing changes in the bubble’s shape and the flow around it. (Image credit: A. Grey; research credit: M. Herrada and J. Eggers; via Vice; submitted by @lediva)

Leidenfrost Explosion
When a water drop hits a surface that’s much hotter than its boiling point, part of it will vaporize immediately. Depending on the temperature, this Leidenfrost effect can be a relatively gentle process — or not. Here, the surface is so hot that the entire drop is boiling before it’s even finished spreading from impact. The vapor in contact with the surface is trying to escape, bubbling up so violently that it rips the original droplet into a spray of tiny droplets. (Video and image credit: L. Gledhill)