Flying lizards are truly gliders, but that doesn’t mean they’re unsophisticated. Newly reported observations of the species in the wild show that flying lizards don’t simply hold their forelimbs out a la Superman. Instead, they reach back with their forelimbs, pressing their arms into the underside of the thin patagium that serves as their flight surface while rotating their hands to grasp the upper side of the patagium. This forms a composite wing with a thicker leading edge and seems to be how the lizards control their glide. Close observation of their flight shows that, while holding their patagium, the lizards actively arch their backs to camber their composite wing. This can increase their maximum lift coefficient, allowing them to glide longer distances. (Image and research credit: J. Dehling, source)
Category: Research

Breaking Up Turbulence
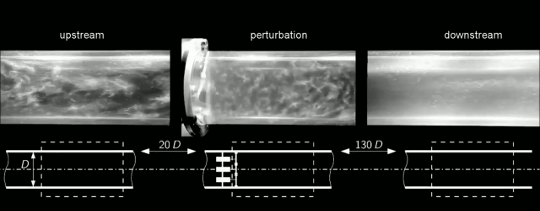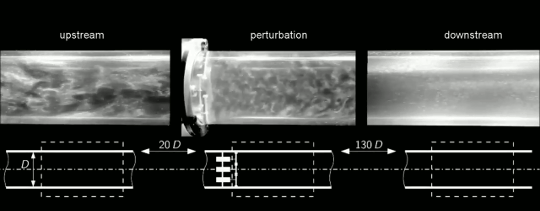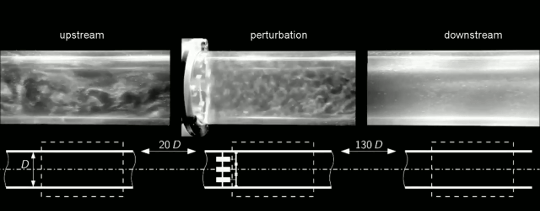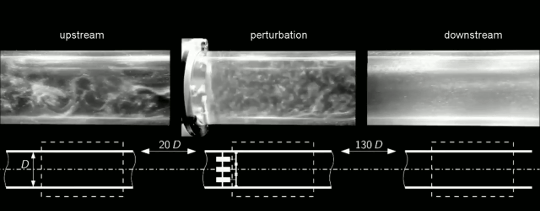
Under most circumstances, we think about flows changing from ordered and laminar to random and turbulent. But it’s actually possible for disordered flows to become laminar again. This is what we see happening in the clip above. Upstream, the flow in this pipe is turbulent (left). Then four rotors are used to perturb the flow (center). This disrupts the turbulence and causes the flow to become laminar again downstream (right). To understand how this works, we have to talk about one of the fundamental concepts in turbulence: the energy cascade.
Turbulent flows are known for their large range of length scales. Think about a volcanic plume, for example. Some of the turbulent motions in the plume may be a hundred meters across, but there are a continuous range of smaller scales as well, all the way down to a centimeter or less in size. In a turbulent flow, energy starts at the largest scales and flows further and further down until it reaches scales small enough that viscosity can extinguish them.
That should offer a hint as to what’s happening here. The rotors are perturbing the flow, yes, but they’re also breaking the larger turbulent scales down into smaller ones. The smaller the largest lengthscales of the flow are, the more quickly their energy will decay to the smallest lengthscales where viscosity can damp them out. This is what we see here. Once the turbulent energy is concentrated at the smallest scales, viscosity damps them out and the flow returns to laminar. Check out the full video below for a cool sequence where the camera moves alongside the pipe so you can watch the turbulence fading as it moves downstream. (Image and video credit: J. Kühnen et al.)
ETA: As it turns out, there’s more going on here than I’d originally thought. Simulations show that breaking up length scales is not the primary cause of relaminarization in this case. Instead, the rotors are modifying the velocity profile across the pipe in such a way that it tends to cause the turbulence to die out. The full paper is now out in Nature Physics and on arXiv.

The Fishbone
The simple collision of two liquid jets can form striking and beautiful patterns. Here the two jets strike one another diagonally near the top of the animation. One is slanted into the screen; the other slants outward. At their point of contact, the liquid spreads into a sheet and forms what’s known as a fishbone pattern. The water forms a thicker rim at the edge of the sheet, and this rim destabilizes when surface tension can no longer balance the momentum of the fluid. Fingers of liquid form along the edge, stretching outward until they break apart into droplets. Ultimately, this instability tears the liquid sheet apart. Under the right conditions, all kinds of beautiful shapes form in a system like this. (Image credit: V. Sanjay et al., source)

Swimming Microdroplets
Simple systems can sometimes have surprisingly complex behaviors. In this video, the Lutetium Project outlines a scheme for swimming microdroplets. Most of the droplets shown are just water, but they’re released into a chamber filled with a mixture of oil and surfactants. All flow through the chamber is shut off, but the droplets swim around in complicated, disordered patterns anyway. To see why, we have to zoom way in. The surfactant molecules in the oil cluster around the droplets, orienting so that their hydrophobic parts are in the oil and their hydrophilic parts point toward the water. They actually draw some of the water out of the droplets. This creates a variation in surface tension that causes Marangoni flow, making the droplets swim. Over time, the droplets shrink and slow down as the surfactants pull away more and more of their water and the variations in surface tension get smaller. (Image and video credit: The Lutetium Project; research credit: Z. Izri et al.)


Growing Fingers
Branching, tree-like structures are found throughout nature. Take a thin layer of a viscous fluid pressed between two glass plates and inject a less viscous fluid like air and you’ll get branch-like structures. These are the result of the Saffman-Taylor instability and usually result in a fairly random outcome because of the instability’s sensitivity to small variations. In a new study, researchers use multiple air injection ports to finely control the formation and growth of air fingers, allowing them to build well-ordered branching structures like the one above. By placing the air ports in an array, the same technique can be used to create fluid meshes. The authors suggest this new technique could have wide-ranging applications including the design of heat exchangers and the growth of artificial tissues. (Image and research credit: T. ul Islam and P. Gandhi, source)

Rolling Along
Leidenfrost drops – droplets deposited onto a surface much hotter than their boiling point – are known for their mobility. With the right surface, they can be propelled, trapped, and even guided through a maze, typically by directing the vapor layer that cushions them. But new work shows that these drops have internal dynamics that also contribute to their propulsion.
By adding tracer particles to each droplet, researchers can visualize flows inside the droplet. Large drops tend to have a flatter shape and contain two or more rotating vortices. Such drops won’t propel themselves without another force in play. But smaller droplets are more spherical and contain only a single rotating flow. Once these drops detach, they roll away! Despite the similarity to wheels, these liquid drops aren’t moving the same way. Remember that the drop is not actually in contact with the surface. To see what sets the drop’s direction, researchers examined the shape of the bottom of the drop. They found that it sits at a slant on its vapor cushion. That pushes evaporating gases out one side, propelling the drop the other way. (Image and video credit: A. Bouillant et al., source)

Twisting to Pack
One conventional method for packing granular materials is to tap them repeatedly, but a new study suggests that twisting is a faster method. Researchers poured thousands of dice into a cylinder, then twisted the container back and forth. When the acceleration caused by the change in direction exceeded a threshold value, the dice worked their way into ordered layers of concentric rings over ~10,000 cycles. Reaching this maximum packing density through tapping requires a specially designed method where the tapping characteristics change over time. With twisting, shear forces transmitted from the walls of the container tend to align the flat surfaces of the dice, providing an efficient method of ordering if the acceleration is large enough. The researchers hope methods like this may be useful where tapping works poorly, such as in microgravity. (Image and research credit: K. Asencio et al.; via APS Physics; submitted by Kam-Yung Soh)

Layered Latte Physics
Latte lovers may be familiar with the layered latte, a beverage with distinctive horizontal layers mixing espresso and milk, but you may not have taken the time to wonder how these layers form. Like many layering phenomena in our oceans, the layered latte is the result of double-diffusive convection. This means that there are two variables that both affect density in the fluid mixture and that they act at different rates.
In the latte, those factors are 1) the different densities of the milk and espresso and 2) density changes caused as the latte cools to room temperature. A layered latte forms when the lighter espresso is poured into denser milk. If it’s poured quickly enough, the momentum of the pour forces some of the espresso down into the milk, despite the buoyant force that tries to keep the espresso on top. So that initial pour sets up a density gradient that runs from pure espresso at the top to pure milk on the bottom, with varying mixtures of the two in between.
The distinct layers won’t form until the latte begins cooling off. Along the walls of the container, heat is lost more quickly, causing fluid to cool and start sinking. But a specific bit of fluid can only sink until the fluid surrounding it is the same density. That can carry a cooler bit of latte to the bottom of a layer, but not into the denser layer below. At this point, our bit of latte moves inward, starts to warm up, and circulates up through the center of its layer. As when it sank, the fluid can only move up until it encounters a layer with equal or lesser density, at which point it must move horizontally instead. This thermal convection, combined with the density gradient formed by the initial pour, sets up the distinctive layers of the latte. The layers are quite stable – neither gentle stirring nor taking a sip will disrupt them for long – provided the drink remains warmer than the surrounding air. (Image credits: kopeattugu/Instagram, N. Xue et al.; research credit: N. Xue et al.; via NYTimes; submitted by Kam-Yung Soh)

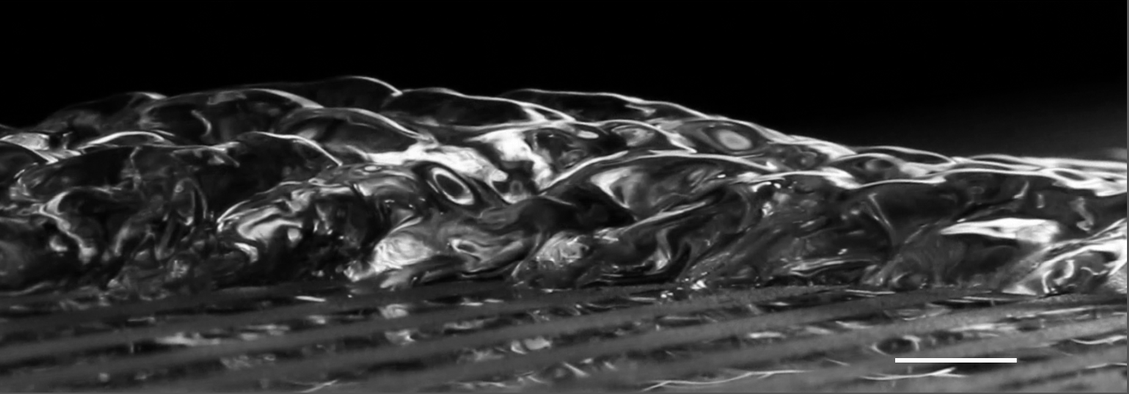
Corrugating Water
The characteristics of a surface can have a major impact on the form a flow takes. The photo above shows a corrugated, almost pinecone-like water surface. It’s the result of a sheet of water flowing over a surface with alternating bands of hydrophobic (water-repelling) and hydrophilic (water-loving) properties. The water sheet narrows over hydrophobic sections and expands over hydrophilic ones. Gravity, inertia, and surface tension compete to create the overall braided appearance. You can see a top-down view of the flow in the original poster. (Image credit: M. Grivel et al., source)