How does water drip, drip, dripping onto stones erode a crater? Water is so much more deformable that it seems impossible for it to wear harder materials away, even over thousands of impacts. To investigate this, a team of researchers developed a new measurement technique: high-speed stress microscopy. In the process, they found that water owes its incredible erosive power to three factors: 1) The drop’s impact creates surface shock waves along the material, which helps increase erosive power; 2) After the shock wave passes, a decompression wave in the material helps loosen surface matter; and 3) The spreading drop sends a non-uniform wave of stress across the material that simultaneously presses and scrubs at the surface. Together, these factors enable simple, repetitive droplet impacts to wear away at hard surfaces. (Image credit: cottonbro; research credit: T. Sun et al.; via Cosmos; submitted by Kam-Yung Soh)
Category: Research

Coalescence Symmetry
When droplets coalesce, they perform a wiggly dance, gyrating as the capillary waves on their surface interfere. When the droplets have matching surface tensions, like the two water droplets in the animation on the lower left, the coalescence dance is symmetric. But for differing droplets, like the water and ethanol droplets merging on the lower right, coalescence is decidedly asymmetric.

Two water droplets merge symmetrically. 
A water droplet and an ethanol droplet merge asymmetrically. The asymmetry arises from the droplets’ different surface tensions. The size and speed of the capillary waves that form on a droplet depend on surface tension, so droplets of different liquids have inherently different capillary waves. During merger, the interference of these capillary waves causes the asymmetry we see. (Image credit: top – enfantnocta, coalescence – M. Hack et al.; research credit: M. Hack et al.)

Featherwings in Flight
The featherwing beetle is tiny, less than half a millimeter in length. At that scale, flying is a challenge, with air’s viscosity dominating the forces the insect must overcome. The featherwing beetle, as its name suggests, has feather-like wings rather than the membranes larger beetles use. But a new study shows that these odd wings work surprisingly well.
The beetle’s bristled wings flap with an exaggerated figure-8 motion, with the wings clapping together in front of and behind the insect. The beetle expends less energy moving its feathery wings than it would if they were solid, and it moves its wing covers at the same time to counter each stroke and keep its body steady. (Image and research credit: S. Farisenkov et al.; video credit: Nature; submitted by Kam-Yung Soh)

Raindrops on the Windshield
When I was a child, I was fascinated by the raindrops that shimmied along the windshield of our car. Some would slide up the glass. Some would run down. And some just seemed to wiggle in place, until the car’s speed changed. As common as this sight is, the physics of these droplets is quite complicated and not completely understood.
Each droplet has a host of forces on it: gravity flattening it or pulling it down an incline; a drag force from the wind flowing over it; and friction between the drop and the surface trying to pin it in place. Recently, scientists have developed a new mathematical model that captures some of the behaviors behind these drops. The work describes the wind speed necessary to move a drop of a given size sitting on a flat surface. The authors also explored how that critical wind speed changes when a drop sits on a tilted surface aligned or against the wind. (Image credit: P. Gupta; research credit: A. Hooshanginejad and S. Lee; via Science News; submitted by Kam-Yung Soh)

Using Turbulence in Flight
When small, heavy particles are in a turbulent flow, they settle faster than in a quiescent one. Their interactions with turbulent eddies sweep them along, extracting energy that lengthens their overall path but reduces the time necessary for them to fall. Using the same principles, researchers are finding ways for rotorcraft and other vehicles to extract energy from turbulence for more efficient flight.
The technique forces a vehicle to behave like a heavy particle by sensing turbulent gusts from its own accelerations and adding forcing to those accelerations when they are in the desired direction of flight. In essence, the vehicle uses the turbulence of its surroundings to find helpful tailwinds. (Image credit: A. Soggetti; research and submission credit: S. Bollt and G. Bewley)

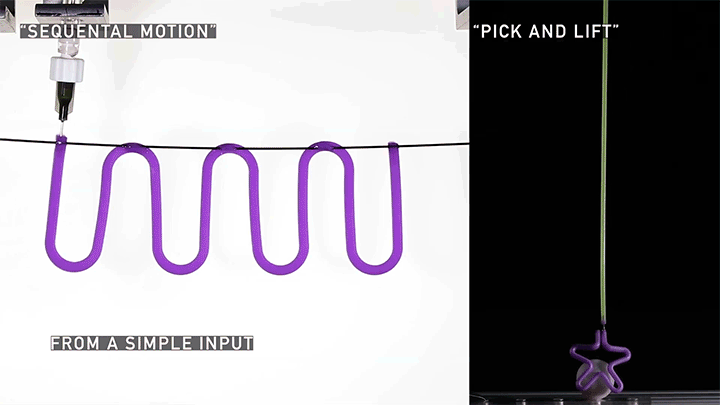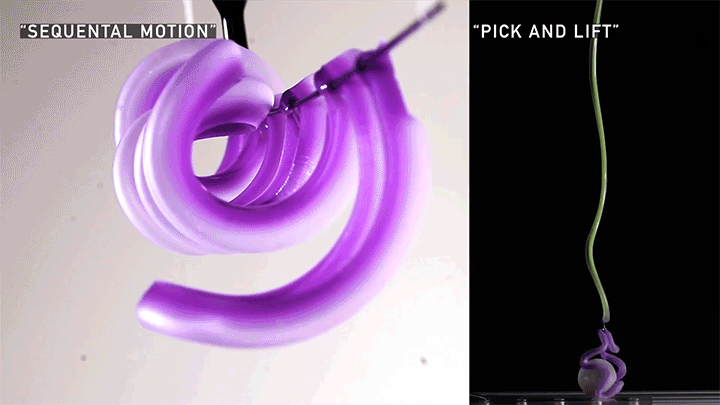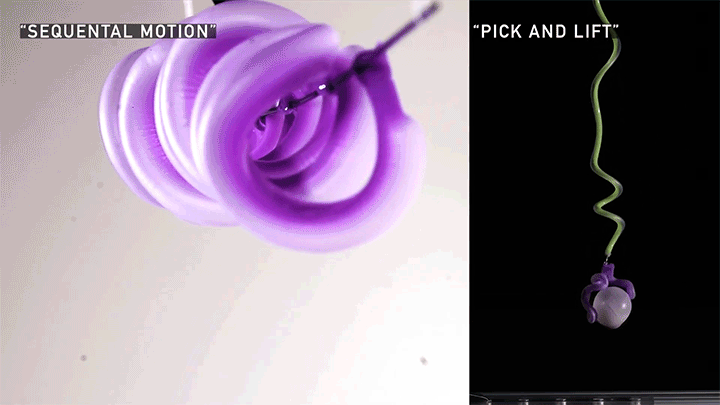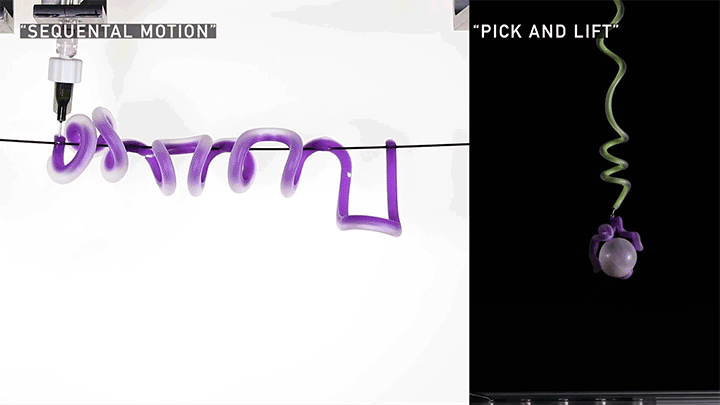
Squishy Actuators
Hard materials don’t always work well in robotics. Here, researchers build soft actuators that can bend, curl, and tighten in order to manipulate objects. They begin by injecting liquid elastomer into a tube (Image 1), followed by a bubble of air. Buoyancy makes the air bubble rise within the tube, creating an asymmetric cross-section where the solidified elastomer has a thin shell along one side and a thicker wall along the other (Image 2). When high-pressure air is pumped into the soft tubes, their asymmetric cross-section makes them bend and twist (Image 3). The team found that they can tune the elastomer tubes to form complex shapes good for gripping and flexing — perfect for a soft robot! (Video and image credit: T. Jones et al.; research credit: T. Jones et al.)

Box Closing Physics
My fellow board game aficionados (and anyone else who regularly opens and closes lidded boxes) have probably noticed the way a lid drops slowly onto its box once aligned. The weight of the lid pressurizes air inside the box, driving a flow through the narrow gap between the walls of the box and the lid. Researchers found that the time it takes for a box to slide closed is closely related to the size and shape of the gap between the walls. Despite gaps of less than 1 millimeter, air moving out of the box typically flows at about 1 meter per second!
With their mathematical model of the flow from a closing box, the group was also able to determine the optimal shape for a fast-closing box, something that may be of interest to manufacturers as well as fans of board games. (Image credit: N. Sharp; research credit: J. de Ruiter et al.; via APS Physics)

Making Yeast-Free Pizza
Yeast is a key ingredient in many pizza doughs; as the yeast ferment sugars in the dough, they produce carbon dioxide which bubbles into the dough, creating the light and airy texture necessary for a good crust. It’s a slow process, though, often requiring several hours for the dough to rise. Recently, researchers studied an alternative pizza-making method that generates bubbles in the dough via pressurization — with no yeast required.
The new technique is similar to the process used to carbonate sodas. The team mixed flour, water, and salt and placed the dough in an autoclave, which allowed them to control both temperature and pressure during baking. They dissolved gas into the dough at high pressure and then carefully released the pressure during baking, allowing the bubbles to grow. They used rheological measurements to compare the characteristics of yeasted and yeast-free doughs at various stages in the leavening and baking processes.
Now that they have the methodology down, they’ve purchased a food-grade autoclave and are looking forward to taste testing their yeast-free creations — none more so than their team member who has a yeast allergy! Since the pressures required for their method are quite mild, they hope it’s a technique that restaurants will take on. (Image credit: B. Huff; research credit: P. Avallone et al.)

Dispelling Ice
In winter weather, delays pile up at airports when planes need de-icing. Our current process involves spraying thousands of gallons of chemicals on planes, but these chemicals are easily removed by shear stress and dissolution, meaning that by the time a plane takes off, there is little to no de-icing agent remaining on the plane. Instead, those chemicals become run-off.
Researchers looking to change that have developed a family of anti-icing coatings — including creams, sprays, and gels — that are easy to use and apply, non-toxic, and much longer lasting than conventional methods. Ice slides easily off their gel coatings, which remain optically transparent even under freezing conditions — and ice can take 25 times longer to form on the gels compared to current anti-icing tech.
The team envisions using their coatings on much more than airplanes. Imagine traffic lights that can’t be obscured by ice or snow, a windshield on your car that never freezes over, or even an anti-icing spray that could protect crops from a sudden freeze! (Image, video, research, and submission credit: R. Chatterjee et al.; see also)