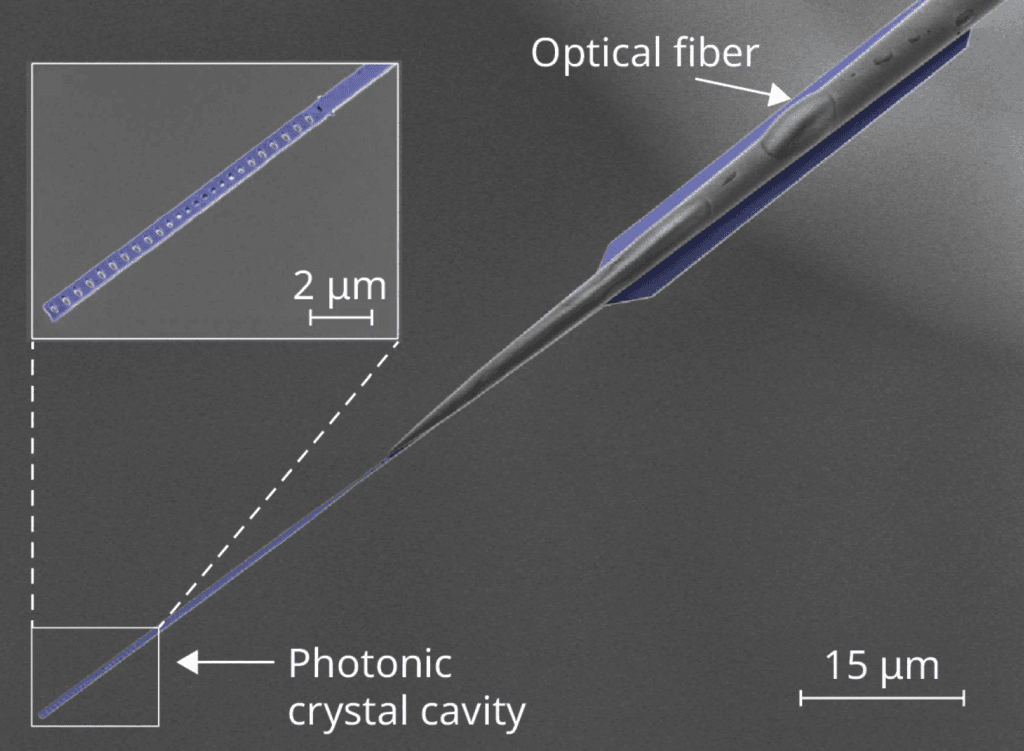
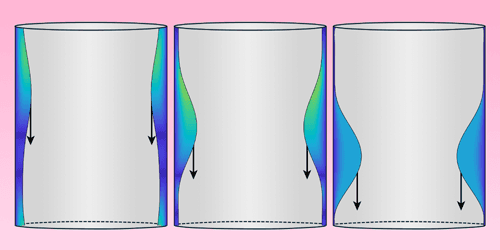
It’s tough to simulate nonlinear wave dynamics, so scientists often test theories in wave flumes, where they can create more controlled waves than what we see in the wild. But conventional wave flumes are big–meters-long, complicated equipment–and can only test a small range of conditions. To reach more extreme nonlinear dynamics, researchers have turned to a chip-based approach. These 100-micron-long wave flumes carry a film of superfluid helium less than 7 nanometers thick. But despite that tiny size, the system can reach levels of nonlinearity five orders of magnitude greater than their full-sized counterparts. (Image and research credit: M. Reeves et al.; via Physics Today)