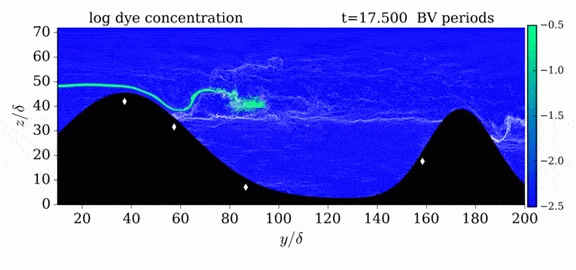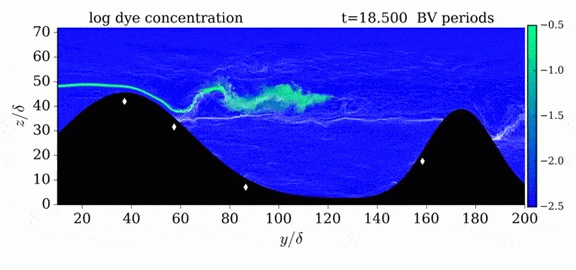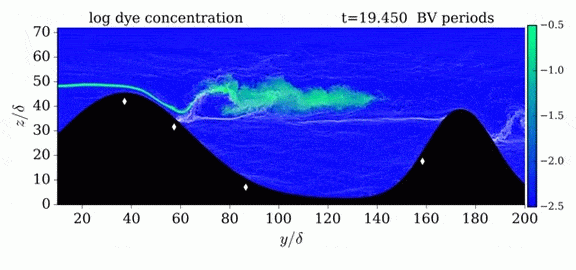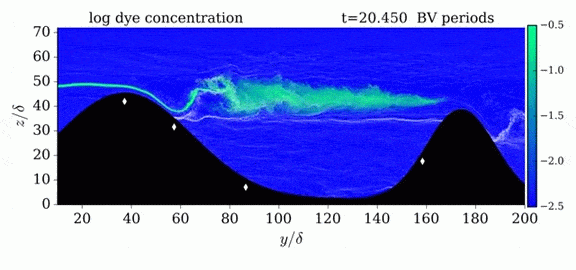
This year’s Tour de France opened with an individual time trial stage in which riders competed solo against the clock. But, according to numerical simulations, some riders may get an unfair aerodynamic advantage in the race if they have a following car. The top image shows the pressure fields around a rider with a car following 5 meters behind versus 10 meters behind. The size of the car means that it displaces air well in advance of its arrival. By following a rider closely, that car’s high pressure region can help fill in a cyclist’s wake, thereby reducing the drag the rider experiences. For a short time trial like the 13.8 km race that kicked off this year’s tour, a rider whose car follows at 5 meter could save 6 seconds over one whose car followed at the regulation 10 meter distance. (As it happens, the stage was decided by a 5 second margin.) Since not all riders get a team follow car, it’s especially important to ensure that those who do aren’t receiving an additional advantage. For more about cycling aerodynamics, check out our previous cycling posts and Tour de France series. (Image credit: TU Eindhoven, EPA/J. Jumelet; via phys.org; submitted by @NathanMechEng)