When a drop of water touches a very hot pan, it will skitter across the surface on a thin layer of water vapor due to the Leidenfrost effect. But what happens when another chemical is added to the droplet? Researchers find that adding a surfactant to the water droplets creates some spectacular results. As the water evaporates, the concentration of the surfactant in the droplet increases causing the surfactant to form a shell around the droplet. The pressure inside the droplet increases until the shell breaks in a miniature explosion much like the popping of popcorn. (Video credit: F. Moreau et al.)
Year: 2012
APS Division of Fluid Dynamics 2012
Just a heads up that I’ll be at the annual American Physical Society Division of Fluid Dynamics meeting in San Diego starting this weekend. There are regular posts lined up, but additional supplemental posts may also go up in addition to plenty of Twitter updates.

Catastrophic Cracking from Cavitation
At your next party, you can break the bottom of a glass bottle with the palm of your hand and the power of fluid dynamics. As shown in the video above, striking the mouth of the bottle accelerates fluid at the bottom, lowering the local pressure below the vapor pressure and causing the formation of cavitation bubbles. When these bubbles collapse, they form very high temperatures and pressures for an instant, and it is this which can break the glass. (Video credit: J. Daily et al., BYU Splash Lab)

The Beauty of the Great Red Spot

Jupiter is home to one of the most famous storms in the solar system, the Great Red Spot, which Earth observations place at a minimum of 180 (Earth) years in duration. Some evidence suggests that it may have been observed by humans as early as 1665. The magnitude of such a storm is almost unimaginable. At its narrowest point, the storm is still as wide as our entire planet and observations from the Voyager crafts indicate that the storm has 250 mph winds. The scale of mixing and turbulence around the storm, seen in photographs, is stunning and beautiful. (Photo credits: NASA/Voyager 1 and Michael Benson; submitted by oneheadtoanother)

Jets from Hollows
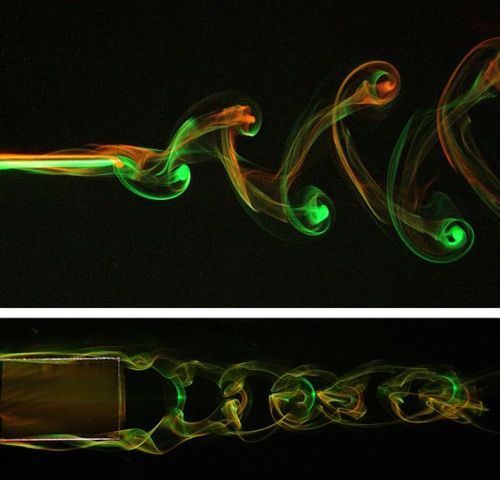
Bubbles rising through a viscous fluid deform and interact. As they collapse into one another, the lower bubble induces a gravity-driven jet that projects upward into the higher bubble. The more elongated the bubble, the faster the jet. The same behavior is seen in the rebound of a cavity at the free surface of a liquid. The authors suggest a universal scaling law for this behavior. (Video credit: T. Seon et al.)

Viscoelastic Jets
Unlike Newtonian fluids, such as air and water, viscoelastic fluids exhibit non-uniform reactions to deformation. In this video, researchers explore the effects of this behavior when a liquid jet falls into another fluid. When fluids move past one another at different speeds in this manner, there is a shearing force which often leads to the wave-like Kelvin-Helmholtz instability between the fluids. Here we see for a variety of wavelengths how the breakdown of a Newtonian and viscoelastic jet differ. The Newtonian jets form clean lines and complicated tulip-like shapes, but the viscoelasticity of the non-Newtonian jets inhibits the growth of these instabilities, surrounding the central jet with wisps of escaping fluid. For more, see Keshavarz and McKinley. (Video credit: B. Keshavarz and G. McKinley)

Dancing Droplet Clusters
When a fluid surface is vibrated, it’s possible to bounce a droplet indefinitely on the surface without the droplet coalescing into the pool. This is because each bounce of the droplet replenishes a thin layer of air that separates the droplet and the pool. If many droplets are added to the surface, as in the video above, a clustering behavior is observed, with many droplets gathering together. There is a limit, however, to the size of the cluster based on the amplitude of vibration. If vibrational amplitudes are pushed to the point of creating Faraday waves–standing waves on the surface of the pool–then large clusters of droplets can be suspended and sustained. (Video credit: P. Cabrera-Garcia and R. Zenit; via io9; submitted by oneheadtoanother)

Fluidic Public Art by Charles Sowers
Artist Charles Sowers creates exhibits and public art focused on illuminating natural phenomenon that might otherwise go unnoticed, and much of his work features fluid dynamics directly or indirectly. “Windswept” and “Wave Wall” are both outdoor exhibits that show undulations and vortices corresponding to local wind flow. Other pieces explore ferrofluids through magnetic mazes or feature foggy turbulence. My own favorite, “Drip Chamber”, oozes with viscous fluids whose dripping forms patterns reminiscent of convection cells. Be sure to check out his website for videos of the exhibits in action. (Photo credits: Charles Sowers; submitted by rreis)

Reader Question: How Useful is Flow Viz?
Reader Andrew asks:
I’ve noticed you’ve posted a bunch of flow visualization/wind tunnel content. I’m just curious where how useful information is obtained from these. Is it just observation? Or are there instruments that are usually used in conjunction with these techniques to provide data?
Great question, Andrew! The answer can vary based on the technique and application. In some cases, flow visualization is used for purely qualitative observation, but in others it can provide more quantifiable data. For example, the water tunnel flow visualization of Google’s heliostat array gave very qualitative data about flow around a given configuration but allowed quick evaluation of many configurations. Flow visualization can also help identify key features for additional study like vortices in a wake. This identification of structure can be so useful that even in computational fluid dynamics, where researchers have all possible information about pressure, temperature, and velocity in a flow field, flow visualization is regularly used to identify underlying structures.
Some flow visualization methods can also give very specific information. Oil-flow visualization gives a snapshot of shear stress at the surface of an object, letting an engineer identify at a glance areas of laminar and turbulent flow as well as regions with vortices and streaks. Naphthalene flow visualization and infrared thermography are both great for identifying the location of laminar-turbulent transition and can do so across the span of an object, which is much easier than trying to traverse a probe across the entire object. And some forms of flow visualization allow for extraction of velocity field information, as in particle image velocimetry. In this technique, tiny particles seed the flow and carefully timed image pairs are taken and correlated to determine the flow field velocity based on the changes in particle positions between images.
Like every measurement, flow visualization methods have their strengths and limitations. But for many applications, flow visualization provides much more than just pretty pictures and thus remains an important tool in any fluid dynamicist’s arsenal!

Detonation in a Bubble
Accidental releases of combustible gases in unconfined spaces can be difficult to recreate in a laboratory environment. Here researchers simulate the conditions using detonation inside a soap film bubble. Combustible gases are pumped inside the soap film and then a spark creates ignition. The resulting flame propagation is visualized using high-speed schlieren photography, making the density gradients in the flame visible. When the mixture of hydrogen fuel to air is balanced, the flame is spherically symmetric with a high flame speed. In contrast, weaker mixtures of fuel/air produce slow flame speeds and mushroom-like flames that leave behind unreacted fuel. This is due to buoyant effects; the time scale associated with buoyancy is smaller than that of the flame speed and chemical reactions when the fuel/air mixture is lean. (Video credit: L. Leblanc et al.)