Photographer Mikko Lagerstedt specializes in Nordic landscapes, like the windswept snow seen here. I love the way he’s captured the snow that gets picked up and blown by the wind. Notice the hazy layer of snow hovering over the foreground. This snow is saltating, just as sand does in the desert. When flakes get picked up by the wind, they follow a ballistic trajectory, much like a cannonball in a high-school physics class. As the snow crashes back down, its impact knocks up more flakes, and the process continues. Repeat enough times, and you’ll see this hazy layer of blowing snow blanketing a snowscape. (Image credit: M. Lagerstedt; via Colossal)
Tag: granular material

Seashore Hunting
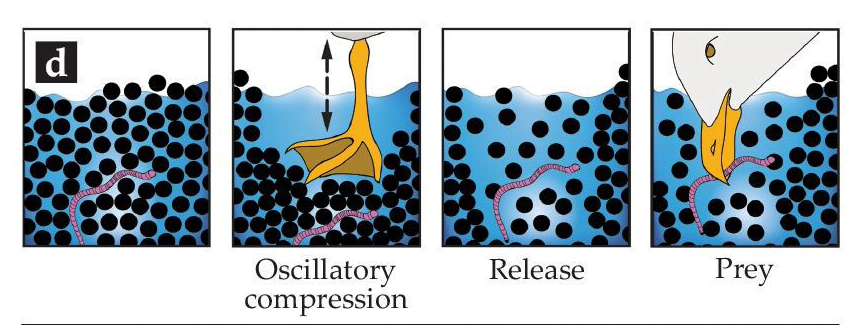
Watch sea gulls, plovers, and other birds hunt in the tidal zone, and you may notice them stepping over and over in the same spot. This is part of bird’s hunting strategy. Each footfall compresses the wet sand and drives water out. Mechanically, this is the same thing that happens when a human walks on wet sand; you’ll see the sand go from a glossy appearance to a matte one as the local water level falls. Once the weight is removed, the water will seep back and the sand appears glossy again.

Illustration of a gull’s hunting process. Compressing the sand by stepping on it drives water out of the area. Once the bird’s foot is removed, water floods back, diluting the sand, and making it easier for the bird to reach its prey without digging. For the birds, the flood of returning water loosens and dilutes the sand. That makes prey easier to expose and reach without the additional effort of digging. (Image credits: bird – C. Davis, illustration – P. Fischer; via Physics Today)

In a Box, Shaken
Tidal areas experience lots of oscillating, back-and-forth flow that builds up patterns in the sand below. In this experiment, researchers investigate a similar situation by filling a box with water and spherical particles, then shaking the box from side-to-side. Inside the box, the particles line up in chains that are perpendicular to the direction of oscillation (think sand ripples parallel to a shoreline). In this simplified system, the team can then look at what forces align the particles, how defects in the pattern shift, and what happens when the oscillation gets bigger. (Image and video credit: T. van Overveld et al.)

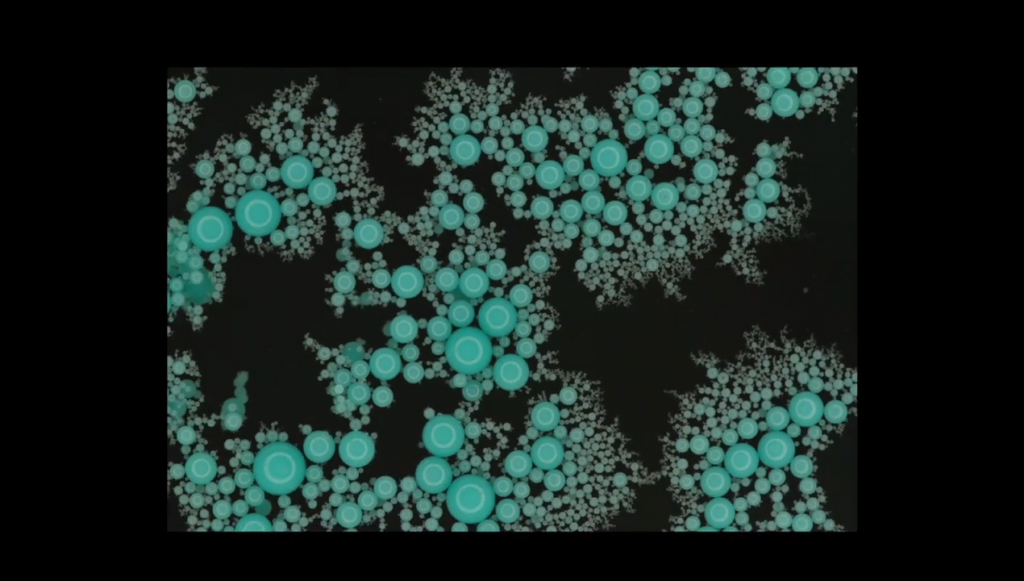
A Fractal Raft From a Spinning Top
File this one under Cool Things I Would Have Never Thought Of. In this video, researchers play around with the flow around a spinning top and end up creating a fractal, granular raft. By immersing a top in dyed fluid, they show the toroidal vortices that form around the spinning toy. Then, instead of dye, they add a stretchy elastomer compound that cures over time. The elastomer stretches into thin ligaments in the swirling flow around the top. Eventually, it breaks apart into spherical drops of all different sizes.
Once the top is removed, the elastomer drops slowly float to the surface. Surface tension and the Cheerios effect draw the drops together, and because of their many sizes, the rafts that form are fractal. (Image and video credit: B. Keshavarz and M. Geri)

Mixing the Perfect Batter
In baking, there’s a point when wet and dry ingredients get combined to form the batter (or dough) that eventually becomes a tasty treat. Experienced bakers know that the ratio of wet-to-dry must be just right for the final product. Too dry and the mixture won’t come together; too wet and the final product is a soggy mess.
Mixing liquids and powders is ubiquitous outside the kitchen, too. Ceramics, concrete, laundry detergent, chocolate — all involve this critical step. To understand how these mixtures transition from fluid to clustered granules to granulations (think wet sand), researchers carefully studied a mixture of glass spheres and glycerol. When there were relatively few particles in the mixture (in technical terms, a smaller “particle volume fraction”), the mixture was fully fluid (top image, orange background). When the ratio of particles-to-liquid was high, the mixture was granular (blue background). And in-between these ratios, whether the mixture formed clumps, or granules, depended on how it was mixed (green background). Vigorous mixing (top row) formed large granules, which consisted of a wet, jammed interior and an outer layer of dry particles (lower image).
Their observations allowed the researchers to predict what ratio of liquid and powder is needed, and how much mixing is necessary, to create a desired outcome. (Image and research credit: D. Hodgson et al.; via Physics Today)

A cross-section of a granule, showing the wet, jammed interior (left) surrounded by a region of dry particles (center, enclosed between red dashes). 
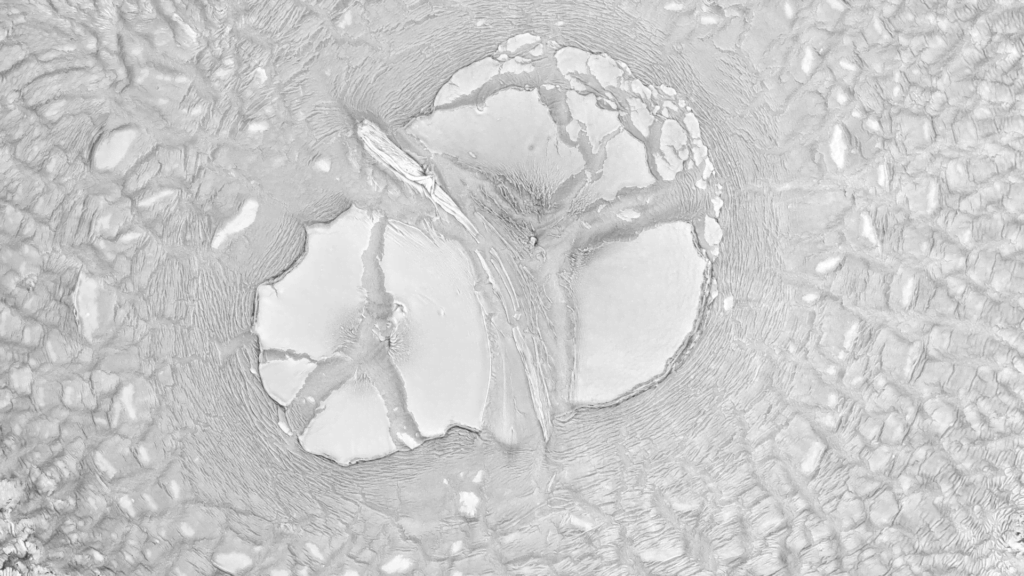
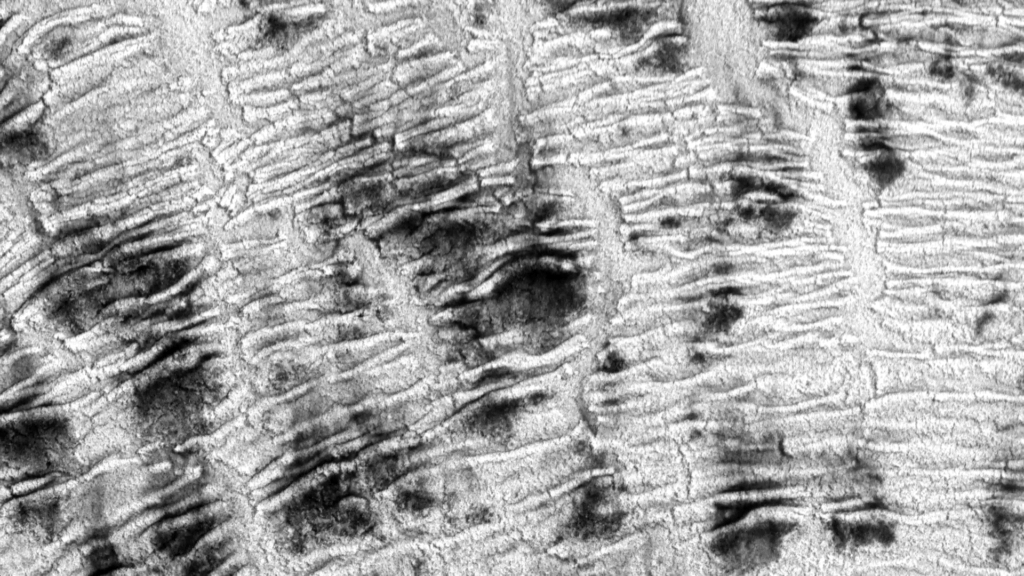
“Haut”
In Susi Sie’s “Haut” the camera seems to fly over ever-shifting landscapes. In reality, these are macro images, created (I think) by dyes and patterns atop a water bath. But they look like vistas we could find on Earth or Mars — giant dune fields, calving glaciers, and river-divided canyons. For something similar in color, check out Roman De Giuli’s “Geodaehan.” (Video credit: S. Sie)

Eroding Grains
When a spacecraft comes in for a landing (or a tag similar to what OSIRIS-REx did), there’s a turbulent jet that points straight into a bed of particles. How those particles react — how they erode and the crater that forms — depends on many factors, including the cohesion between particles. In these experiments, researchers investigated such a jet (in air) and its impact on particles with differing amounts of cohesion.
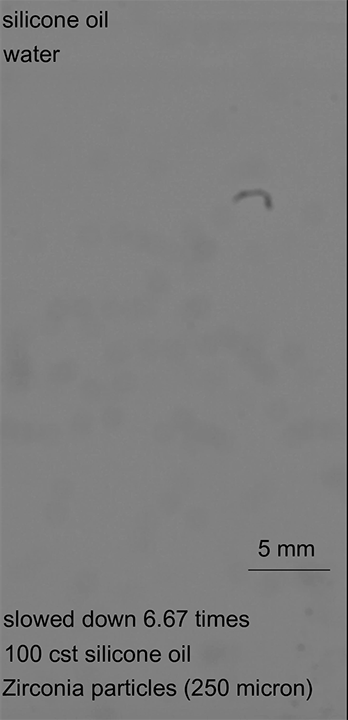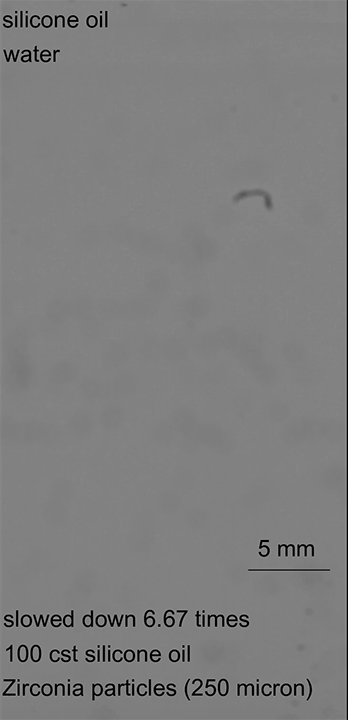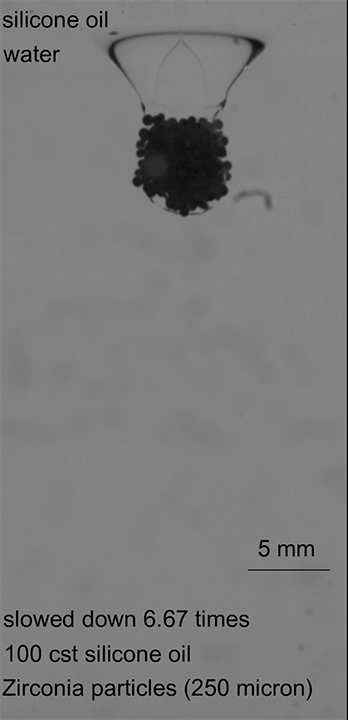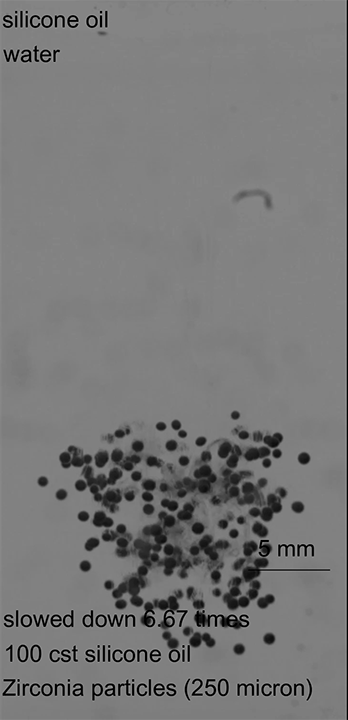
When there is little cohesion between particles, erosion takes place a single particle at a time (Image 1). Once there’s some cohesion, the jet’s velocity has to be higher to trigger erosion (Image 2). Once erosion does begin, it includes both singular and clumped particles. In highly cohesive beds, velocities must be even higher to create erosion, which takes place with large clusters of particles flying off together (Image 3). (Image and research credit: R. Sharma et al.)

A Levitated Boil
When acoustically levitated, objects tend to clump together and move like a single, large solid. But researchers found more fluid-like states for their levitated particles when the particles were smaller. At low acoustic power, the particles behave like a liquid and shift primarily within a plane. But as the acoustic power increases, the granular liquid begins to “boil” and transition into a gaseous state, with particles moving in all directions. It’s amazing how often these metaphors (e.g., treating a group of particles as a “liquid”) hold true when observing different physical systems! (Image and video credit: B. Wu et al.)

Encapsulating Drops
Sometimes a droplet needs a little protection while it’s traveling to its destination. When that’s the case, we often try to encapsulate it in a layer of material that won’t be affected by whatever environment the drop is traveling through. In this study, researchers aimed to give their drops not one but two layers of protection — in as simple a way as possible.
The team began with three layers of liquid. The lowest layer was water, the middle layer was an oil, and the top layer was a mixture of water and isopropyl alcohol. Next, they added glass particles that were denser than the alcohol, but less dense than the oil. This caused the particles to form a clump — a granular raft — along the interface between the alcohol and the oil (not shown). When the layer of particles became heavy enough, it began to sink into the oil, carrying some of the alcohol with them. This conglomeration formed the initial droplet of alcohol mixture encased in an armor of glass beads.
As this armored droplet sank, it approached the second interface: the oil-water interface. At this juncture, the team observed three different outcomes. When the glass particles were small or light, the armored drop would come to a rest at the oil-water interface. As the drop deformed, water would pierce the armor, causing the whole drop to rupture (Image 1).
In the second case, heavier particles caused the armored drop to sink through the oil-water interface, but a low oil viscosity meant that the oil film drained from the bottom of the drop before the drop was fully encapsulated. Once again, this let the water through and ruptured the droplet (Image 2).
In the final case, armored drops with just the right bead density and oil viscosity would sink through the oil-water interface until the oil pinched off behind the drop. This pinch-off allowed the oil to redistribute around the drop, encapsulating it in layers of both oil and particles, thereby protecting it as it continued its journey (Image 3). (Image credits: top – Girl with red hat, experiment – A. Hooshanginejad et al.; research credit: A. Hooshanginejad et al.)

Slow to Relax
Oobleck is a decidedly weird substance. Made from a dense suspension of cornstarch in water, oobleck is known for its mix of liquid-like and solid-like properties, depending on the force that’s applied. In a recent study, researchers took a look at what happens when you really push oobleck to the extreme. When the force applied to oobleck is small or slowly added, the water between cornstarch particles helps keep the particles apart and free of contact. It’s when the force is large that those particles start jamming up against each other and having friction between them, and then the oobleck suddenly acts like a solid. But what happens once that force is removed?
When the force is gone, we expect the particles to repel and for water to squeeze back into the spaces between them, breaking up the friction and allowing the oobleck to relax back to a liquid-like form. But the team found that sometimes the oobleck doesn’t relax as easily as expected; instead, it seems to retain some memory of its solid-like state, due to persisting friction between particles. (Image credit: T. Cox; research credit: J. Cho et al.)