A drop of fluorescent dye falling into quiescent water forms fantastical structures that are a mixture of vorticity, turbulence, and molecular diffusion. The horseshoe-like shape near the front of the drop is a typical shape for two fluids strained by moving past one another. The main section of the drop billows outward like a parachute, but the turbulence of its wake stretches the dye into fine threads that quickly disperse in the water. (Photo credit: D. Quinn et al.)
Tag: droplets

“Levitating Water”
Al Seckel, a cognitive neuroscientist and expert on illusions, created this “Levitating Water” installation, in which multiple streams of water appear as a series of levitating droplets thanks to a strobing light. The well-timed strobe lighting tricks the brain into seeing many different falling droplets as the same, nearly stationary droplet. The effect is similar to the one created by vibrating a stream of falling water. (Video credit: wunhanglo)

Droplet Bounce
This high-speed video shows the remarkable resilience of a water droplet upon impact against as a solid surface. The droplet deforms into a pancake-shape, with its center depressing almost flat before rebounding upward. The rest of the drop follows, splitting into several droplets as capillary waves dance across its surface. When one satellite drop almost escapes, the main droplet just barely comes in contact with it, the coalescence enough to tip surface tension into pulling them together instead of breaking them apart. (Video credit: K. Suh/ChemistryWorldUK)

Bouncing on a Pool
There’s something wonderfully serene about watching water droplets skate their way across the surface of a pool. Here the pool of water is being vibrated at a frequency just below the Faraday instability – meaning that no standing waves form on the surface. Instead, the bounce is just enough to create a thin layer of air between the droplet and the pool to prevent coalescence. With each bounce, gravity’s effect on the water tries to drain the air away, but each rebound lets more air rush in to hold the droplet up. Eventually, gravity wins and the droplets coalesce into the pool. In high-speed that process is mesmerizing, too. (Video credit: K. Welch)

Electrowetting
The electrowetting effect can change the shape of a liquid droplet on a surface by applying a voltage across the surface and droplet. Surface tension is a kind of measure of the energy required to maintain a certain drop shape, and that energy can be both chemical and electrical. In the video above, the droplet maintains a small contact area naturally (with no voltage). It expands and flattens under an electrical charge. Varying the voltage will change the degree to which the droplet flattens, but only to a point. Electrowetting is used to control variable lenses and some types of electronic displays. The technology may be used to replace current generation LCDs. (Video credit: V. Arya/Duke University)

Encapsulating Droplets
In applications like drug delivery, it’s often desirable to encapsulate one or more liquid droplets in an additional immiscible fluid. These drops-within-drops, called double emulsions, are typically a multi-step process, created from the innermost drop outward. In this new microfluidic technique, though, researchers are able to create multi-component emulsions in a single step. A double-bored capillary tube creates the two inner droplets (both water, dyed different colors) while oil flows down the outside of the injection tube to encapsulate the droplets. The multi-component double emulsions then flow as one to the right in the outer carrier fluid. The spacing of the capillary tubes is critical to prevent the inner droplets from coalescing with one another. (Video credit: L. L. A. Adams et al.)

Breaking Up a Ferrofluid
Ferrofluids are known for their fascinating behaviors when subjected to magnetic fields, especially for the distinctive peaks they can form. In this video, we see a very thin ferrofluid drop on a pre-wetted surface just as a uniform perpendicular magnetic field is applied. Immediately the droplet breaks up into tiny isolated peaks that migrate out to the circumference. The interface breaks down from center, where the drop height is largest, and moves outward. Simultaneously, the diffusion of ferrofluid from the circumferential droplets into the surrounding fluid lowers the magnetization of those droplets, making it more difficult for them to repel their neighbors. As a result, they drift outward more slowly and get caught by the faster-moving droplets from within. (Video credit: C. Chen)

Evaporation and Surface Effects
Surface properties can have surprising effects on fluid behavior. This image shows the evaporation of several droplets over time. All of the initial droplets are of the same volume, but they are placed on a surface which is a) superhydrophobic, b) hydrophobic, or c) hydrophilic. The more hydrophobic the surface, the larger the initial contact angle between the droplet and surface and the smaller the wetted area of the surface. Yet despite this seemingly large surface area exposure to air, the droplet on the superhydrophobic surface is the slowest to evaporate. (Photo credit: C. Choi)

Bouncing to Mix Oil and Water
Mixing immiscible liquids–like oil and water–is tough. The best one can usually do is create an emulsion, in which droplets of one fluid are suspended in another. The series of images above shows a double emulsion consisting of oil and water that’s been formed by bouncing the compound droplet on a vibrating bath. The vibration of the liquid surface keeps the droplet from coalescing with the bath and the deformation provides mixing. The top row shows the initial impact while the bottom row of images shows the droplet after many bounces. As time goes on, the layer of oil around the compound drop becomes a cluster of tiny droplets contained within the water portion of the drop. (Photo credit: D. Terwagne et al.)

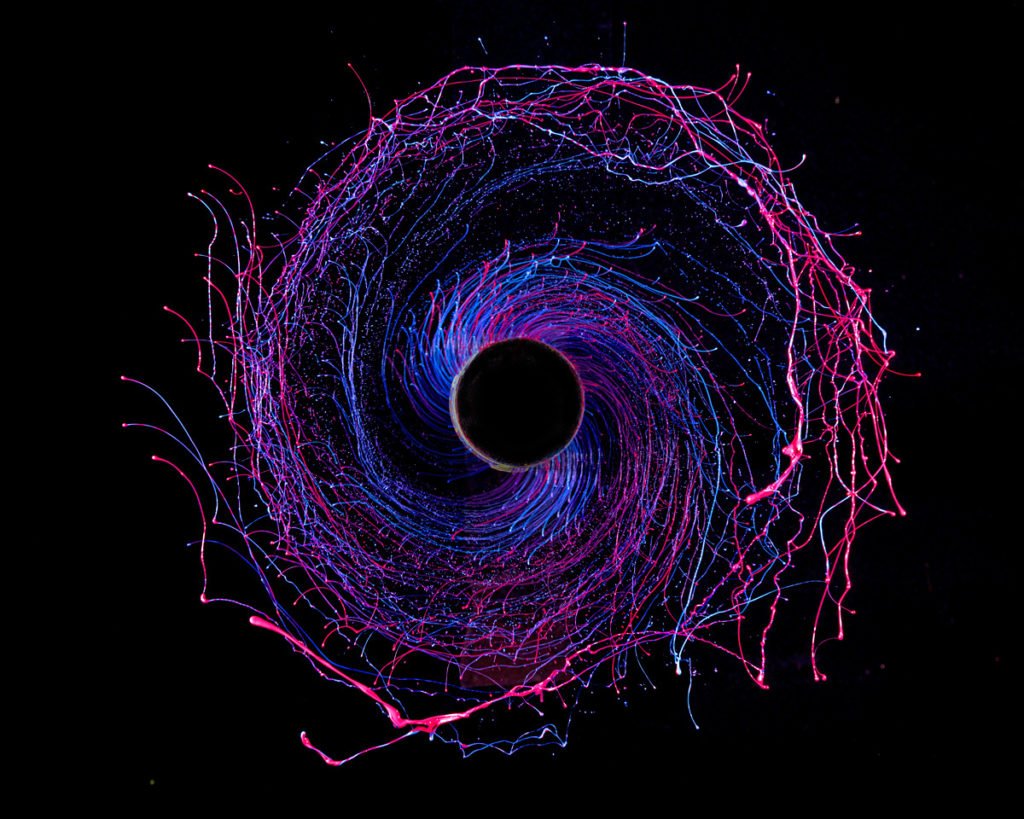
Colorful Spirals
Artist Fabian Oefner captures these colorful portraits of fluid instability by dripping acrylic paints onto a metal rod, which is connected to a drill. When the drill is switched on, paint is flung away from the rod, creating these snapshots of centripetal force and surface tension. Note how droplets gather at the ends of the spiral arms like in a Plateau-Rayleigh or a rimming instability. For more, check out Oefner’s webpage, which includes a video showing how the images are made, or his previously featured work, “Millefiori”. (Photo credit: F. Oefner; submitted by Stephen D.)