When liquids hit a surface much hotter than their boiling point, a thin layer of gas can form between the drop and surface, allowing the drop to glide along. This Leidenfrost effect is what makes drops of water skitter across a hot pan. But what happens when the pan isn’t flat? The video above shows a Leidenfrost drop on a ratchet-like surface. Instead of gliding or skittering randomly, the drop self-propels toward the steepest section of the ratchet This behavior allows researchers to design surfaces that guide the drops on an intended path. (Video credit: G. Lagubeau and D. Quéré)
Tag: droplets

The Red Crown
A drop of red dye falls into a thin layer of milk, forming a crown splash. Notice the pale edges of the droplets at the rim of the crown; this is milk that has been entrained by the original drop. The rim and satellite droplets surrounding the splash are formed due to surface tension effects, chiefly the Plateau-Rayleigh instability–the same effect responsible for breaking a falling column of liquid into droplets like in a leaking faucet. The instability will have a most unstable wavelength that determines the number of satellite droplets formed. (Photo credit: W. van Hoeve et al., University of Twente)

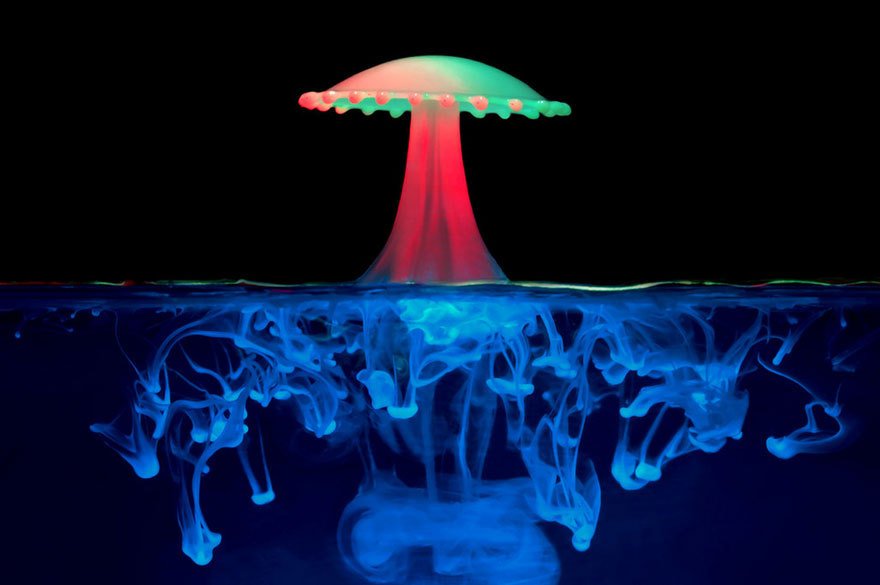
Liquid Sculptures
Artist Corrie White uses dyes and droplets to capture fantastical liquid sculptures at high-speed. The mushroom-like upper half of this photo is formed when the rebounding jet from one droplet’s impact on the water is hit by a well-timed second droplet, creating the splash’s umbrella. In the lower half of the picture, we see the remains of previous droplets, mixing and diffusing into the water via the Rayleigh-Taylor instability caused by their slight difference in density relative to the water. There’s also a hint of a vortex ring, likely from the droplet that caused the rebounding jet. (Photo credit: Corrie White)

Droplet Springs
Prior to reaching terminal velocity, a falling droplet typically oscillates between a prolate shape (like an American football about to be kicked) and an oblate one (like that same football when thrown or carried). As explained by Minute Laboratory, this oscillation behaves very similarly to a mass on a spring. For a spring/mass system, the frequency of oscillation is related to the spring’s stiffness; for the falling droplet, it is instead governed by surface tension. If only high schools had high-speed cameras, this would make a fantastic fluids lab experiment! (Video credit: Minute Laboratory; submitted by Pascal W.)
FYFD is sporting a new look and new about and resources pages for those who want more fluidsy goodness.

Bouncing and Break-Up
In the collage above, successive frames showing the bouncing and break-up of liquid droplets impacting a solid inclined surface coated with a thin layer of high-viscosity fluid have been superposed. This allows one to see the trajectory and deformation of the original droplet as well as its daughter droplets. The impacts vary by Weber number, a dimensionless parameter used to compare the effects of a droplet’s inertia to its surface tension. A larger Weber number indicates inertial dominance, and the Weber number increases from 1.7 in (a) to 15.3 in (d). In the case of (a), the impact of the droplet is such that the droplet does not merge with the layer of fluid on the surface, so the complete droplet rebounds. In cases (b)-(d), there is partial merger between the initial droplet and the fluid layer. The impact flattens the original droplet into a pancake-like layer, which rebounds in a Worthington jet before ejecting several smaller droplets. For more, see Gilet and Bush 2012. (Photo credit: T. Gilet and J. W. M. Bush)

Champagne Science
Today many a glass of champagne will be raised in honor of the end of one year and the beginning of a new. This French wine, known for its bubbly effervescence, is full of fascinating physics. During secondary fermentation of champagne, yeast in the wine consume sugars and excrete carbon dioxide gas, which dissolves in the liquid. Since the bottle containing the wine is corked, this increases the pressure inside the bottle, and this pressure is released when the cork is popped. Once champagne is in the glass, the dissolved carbon dioxide will form bubbles on flaws in the glass, which may be due to dust, scratches, or even intentional marks from manufacturing. These bubbles rise to the surface, expanding as they do so because the hydrodynamic pressure of the surrounding wine decreases with decreasing depth. At the surface, the bubbles burst, creating tiny crowns that collapse into Worthington jets, which can propel droplets upward to be felt by the drinker. For more on the physics of champagne, check out Gerard Liger-Belair’s book Uncorked: The Science of Champagne and/or Patrick Hunt’s analysis. Happy New Year! (Video credit: AFP/Gerard Liger-Belair)

Bouncing in a Corral
About a year ago, we featured a video in which a fluid droplet bouncing on a vibrating pool demonstrated some aspects of the wave-particle duality fundamental to quantum mechanics. Work on this system continues and this new video focuses on studying some of the statistics of such a bouncing droplet–called a walker in the video–when it is confined to a circular corral. Using strobe lighting and capturing one frame per bounce, the vertical motion of these droplets is filtered out and the walking motion and the surface waves that guide it are captured. When the droplet is allowed to walk for an extended time, its path appears complicated and seemingly random, but it is possible to build a statistical picture and a probability density field that describe where the walker is most likely to be, much the way one describes the likelihood of locating a quantum particle. Parallels between the physical macroscale system and quantum-mechanical theory are drawn. (Video credit: D. Harris and J. Bush; submission by D. Harris)
Reader Question: Snow from Boiling Water?
[original media no longer available]
Reader kylewpppd asks:
Have you seen the post of a man in Siberia throwing boiling water off of his balcony? Can you provide a better explanation of what’s going on?
As you can see in the video (and in many similar examples on YouTube), tossing near boiling water into extremely cold air results in an instant snowstorm. Several effects are going on here. The first thing to understand is how heat is transferred between objects or fluids of differing temperatures. The rate at which heat is transferred depends on the temperature difference between the air and the water; the larger that temperature difference is the faster heat is transferred. However, as that temperature difference decreases, so does the rate of heat transfer. So even though hot water will initially lose heat very quickly to its surroundings, water that is initially cold will still reach equilibrium with the cold air faster. Therefore, all things being equal, hot water does not freeze faster than cold water, as one might suspect from the video.
The key to the hot water’s fast-freeze here is not just the large temperature difference, though. It’s the fact that the water is being tossed. When the water leaves the pot, it tends to break up into droplets, which quickly increases the surface area exposed to the cold air, and the rate of heat transfer depends on surface area as well! A smaller droplet will also freeze much more quickly than a larger droplet.
What would happen if room temperature water were used instead of boiling water? In all likelihood, a big cold bunch of water would hit the ground. Why? It turns out that both the viscosity and the surface tension of water decrease with increasing temperature. This means that a pot of hot water will tend to break into smaller droplets when tossed than the cold water would. Smaller droplets means less mass to freeze per droplet and a larger surface area (adding up all the surface area of all the droplets) exposed. Hence, faster freezing!

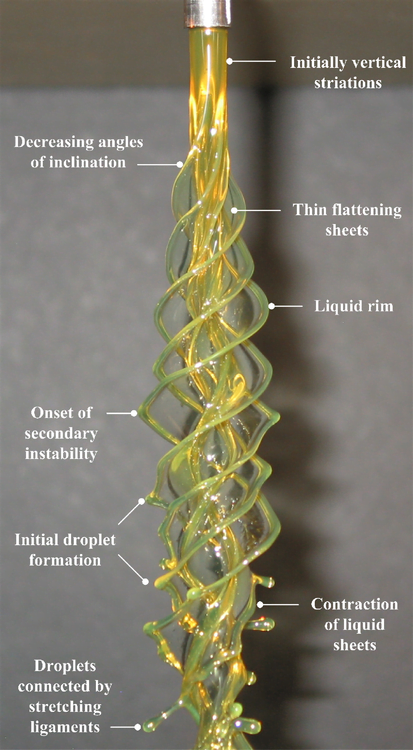
Swirling Jets
In fluid dynamics, we like to classify flows as laminar–smooth and orderly–or turbulent–chaotic and seemingly random–but rarely is any given flow one or the other. Many flows start out laminar and then transition to turbulence. Often this is due to the introduction of a tiny perturbation which grows due to the flow’s instability and ultimately provokes transition. An instability can typically take more than one form in a given flow, based on the characteristic lengths, velocities, etc. of the flow, and we classify these as instability modes. In the case of the vertical rotating viscous liquid jet shown above, the rotation rate separates one mode (n) from another. As the mode and rotation rate increase, the shape assumed by the rotating liquid becomes more complicated. Within each of these columns, though, we can also observe the transition process. Key features are labeled in the still photograph of the n=4 mode shown below. Initially, the column is smooth and uniform, then small vertical striations appear, developing into sheets that wrap around the jet. But this shape is also unstable and a secondary instability forms on the liquid rim, which causes the formation of droplets that stretch outward on ligaments. Ultimately, these droplets will overcome the surface tension holding them to the jet and the flow will atomize. (Video and photo credits: J. P. Kubitschek and P. D. Weidman)

“Kusho”
Artist Shinichi Maruyama uses photography to freeze the transient motion of fluids into water sculptures. Inertia, gravity, and surface tension are at war in each piece. Plateau-Rayleigh instabilities break long filaments of liquid into droplets that splash, collide, and reform. To see how he makes this art, check out his videos. (Photo credits: Shinichi Maruyama)