Light rain after a dry spell often produces a distinctive earthy scent called petrichor that is associated with plant oils and bacteria products. How these chemicals get into the air has been unclear, but new research suggests that the mechanism may come from the rain itself. When water falls on a porous surface like soil, tiny air bubbles get trapped beneath the drop. These bubbles rise rapidly due to buoyancy and, upon reaching the surface, burst and release tiny droplets known as aerosols. Depending on the surface properties and the drop’s impact speed, a single drop can produce a cloud of aerosol droplets. The research team is now investigating how readily bacteria or pathogens in the soil can spread through this mechanism. Other human-focused research has already shown that these tiny aerosol droplets can persist in the air for remarkably long periods and may help spread diseases. (Video credit: Massachusetts Institute of Technology; research credit: Y. Joung and C. Buie; submitted by Daniel B and entropy-perturbation)
Tag: droplet impact

Viscous Droplet Impacts
Viscosity can have a notable effect on droplet impacts. This poster demonstrates with snapshots from three droplet impacts. The blue drops are dyed water, and the red ones are a more viscous water-glycerol mixture. When the two water droplets impact, a skirt forms between them, then spreads outward into a sheet with a thicker, uneven rim before retracting. The second row shows a water droplet impacting a water-glycerol droplet. The less viscous water droplet deforms faster, wrapping around and mixing into the other drop before rebounding in a jet. The last row switches the impacts, with the more viscous drop falling onto the water. As in the previous case, the water deforms faster than the water-glycerol. The two mix during spreading and rebound slower. In the last timestep shown, the droplet is still contracting, but it does rebound as a jet thereafter. (Image credit: T. Fanning et al.)

Raindrops on Sand
Here is a high-speed look at the impact of a raindrop on a sandy beach. In this case, a water droplet is falling on a bed of uniform glass beads, but the situation is effectively the same. Depending on the speed of the drop at impact, many types of craters are possible. The higher the impact velocity, the greater the momentum of the drop at impact and the more likely the drop is to tear apart when surface tension can no longer hold it together. Interestingly, there is remarkable similarity between the shape and behavior of these liquid drop impacts and those of a catastrophic asteroid impact. (Video credit: R. Zhao et al.)

Momentary Crown
When a drop falls on a liquid film, its impact drives a thin liquid sheet called the ejecta upward and outward from the point of impact. Within milliseconds, tiny perturbations develop in the ejecta and begin growing exponentially. These become the distinctive spikes of the crown. The momentum from the impact drives the ejecta and spikes further outward until it overcomes surface tension’s ability to hold the liquid crown together. Tiny droplets escape the crown before the ejecta comes crashing down. The whole process takes only a few hundred milliseconds from start to finish. (Photo credit: S. Jung et al.)

Bounce or Freeze?
Icing is a major problem for aircraft. When ice builds up on the leading edge of a wing it creates major disruptions in flow around the wing and can lead to a loss of flight control. One of the important factors in predicting and controlling ice building up is knowing when and where water droplets will freeze. The video above shows how surface conditions on the wing affect how an impacting droplet freezes. On a subzero hydrophilic surface, a falling droplet spreads and freezes over a wide area, which would hasten ice buildup. A hydrophobic surface is slightly better, with the droplet freezing over a smaller area, whereas a superhydrophobic surface shows no ice buildup. Unfortunately, at present superhydrophobic surfaces and surface treatments are extremely delicate, making them unsuitable for use on aircraft leading edges. (Video credit: G. Finlay)

What’s in a Splash?
A droplet falling onto a solid, dry surface seems like a simple situation, one that would be easy to understand. But splashes can be unpredictable. Velocity, viscosity, and surface tension all play clear roles, but the surrounding air also has an impact – drop the air pressure low enough and a droplet won’t splash. A new paper has tackled the problem, producing a mathematical model in agreement with experimental results. So why do some drops splash and others don’t? When a drop falls, its momentum flattens it into a pancake shape while surface tension struggles to hold it together. The spreading edge, called the lamella, can pull away from the surface. When it does, a pocket of high pressure forms beneath it due to lubrication effects, and the faster airflow over the top of the lamella creates a suction effect. This is analogous to a wing producing lift. Like the momentum that spread the droplet, the lift force pulls the lamella and ejecta sheet further up and outward, overcoming the restoring force of surface tension and tearing the droplet apart. For more on the effect, check out the research paper or this Inside Science article. (Video credit: G. Riboux and J. Gordillo; via Inside Science)

The March of Drops
I love science with a sense of humor. This video features a series of clips showing the behavior of droplets on what appears to be a superhydrophobic surface. In particular, there are some excellent examples of drops bouncing on an incline and droplets rebounding after impact. For droplets with enough momentum, impact flattens them like a pancake, with the rim sometimes forming a halo of droplets. If the momentum is high enough, these droplets can escape as satellite drops, but other times the rebound of the drop off the superhydrophobic surface is forceful enough to overcome the instability and draw the entire drop back off the surface. (Video credit: C. Antonini et al.)

Inside a Splash

When a droplet strikes a pool, a thin, fast-moving sheet of liquid expands outward from the region of contact. These ejecta sheets come in many forms depending on surface tension, viscosity, air pressure, and droplet momentum. When the ejecta sheet curls downward to touch the pool, it can spray microdroplets outward or trap a layer of air underneath the droplet. For more, see this video by S. Nagel et al., and the papers Thoroddsen (2002) and Thoroddsen et al. (2008). (Photo credits: S. Thoroddsen et al.; GIF from this video by S. Thoroddsen et al.)

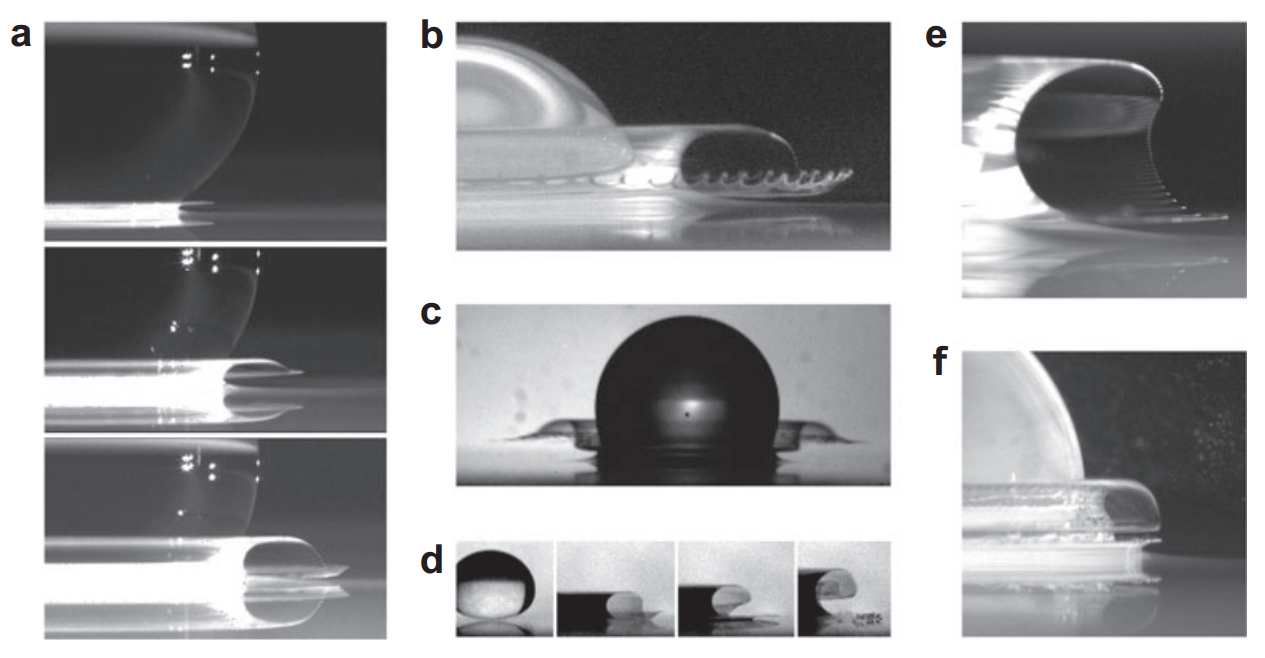
Viscosity’s Impact
Everyone has seen drops of liquid falling onto a dry surface, yet the process is still being unraveled by researchers. We have learned, for example, that lowering the ambient air pressure can completely suppress splashing. Viscosity of the fluid also clearly plays a role, but the relationship between these and other variables is unclear. The images above show two droplet impacts in which the viscosity differs. The top image shows a low viscosity fluid, which almost immediately after impact forms a thin expanding sheet of fluid that lifts off the surface to create a crownlike splash. In contrast, the higher viscosity fluid in the bottom image spreads as a thick lamella with a thinner outer sheet that breaks down at the rim. Researchers found that both the high- and low-viscosity fluids have splashes featuring these thin liquid sheets, but the time scales on which the sheet develops differ. Moreover, lowering the ambient pressure increases the time required for the sheet to develop regardless of the fluid’s viscosity. (Image credit: C. Stevens et al.; submitted by @ASoutIglesias)

Air Pressure Affects Splashes
When a drop falls on a dry surface, our intuition tells us it will splash, breaking up into many smaller droplets. Yet this is not always the case. The splashing of a droplet depends on many factors, including surface roughness, viscosity, drop size, and–strangely enough–air pressure. It turns out there is a threshold air pressure below which splashing is suppressed. Instead, a drop will spread and flatten without breaking up, as shown in the video above. For contrast, here is the same fluid splashing at atmospheric pressure. This splash suppression at low pressures is observed for both low and high viscosity fluids. Although the mechanism by which gases affect splashing is still under investigation, measurements show that no significant air layer exists under the spreading droplet except near the very edges. This suggests that the splash mechanism depends on how the spreading liquid encroaches on the surrounding gas. (Video credit: S. Nagel et al.; research credit: M. Driscoll et al.)