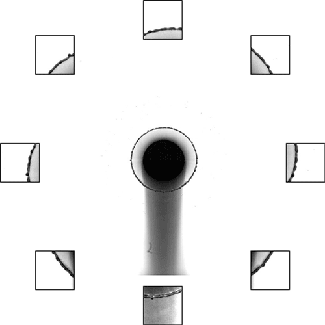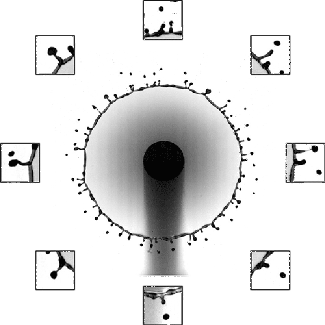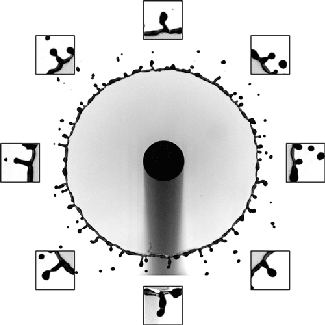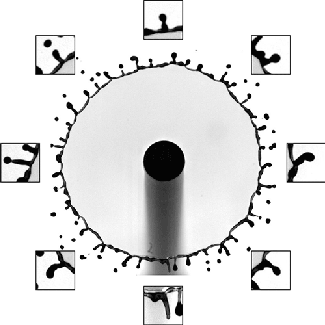
Although many people have studied droplet impacts over the years, there’s been remarkably little work done with oil-on-water impacts. One of the things that makes this situation different is that the oil and water are completely immiscible, which means we can see aspects of the impact process that are invisible with, say, water-on-water impacts.
The animation above shows an underwater view of the oil droplet’s impact. The energy of the initial impact creates an expanding crater and an unstable crown splash. That crown splash contains both water and oil. After it ejects some droplets, the rim stabilizes, but we can still see small perturbations along its edge as it starts to retract. In the water, high surface tension damps out these perturbations. Not so for the oil! As the crater retracts, the small disturbances along the rim get stretched into mushroom-shaped fingers that point inward toward the impact site. Because the index of refraction is different between oil and water, we can see the fingers clearly near the end of the animation. (Image and research credit: U. Jain et al.; submitted by Utkarsh J.)