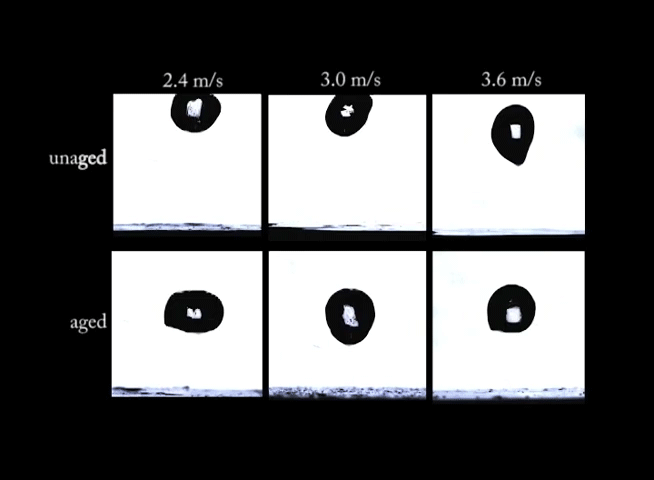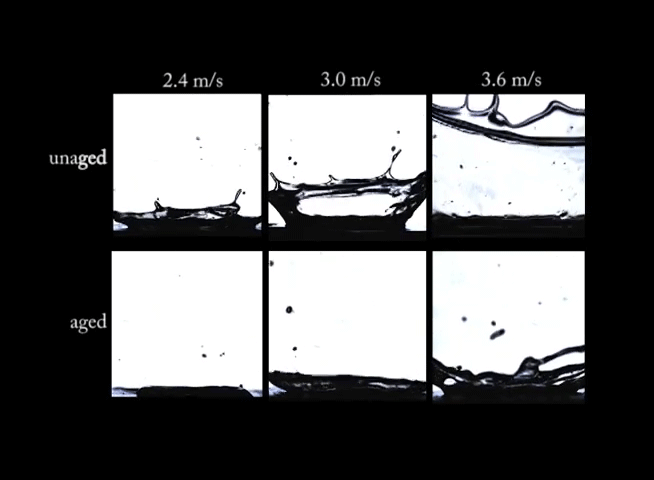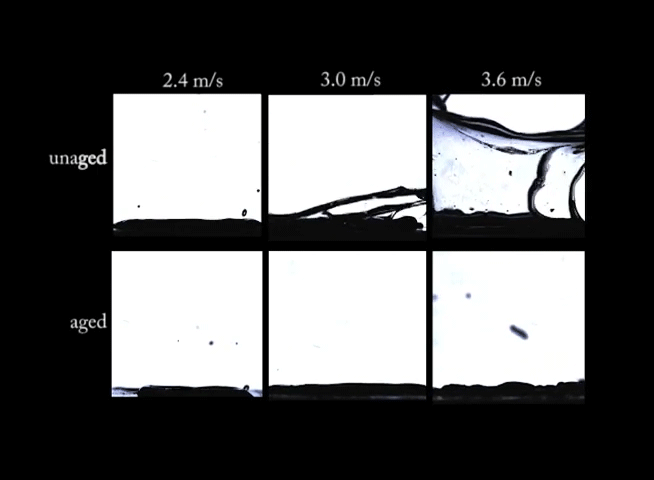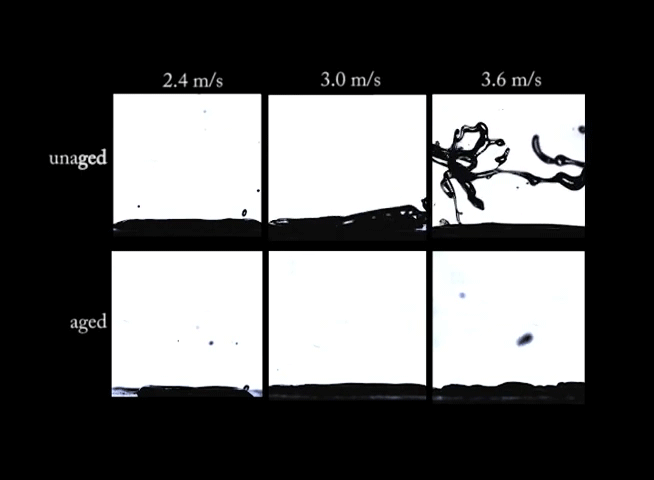
If you’ve ever left a sealed container of Playdoh untouched for months, you know that there’s a big difference between the fresh stuff and what’s left in that can. Aging can have big effects on non-Newtonian fluids. In this video, we see drops of a synthetic clay impacting at different speeds. In the top row of images, the clay is fresh and unaged; on impact, the clay forms large crown-like splashes. In the bottom row, however, the aged clay behaves quite differently. Instead of a splash, the drops make more of a splat. (Image and video credit: R. Ewoldt et al.)
Tag: droplet impact

Jets Beneath Leidenfrost Drops
When a droplet impacts, it’s not unusual for converging ripples to form an upward jet, like the one seen here. But under the right circumstances, jets can form downward, too. This study looks at the ultrafast jets that can form beneath an impacting Leidenfrost drop.
These Leidenfrost drops are striking a surface much hotter than their boiling point, so a large vapor cavity forms quickly beneath them. Using x-ray imaging, the researchers were able to capture the dynamics of this cavity’s formation and collapse (Image 2). The field of view in the animation shows only a portion of the drop’s cavity, so Image 3 may help you orient relative to the drop at large.
Initially, we see the center of the droplet hitting the surface, followed by the fast growth of a vapor cavity. Rippling capillary waves converge on top of the cavity, creating a pinch-off. From there, a bubble rises up while a fast jet shoots downward. (Image credit: water jet – A. Min, others – S. Lee et al.; research credit: S. Lee et al.)

Droplets on Inclined Walls
When a droplet impacts an inclined surface, it spreads asymmetrically. The splash shape is largely elliptical, as researchers found when modeling such impacts over a range of inclination angles. Understanding such splash patterns is important not only for industrial applications like printing but in areas like forensic science. (Image and research credit: P. García-Geijo et al.)

Freezing Splats
When a drop hits a surface colder than its freezing point, there’s a competition between retraction and solidification that determines the final shape of the splat. For many materials, like wax or soldering metals, the contact angle between their liquid and solid phase is zero, so there’s no major shape change once solidification begins. But water — as is so often the case — is an exception.
Water and ice have a non-zero contact angle, which means that retraction can continue even after the drop begins freezing. As a result, the final shape of the splat varies depending on how cold the surface is. For a surface only a little colder than the freezing point, the final splat forms a spherical cap (Image 1). But once the surface is colder, freezing happens before the water can fully retract and the final splat forms a ring (Image 2). (Image and research credit: V. Thiévenaz et al.)

How Animals Stay Dry in the Rain
Getting wet can be a problem for many animals. A wet insect could quickly become too heavy to fly, and a wet bird can struggle to stay warm. But these animals have a secret weapon: tiny, multi-scale roughness on their wings, scales, and feathers that helps them shed water. Watch the latest FYFD video to learn how! (Image and video credit: N. Sharp; research credit: S. Kim et al.)

Particle-filled Splashes
Adding particles to a liquid can significantly alter its splash dynamics, as shown in this new study. In the first image, a purely-liquid droplet spreads on impact into a thin liquid sheet that destabilizes from the rim inward, ripping itself into a spray of droplets. At first glance, the particle-filled droplet in the second image behaves similarly; it, too, spreads and then disintegrates. But there are distinctive differences.
During expansion, the particles increase the drop’s effective viscosity, meaning that the splash sheet does not expand as far. That apparent viscosity increase is also part of why the drops the splash sheds are bigger than those without particles. The other part of that story comes from the retraction, where the variations in thickness caused by the particles and their menisci create preferential paths for the flow. As a result, the particle-filled splash breaks up faster and into larger droplets compared to its purely-liquid counterpart. (Image and research credit: P. Raux et al.)

Watching a Droplet Freeze
Whether it’s rain hitting an airplane wing or droplet-based 3D printing, the dynamics of a droplet impacting and solidifying on a surface are important. This new study observes the process from below, tracking the progress of freezing on a scale of hundreds of nanoseconds.
All three of the drops you see above are liquid hexadecane. Each droplet was the same size and impacted at the same velocity. What differs in each image is how much colder the surface was than hexadecane’s melting point. The leftmost image shows a droplet on a surface only a few degrees cooler than the melting point. The initial expanding ring shows the droplet’s contact line expanding as it impacts. Then frozen crystals appear and grow inside the drop until the entire thing freezes.
With a slightly colder surface (middle image), frozen crystals form while the contact line is still expanding, and rather than form in distinctive spots, they form as a cloud that quickly expands throughout the drop.
But with an even colder surface (right image), something entirely new happens. As the drop freezes, we see multiple dark rings expand through the drop. Each of these rings is made up of frozen crystals. The researchers argue that we’re seeing a combination of freezing and hydrodynamics here. Essentially, whenever the frozen crystals get large enough, the outward flow of the impacting drop sweeps them toward the contact line. As new crystals grow near the center of the drop, they’re dragged out in a subsequent wave. (Image, research, and submission credit: P. Kant et al.)

Bouncing Off Hydrophilic Surfaces
Droplets typically bounce off hydrophobic surfaces due to air trapped beneath the liquid that prevents contact between the drop and surface. But even extremely smooth, hydrophilic surfaces can elicit a bounce under the right circumstances, as shown in a new study.
The key is that the droplet must bounce at exactly the right speed. If the bounce has too much momentum, it will squeeze the nanometer-sized air cushion too thin, allowing contact. Too slow and the Van der Waals attraction between the droplet molecules and wall molecules will have time to act. But between those lies a sweet spot where the dimple and cushion of air beneath the drop keep it from impacting. (Image credit: droplets – klickblick, drop bounce – J. Kolinski, bounce sim – J. Sprittles et al.; research credit: M. Chubynsky et al.; submitted by James S.)

Pearls On a Puddle
Leave a drop of coffee sitting on a surface and it will leave behind a ring of particulates once the water evaporates. But what happens to a droplet made up of multiple liquids that evaporate differently? That’s the subject of this new study. Researchers mixed a volatile drop (isopropyl alcohol) with a smaller amount of a non-volatile liquid and observed how this changed the droplet’s splash rim and evaporation pattern.
When the surface tension difference between the two liquids was large, the researchers found that the splash formed fingers along its rim (Image 1). The fingers consist almost entirely of the non-volatile component, driven to the outskirts of the drop by Marangoni forces. The dark and light bands you see in the image are interference fringes, which the researchers used to track the film’s thickness.
When the researchers used liquids with similar surface tensions, the droplet rim instead formed pearl-like satellite droplets. Once the volatile liquid evaporated away, the remaining liquid merged into a thick film. (Image and research credit: A. Mouat et al.; via APS Physics; submitted by Kam-Yung Soh)

Surface Jets in Coalescing Droplets
What goes on when droplets merge is tough to observe, even with a high-speed camera. There are many factors at play: any momentum in the droplets, surface tension, gravity, and Marangoni forces, to name a few. A new study that simultaneously records multiple views of coalescence is shedding some light on these dynamics.
The results are particularly interesting for droplets that are somewhat physically separated so that they only coalesce after one drop impacts near the other. In this situation, with droplets of equal surface tension, researchers observed a jet that forms after impact (Image 1) and runs along the top surface of the coalescing drops (Image 2). That location is a strong indication that the jet is created by surface tension and not other forces.
To test that further, the researchers repeated the experiment but with droplets of unequal surface tension. They found that when the undyed droplet’s surface tension was higher (Image 3), Marangoni forces enhanced the surface jet, as one would expect for a surface-tension-driven phenomenon. But if the dyed droplet had the higher surface tension (Image 4), it was possible to completely suppress the jet’s formation. (Image, research, and submission credit: T. Sykes et al., arXiv)