If you try to build a pyramid of dry glass beads, you’ll have a hard time of it. The frictional forces simply aren’t enough to hold the beads together against the force of gravity. If you add a little water, though, the story is different. The intermolecular forces inside water give it a lot of cohesion, which helps it fill the narrow gaps between beads. That added capillary force gives just enough additional sticking power to hold a pyramid of beads together. (Image and video credit: amàco et al.)
Tag: capillary action

The Drinking Bird
At first glance, the drinking bird is a simple desk toy, but the physics and engineering behind the device is clever enough to have challenged many great minds. In this video, Bill Hammack dissects the drinking bird, revealing the heat engine beneath the felt and feathers. The bird’s drinking is driven by thermodynamics and the relative pressures of fluids in its head and body. When the beak is wetted, fluid wicks up the felted head and slowly evaporates, thereby cooling the vapor inside the head. Some of that vapor condenses, lowering the vapor pressure in the head and allowing liquid to rise from the body. When enough fluid reaches the head, the bird tips forward. This allows vapor to rise up the liquid column into the head, equalizing the pressure between the two ends. The bird sits up with a freshly wetted head and starts the cycle over. Check out the full video for more detail, including a look at what other methods can drive the bird, including bourbon and light bulbs. (Video and image credit: B. Hammack; via J. Ouellette)


Water Atop Oil
At first glance, this image looks much like the impact of any drop on a pool of the same liquid, but that’s not what you’re seeing. This is the impact of a water droplet on a thin film of oil, and the immiscibility of those two fluids has important effects on the collision. When the water drop impacts, it spreads and forms a compound crown that rises out of the fluid. Eventually, that momentum runs out and the crown falls into the liquid.
Water’s intermolecular forces are strong enough to pull the remains of the droplet back in on itself. As that fluid collides at the center, it gets forced up into a central jet with enough energy to eject a droplet or two at its tip. Even though this looks like a Worthington jet, it’s not. Worthington jets form after the collapse of a cavity in the impacted liquid – in other words, they form on pools, not on films. Despite the visual similarity, this central jet is formed entirely differently! (Image and research credit: Z. Che and O. Matar, source; submitted by O. Matar)

Building with Sand
Sand and water make a remarkable team when it comes to building. But the substrate – the surface you build on – makes a big difference as well. Take a syringe of wet sand and drip it onto a waterproof surface (bottom right), and you’ll get a wet heap that flows like a viscous liquid. Drop the same wet sand onto a surface covered in dry sand (bottom left), and the drops pile up into a tower. Watch the sand drop tower closely, and you’ll see how new drops first glisten with moisture and then lose their shine. The excess water in each drop is being drawn downward and into the surrounding sand through capillary action. This lets the sand grains settle against one another instead of sliding past, giving the sand pile the strength to hold its weight upright. (Video and image credit: amàco et al.)


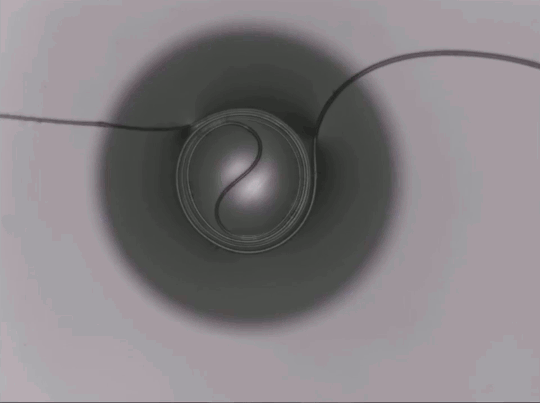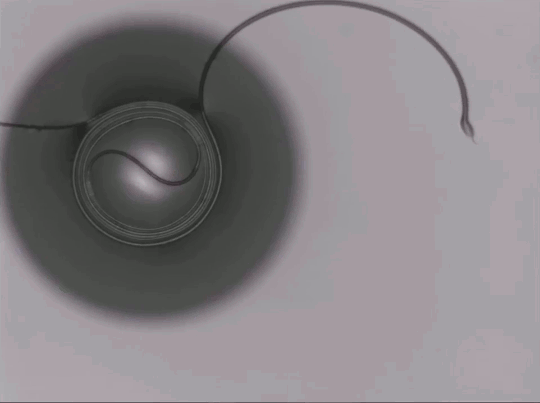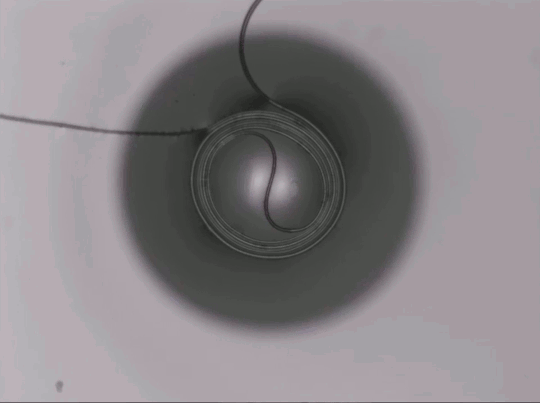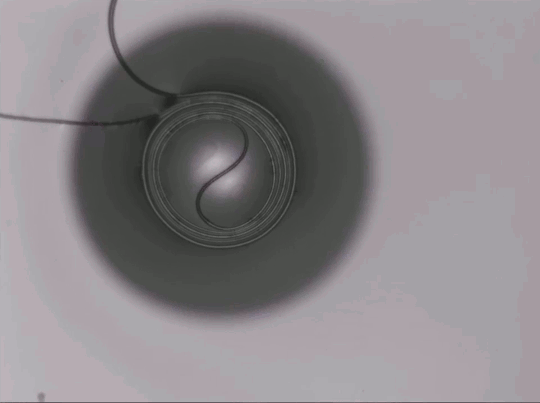
Capillary Action in Microgravity
On Earth, gravity dominates over many fluid effects, but in microgravity a different picture emerges. This animation shows a two-channel apparatus partially filled with silicone oil being dropped. While in free-fall, the liquid experiences microgravity conditions and the height of the fluid in the two connected channels changes. The oil meniscus climbs up the walls of the tubes thanks to capillary action. This is the result of intermolecular forces between the liquid and solid walls. Capillary action is most effective in narrow tubes where surface tension and the adhesion between the liquid and solid can actually propel liquid up the walls, as seen here. On Earth we mostly ignore capillary action, except in very small spaces, but for space systems, it is a major force to reckon with in designing flows. (Image credit: NASA Glenn Research Center, source)

Wrapping Up

It’s often at the intersection of topics that we can learn something new and fascinating. The latest video from The Lutetium Project shows examples of this at the intersection of solid mechanics and fluid dynamics with a look at elastocapillarity. Breaking that word down, that’s where elasticity – that stretchy quality associated with solids – meets capillarity – the surface-tension-dominated behavior of a fluid. In particular, they explore some of the mind-boggling and surprising interactions that happen between drops, bubbles, and thin flexible fibers smaller than the width of a human hair. Check out the full video below. (Images credit: K. Dalnoki-Veress et al.; video credit: The Lutetium Project)

Granular Plugs

Imagine filling a narrow tube with a mixture of water and tiny glass beads. Then take a syringe and very slowly start drawing out the water. As the water gets sucked out of the tube, air will be pulled into the opposite end. The meniscus where the air and water meet sweeps up the glass beads like a liquid bulldozer. As the experiment continues, pressure builds up and air starts filtering through the beads, changing the viscous and frictional forces the system experiences. Eventually, the grains break off, leaving a chunk of glass beads – known as a plug – behind. Keep draining the tube and more plugs form. Check out the video below to see it in action! (Image/video credit: G. Dumazer et al., source; research paper; open synopsis; submitted by B. Sandnes)

Coffee-Making in Space
In this video, Kjell Lindgren demonstrates his technique for making coffee aboard the Space Station. Astronauts usually drink coffee reconstituted from powder, or, on special occasions, enjoy a beverage from their special espresso machine. But Lindgren uses a pour-over method by attaching a pod of coffee grounds to the underside of a Capillary Beverage Experiment cup – a specially-designed 3D printed cup that uses capillary action and surface tension to guide fluids. Then, by forcing hot water from a syringe through the grounds and into the cup, he gets a result that’s not too different from the way many people enjoy their coffee here on Earth. I must say, though, that my favorite part of this video is how he just starts spinning to separate the air and water in the syringe! (Video credit: NASA; via IRPI LLC)

Drinking in Space
Earlier this year, the Capillary Beverage experiment launched to the International Space Station with new open-topped “Space Cups” for astronauts to test. Now those of us back on Earth are getting a glimpse of the cups in microgravity action. The geometry of the cups is wide on the back-end with a tightening v-shape near the mouth. This shape guides the liquid by using capillary action to wick it toward the spout.
One of the key goals of the experiment was to observe how the liquid drained–what shape it assumed in the cup and where and how much liquid was left behind. The researchers want to compare the real-life performance of the cups with their numerical models and simulations, which will help design future microgravity liquid transport systems for fuel, waste management, and other applications.
Although the experiments have a wider purpose, the space cups also do a great job allowing astronauts to drink from more than just pouches. Check out the gallery demo above to see how they hold up against astronaut silliness! (Video and image credits: NASA/IRPI LLC, GIF source)