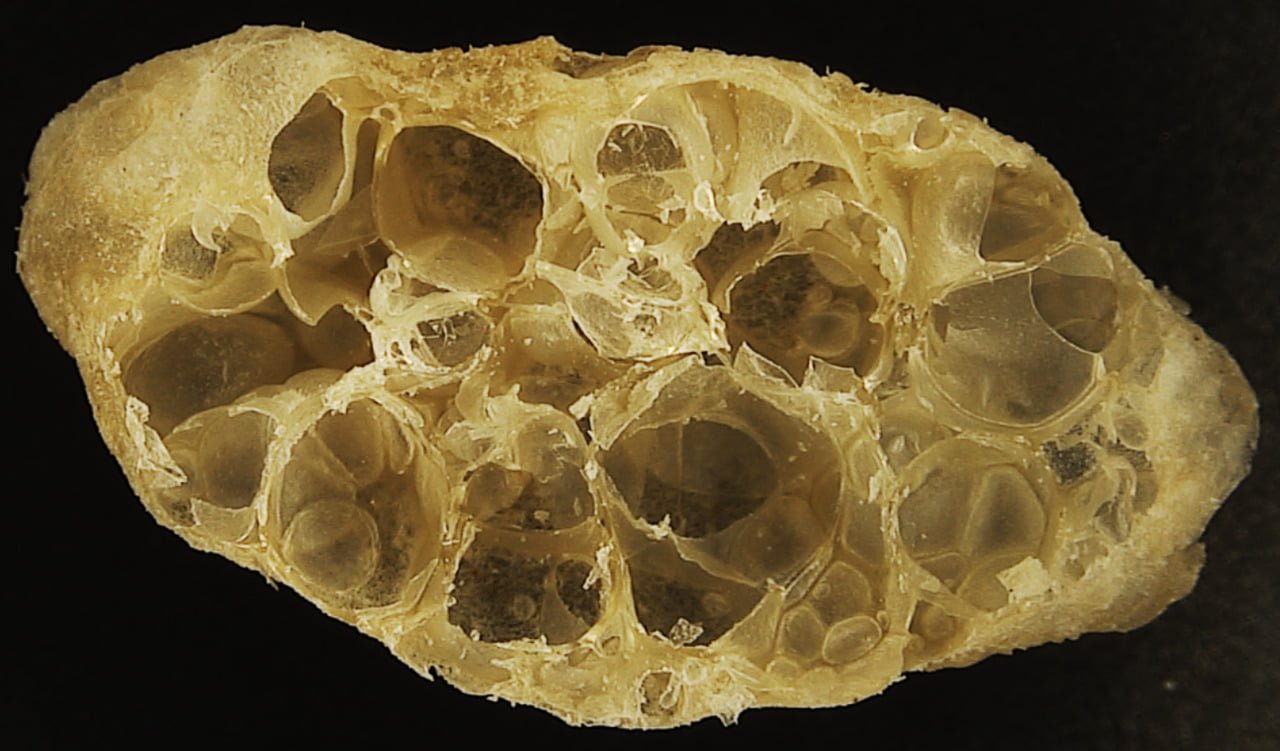
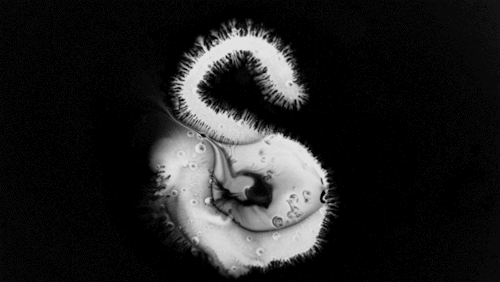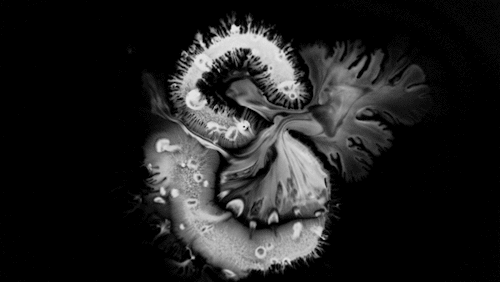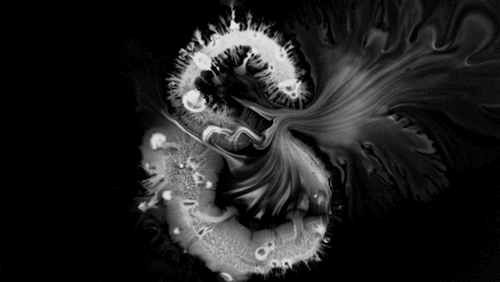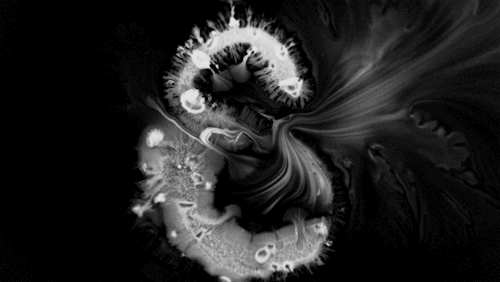
Carnivorous pitcher plants supplement their nutrient-poor environments by capturing and consuming insects. The viscoelastic fluid inside them helps trap prey, but fluid dynamics plays a role elsewhere on the plant as well. The inner and outer surfaces of the pitcher are covered in macroscopic and microscopic grooves, seen above, oriented toward the interior of the plant.
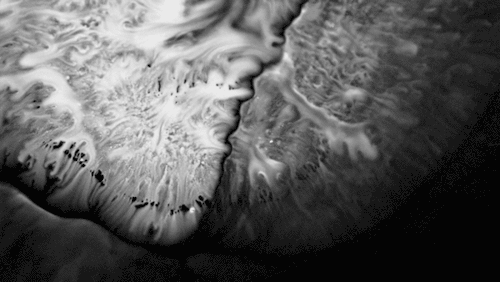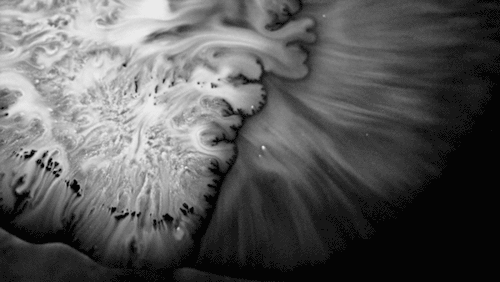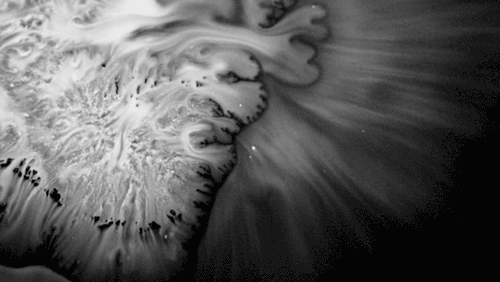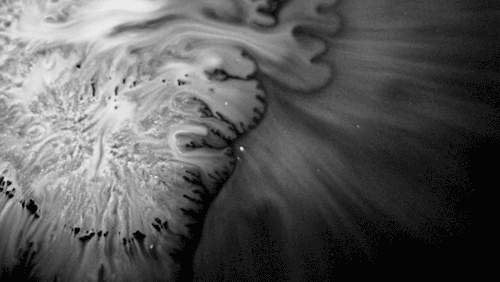
Researchers found that these grooves trap droplets on the slippery plant through capillary action. Once adhered, the droplet cannot easily move across the grooves, but it can slip along them, carrying the droplet and any insect stuck to it, into the plant. By replicating pitcher-plant-inspired grooves on manmade surfaces, researchers found they were able to better control droplet motion on slippery, lubricant-infused surfaces than in previous work. (Image and research credit: F. Box et al.; via Royal Society; submitted by Kam-Yung Soh)