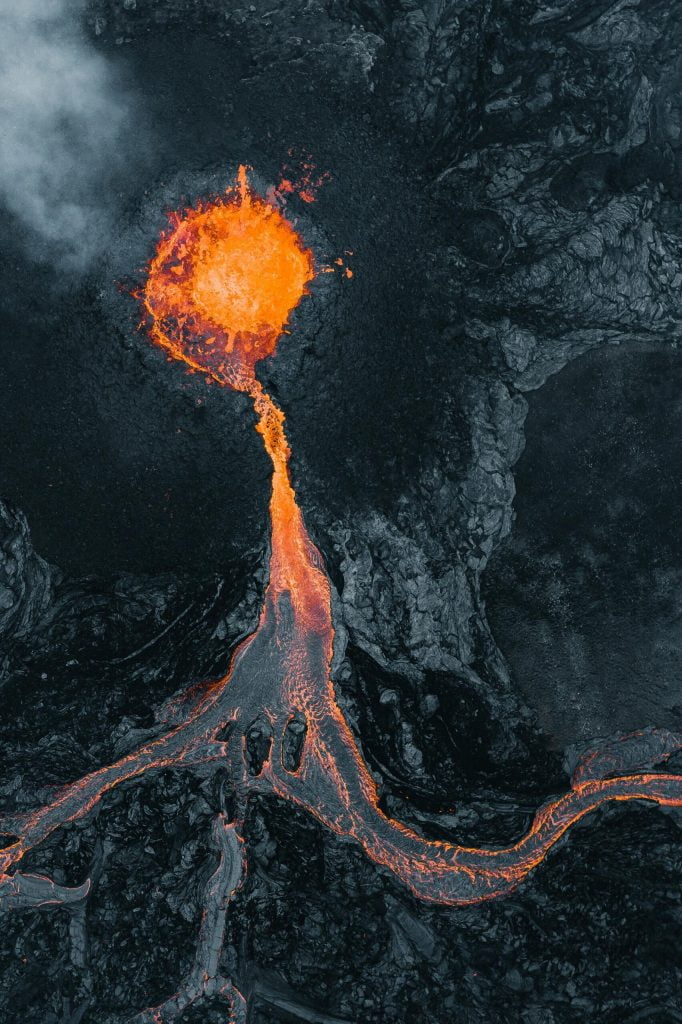
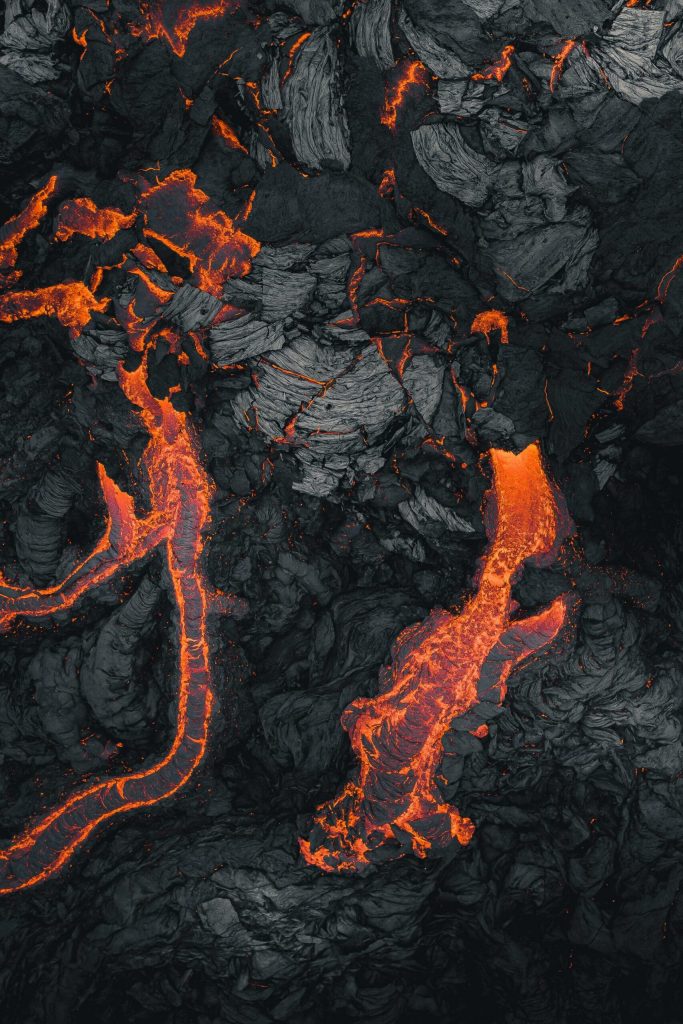
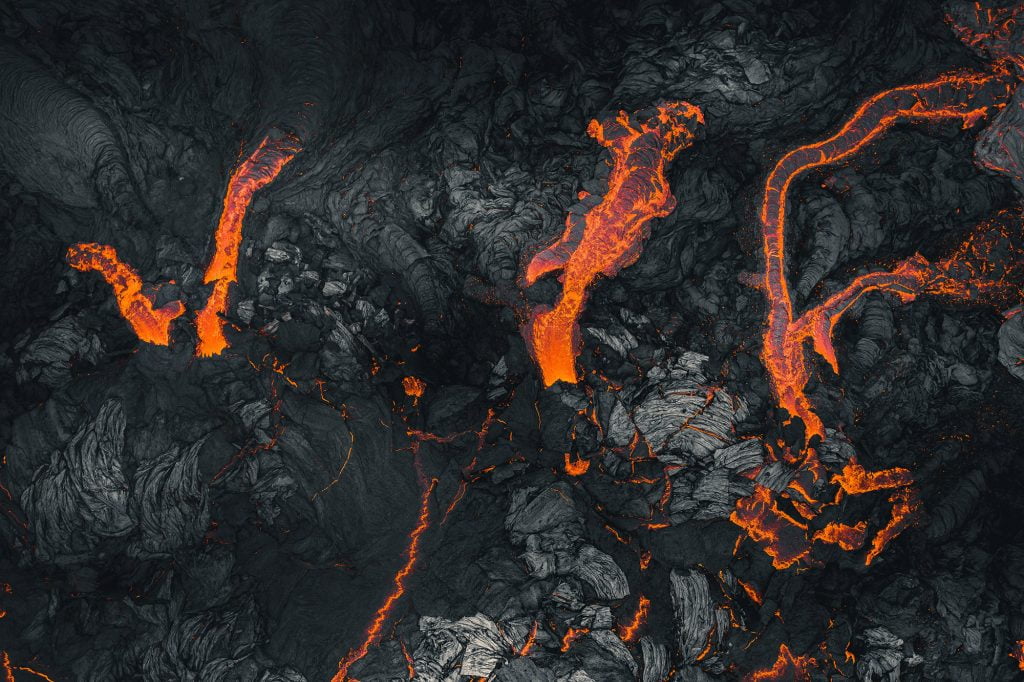Lava flows are endlessly fascinating to watch. They’re a destructive act of creation that seems in many ways familiar; after all, lava moves the same way we see other viscous fluids move. But it’s so much more extreme in its temperature, viscosity, and destructive potential. These beautiful aerial photos by photographer Thrainn Kolbeinsson show the recent eruption at Iceland’s Fagradalsfjall volcano. I love the vivid texture of the lava in these shots and the sharp contrast between the hot and cooling flows. You can see the pahoehoe forming before your very eyes! (Image credit: T. Kolbeinsson; via Colossal)
Search results for: “viscous”

Viscoelastic Coiling
Drizzle honey or syrup from high enough, and you’ll see it coil like a liquid rope. This feature of viscous fluids also extends to polymer-filled viscoelastic fluids. But recent work shows that the elasticity of these fluids delays the onset of coiling; put differently, if you pour two fluids of comparable viscosity, the viscoelastic one will have to fall farther before it will start coiling. The authors also found that the coiling frequency for a viscoelastic fluid is smaller than a viscous one, given the same experimental conditions. (Image credit: flo222; research credit: Y. Su et al.)

Chaotic Mixing in Porous Media
One of the peculiar characteristics of viscous, laminar flows is that they are reversible. Squirt dye into glycerin, stir it one way, then the opposite direction, and the dye returns to its initial position. But this neat trick only works in simple geometries; in a more complex environment, like the pores between packed gravel, flows cannot make their way back to their initial state.
That’s the idea at the heart of this new study of mixing in porous media. Researchers took a bed of packed beads and pushed a slow, steady flow of dye into the bed. Then they steadily withdrew fluid to reverse the flow and observed how the dye they’d injected appeared at the surface of the bed (top image). If the flow were perfectly reversible, we’d expect the dye to return to its injection point. But instead the dye is spread chaotically across the surface, giving researchers a snapshot of the chaotic mixing taking place between beads. (Image and research credit: J. Heyman et al.; via APS Physics)
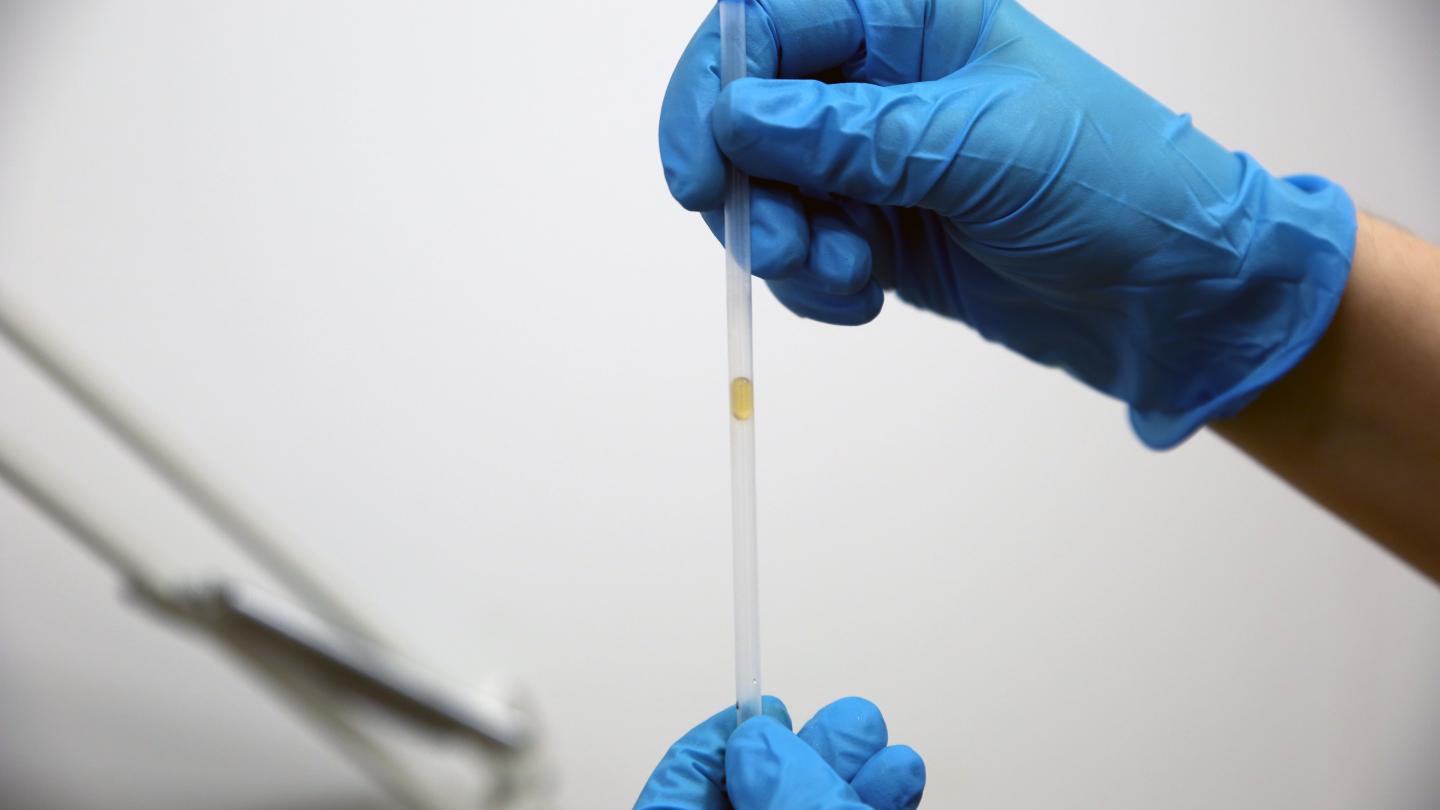
When Honey Flows Faster Than Water
With its high viscosity, no one would ever pick honey to beat water in a race. But a new study shows there’s at least one circumstance where honey wins: inside a narrow, superhydrophobic tube with one or both ends closed. Inside these specially coated tubes a narrow cushion of air stays between the drop and the wall, reducing friction and increasing flow speed for both fluids.
But when one or both ends of the tube are blocked, the drops can only move when air squeezes past. In less viscous fluids, like water, the researchers found rapid internal flows inside the drop. These flows pressed the surface of the drop outward, reducing the air cushion and making it harder for air to squeeze past so that the drop could flow. In contrast, honey showed very little internal flow and so was able to flow through the tubes ten times faster than water! (Image and research credit: M. Vuckovac et al.; via Physics World; submitted by Kam-Yung Soh)

Viscoplastic Drop Impact
There are many materials that don’t behave exactly as a fluid or a solid, instead displaying characteristics of both. In this video, we see drops of hair gel falling into water. The gel is viscoplastic – showing some of the viscous behavior of a fluid and some of the plastic behavior (the inability to change back to its initial shape) of a solid.
On impact, the gel deforms due to the forces on it, but the final shape does not depend solely on the amount of force; instead, it’s the rate at which the forces are applied that determines the final shape. By tuning the impact speed and the gel stiffness, it’s possible to make many final capsule shapes, something that could be useful in applications like drug manufacturing. (Image and video credit: M. Jalaal et al.)

“Chocolate Lullaby”
In this music video for the song “Chocolate Lullaby,” the Macro Room team feature all kinds of fluid dynamical phenomena. It begins with pouring viscous fluids, which, like honey or cake batter, fold and stack before they spread. From there things get significantly less viscous and more turbulent. There’s some neat coalescence, billowing streams colliding, and some gorgeous turbulence. Enjoy! (Image and video credit: Macro Room)

Wrinkles on Bubble Collapse
A viscous bubble wrinkles when it collapses, and scientists long assumed this behavior was caused by gravity. But a new experiment shows that the buckling is, instead, driven by surface tension.
To test gravity’s influence on bubble collapse, the researchers popped bubbles in three orientations: the (normal) upright orientation (Images 1 and 2), upside-down (Image 3), and sideways (Image 4). In all cases, the bubble’s thin film wrinkled as it collapsed, indicating that gravity had little influence on the process. Instead the authors concluded that surface-tension-driven collapse causes the dynamic buckling of the film. (Image and research credit: A. Oratis et al.; submitted by Zander B.)

Floating in Levitating Liquids
When it comes to stability, nature can be amazingly counter-intuitive, as in this case of flotation on the underside of a levitating liquid. First things first: how is this liquid layer levitating? To answer that, consider a simpler system: a pendulum. There are two equilibrium positions for a pendulum: hanging straight down or pointing straight up. We don’t typically observe the latter position because it’s unstable; the slightest disturbance from that perfectly vertical situation will make it fall. But it’s possible to stabilize an inverted pendulum simply by shaking it up and down. The vibration creates a dynamic stability.
The same physics, it turns out, holds for a layer of viscous fluid. With the right vibration, the denser fluid can levitate stably over a layer of air. Inside this vibrating layer, the rules of buoyancy are a little different because the vibration modifies the effects of gravity. As a result, bubbles deep in the liquid layer sink (Image 1). The researchers used this behavior to create their levitating layer (Image 2). The shaking also serves to stabilize objects floating on the underside of the liquid layer, allowing the boat in Image 3 to float upside down! (Image and research credit: B. Apffel et al.; via NYTimes; submitted by multiple sources)

Granular Fingers
Finger-like shapes often form on fluids injected between glass plates, but what happens when that injected fluid contains particles? That’s the situation in this recent study, where researchers sandwiched a fluid between two glass plates and then injected a second, similar fluid laced with particles.
Despite the differences from the traditional Saffman-Taylor set-up, the granular-filled fluid still forms fingers as long as there’s even a slight density difference between the original and injected fluids. It doesn’t even matter which of the two fluids has the greater density! (Image and research credit: A. Kudrolli et al.)

Lava Barriers
Inspired by protecting people and property from lava flows, researchers investigated how viscous fluids flow downhill past large obstacles. As seen above, when the obstacle is tall enough that the flow does not overtop it, there’s substantial deflection of the fluid both up- and downstream. Upstream of the barrier, the flow gets deeper, and downstream there’s a dry region left behind.
The researchers modeled these flows numerically, leading to equations designers can use to predict the necessary height, strength, and shape of barrier necessary to protect areas from encroaching lava. (Image and research credit: E. Hinton et al.)