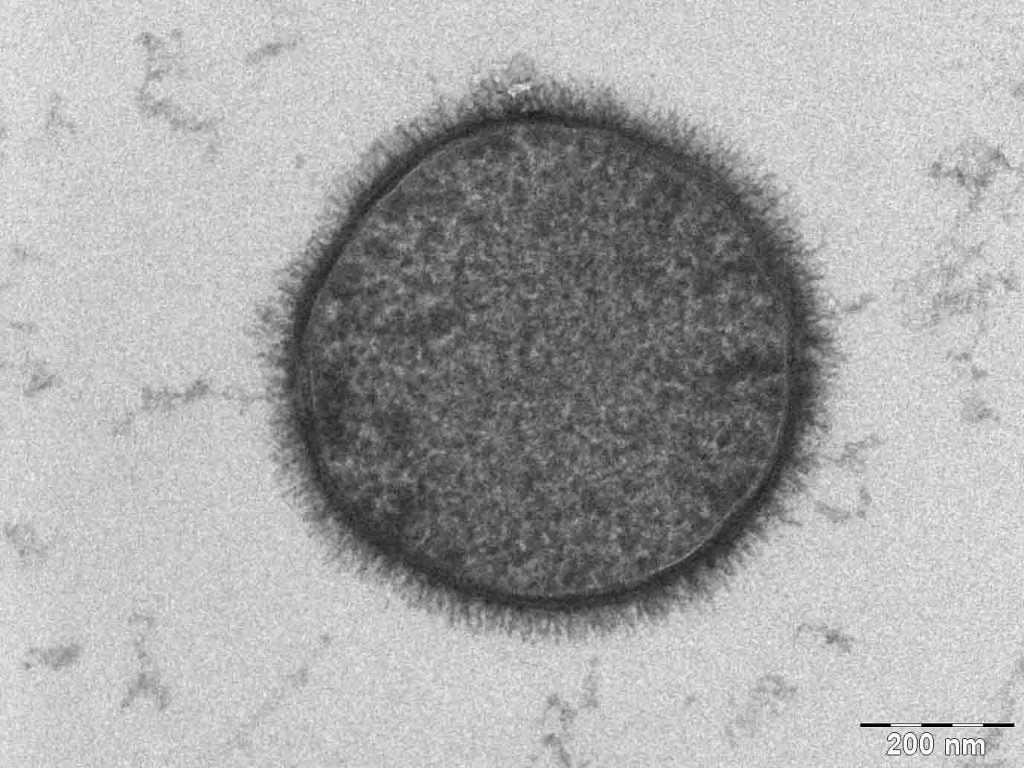
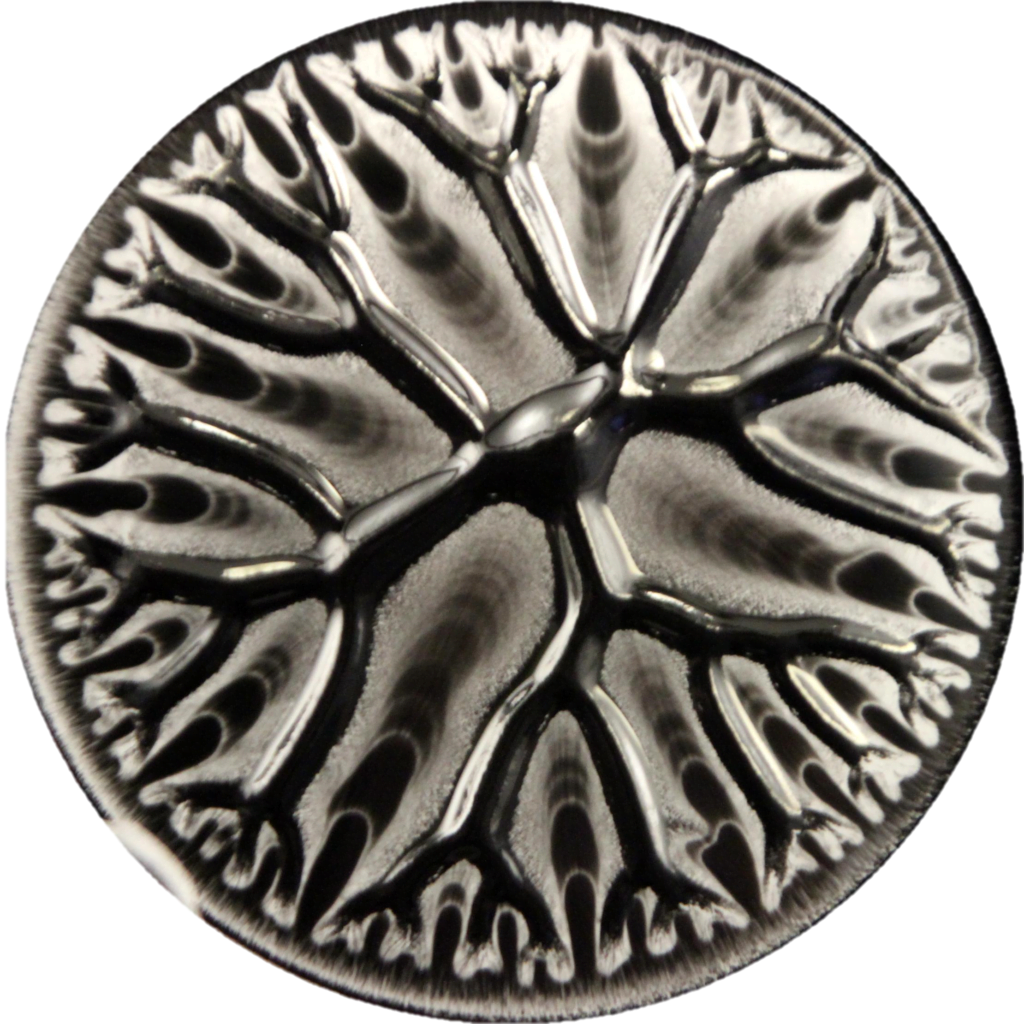
“What happens when two scientists, a composer, a cellist, and a planetarium animator make art?” The answer is “Dendritic,” a musical composition built directly on the tree-like branching patterns found when a less viscous fluid is injected into a more viscous one sandwiched between two plates.
Normally this viscous fingering instability results in dense, branching fingers, but when there’s directional dependence in the fluid, the pattern transitions instead to one that’s dendritic. In this case, that directionality comes from liquid crystals, whose are rod-like shape makes it easier for liquid to flow in the direction aligned with the rods.
For more on the science, math, and music behind the piece, check out this description from the scientists and composer. (Video, image, and submission credit: I. Bischofberger et al.)