When less viscous fluids are injected into a more viscous medium, the low-viscosity fluid forms finger-like protrusions into the background fluid. This is known as the Saffman-Taylor instability. The video above shows this effect but in a more dynamic setting. Blue-dyed water and a clear solution of water and glycerol fifty times more viscous than the water are injected in alternating fashion to a microfluidic channel. The blue water spreads into the clear glycerol solution via fingers that quickly diffuse, creating a homogeneous–or uniform–mixture. (Video credit: Juanes Research Group)
Search results for: “viscous”

Viscous Fluid Falling on a Moving Belt
In this video a very viscous (but still Newtonian) fluid is falling in a stream onto a moving belt. Initially, the belt is moving quickly enough that the viscous stream creates a straight thread. As the belt is slowed, the stream begins to meander sinusoidally and ultimately begins to coil. Aside from some transient behavior when the speed of the belt is changed very quickly, the behavior of the thread is very consistent within a particular speed regime. This is indicative of a nonlinear dynamical system; each shift in behavior due to the changing speed of the belt is called a bifurcation and can be identified mathematically from the governing equation(s) of the system. (Video credit: S. Morris et al)

Viscous Fingers
A Hele Shaw cell is little more than two glass plates separated by a thin layer of viscous fluid. The cell serves as a good test bed for viscous, low Reynolds number flows such as those found in microfluidics. Here a less viscous fluid is injected into the center of the cell, causing the finger-like protrusions of the less viscous fluid into the more viscous one via the Saffman-Taylor instability.

Viscous Fingers
The Saffman-Taylor instability occurs when a less viscous fluid is injected into a more viscous one, usually in a Hele-Shaw cell. Here oil paint and mineral spirits were painted onto flat surfaces that were pressed together before being pulled apart. The result is viscous fingering of the fluids. #

Viscous Fingers
This photo shows the Saffman-Taylor instability in a Hele-Shaw cell. Here a viscous fluid has been placed between two glass plates and a second less viscous fluid inserted, resulting in a finger-like instability as the less viscous fluid displaces the more viscous one. This is an effect that can be easily explored at home using common liquids like glycerin, water, dish soap, or laundry detergent. #
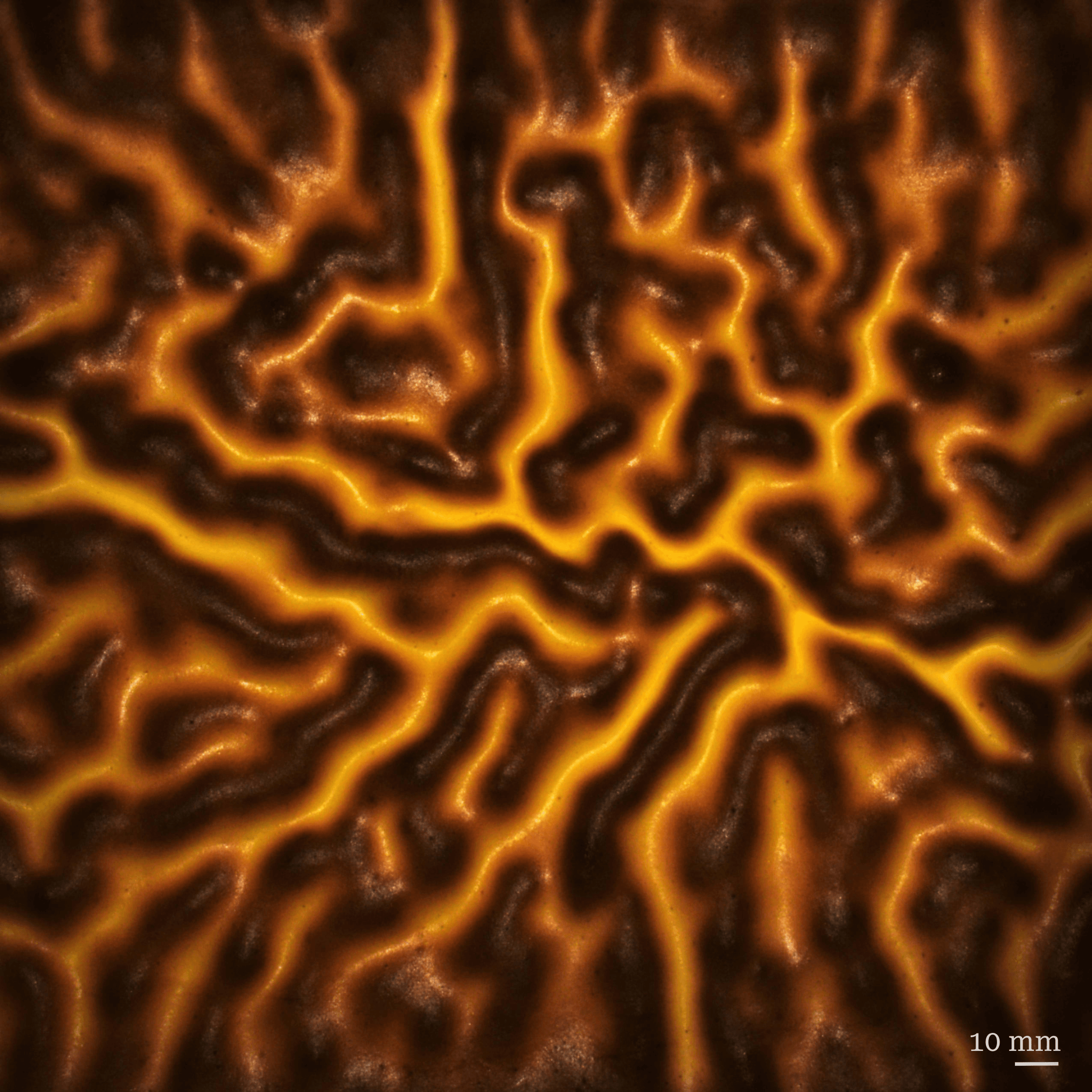
Setting the Stripes on a Tiger (Cake)
A tiger skin cake forms a distinctive pattern of light and dark patches as it bakes. Its current popularity seems to have expanded outward from China; I found a lot of Swiss-roll-style recipes that use it as an outer wrapper. Here, researchers look at how the wrinkled surface forms. The viscous batter quickly forms a solid skin on its surface, and, as the cake grows, the skin is forced to bend and wrinkle to accommodate the growth. Interestingly, the length-scale of the wrinkling pattern depends on the batter’s depth. For larger stripes, use a thicker layer of batter! (Image credit: K. Koutova et al.)

Research poster showing the wrinkling pattern formed on a tiger skin cake. 
Fluids Can Fracture
Fracture is a sudden, brittle breaking-apart that we generally associate with solid materials that get stressed too far. Some viscoelastic, non-Newtonian fluids have been known to fracture, but that was generally thought to be unusual. But a recent study turns that idea on its head, revealing that even simple, albeit highly viscous, liquids can fracture.

A viscous hydrocarbon fluid gets stretched at 100 mm/s, drawing it into a thinning shape. When you stretch a liquid, the general expectation is what you see above: the liquid gets drawn into an ever thinner shape. But researchers found that–when stretched quickly–that same simple hydrocarbon liquid cracked open:

A viscous hydrocarbon fluid gets stretched at 300 mm/s, causing it to fracture like a solid. There’s even an audible snap, which you can hear in the video below. The results were so surprising that they repeated the experiment several times and with different viscous (but Newtonian) liquids. The results held. When the liquids were pulled to a critical stress, they audibly snapped and fractured like a solid.
The next question, of course, is why this happens. The authors suspect (but have yet to show) that cavitation may be at play in the initiation of the crack that separates the liquid in two. (Image, video, and research credit: T. Lima et al.; via Gizmodo)

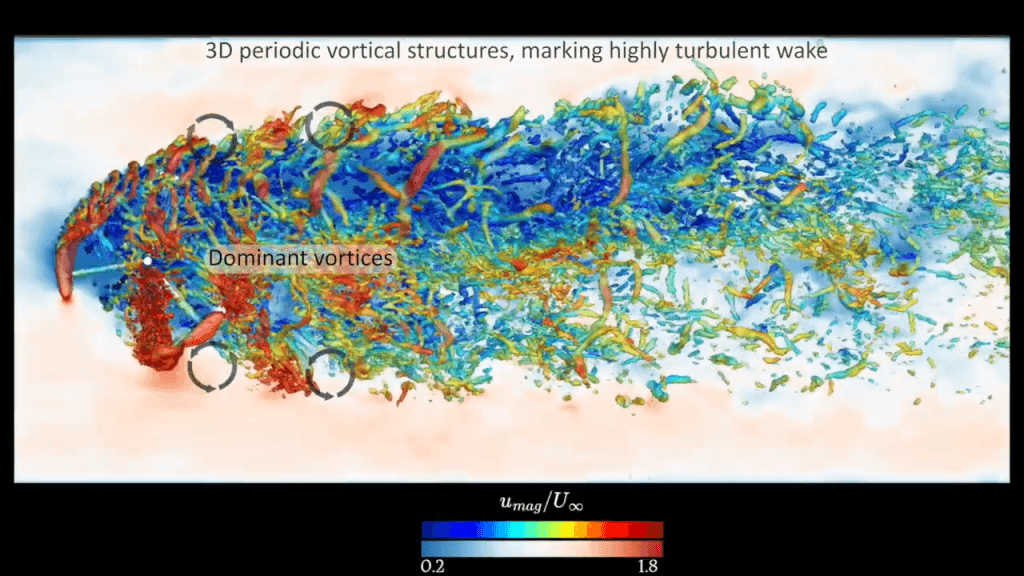
Understanding Fish and Turbines
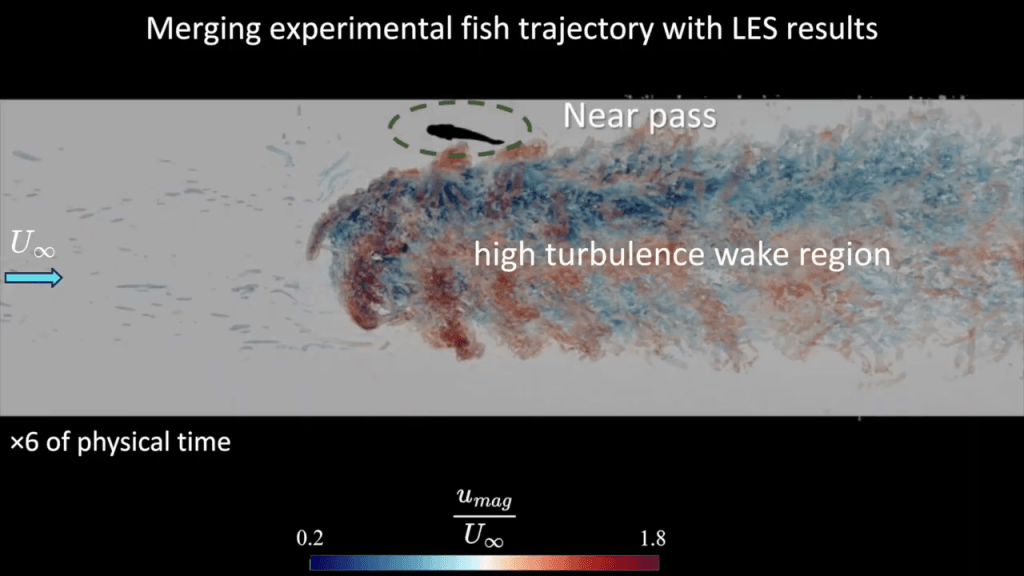
Fish detect turbulence in the water around them; among other things, this helps them avoid colliding with objects. Here, researchers are looking to understand how fish interact with underwater turbines. Experiments give them a set of trajectories that actual fish follow when dealing with the experimental turbine. But to understand what the fish is detecting, the researchers build a digital facsimile of the turbine and use Large Eddy Simulation (LES) to calculate the turbine’s wake.
By overlaying the fish trajectories onto the simulated flow structures, they can better understand what flows the fish is and is not comfortable with. That knowledge helps engineers design turbines with smaller ecological impact. (Video and image credit: H. Seyedzadeh et al.)

Turbulence and Bioluminescence
If you’ve ever seen crashing waves glowing blue, you’ve been treated to bioluminescence. Although many creatures can bioluminesce, tiny dinoflagellates–a type of marine phytoplankton–are one of the easiest to spot. These microscopic organisms create a flash of light in response to viscous stresses. Their response to flow-induced stresses is so robust that they can be used to visualize stress fields.
In a new study, researchers explored how turbulence affects the dinoflagellate’s luminescence. They mathematically modeled the dinoflagellate as an elastic dumbbell that emitted light based on its extent and rate of deformation. Then they explored how this model dinoflagellate behaved in different types of turbulent flows. They found that the fluctuations and intermittency of turbulent flows both encouraged the radiant displays. (Image credit: T. McKinnon; research credit: P. Kumar and J. Picardo)

Making Bubbles in Magma
When bubbles form in magma deep below the earth, volcanic eruptions follow. Scientists believe this happens when decompression of the magma allows volatile compounds to come out of solution and form bubbles–just as opening a bottle of seltzer allows carbon dioxide to bubble out. But a new study indicates that decompression may not be the only source of bubbles.

The team found that supersaturated fluids can nucleate bubbles when they’re sheared–even without decompression. They demonstrated this in the lab, not with magma but with a low-temperature magma analog, seen above. The more saturated with volatiles the fluid is, the less shear is needed to trigger bubbles.
Viscous shear is everywhere for magma, so this bubble formation mechanism is likely common. Better understanding how and when bubbles form in magma directly affects predictions for eruptions–especially for determining whether they’re likely to be explosive or effusive. (Image credit: volcano – A. Bonnerdeaux, experiment – O. Roche et al.; research credit: O. Roche et al.; via Physics World)