Using electromagnetism, researchers are bending and shaping soft liquid wires even against gravity. The team used galinstan — an alloy of gallium, indium, and tin that remains liquid at room temperature. On its own, galinstan has a high surface tension and forms droplets. But with a voltage applied, that surface tension is suppressed, making the liquid form a long, thin, still-liquid wire. Adding a magnetic field allowed the researchers to manipulate the falling stream of liquid, even levitating loops of the metal against the force of gravity! (Image, video, and research credit: Y. He et al.; via Cosmos; submitted by Kam-Yung Soh)
Search results for: “droplet”
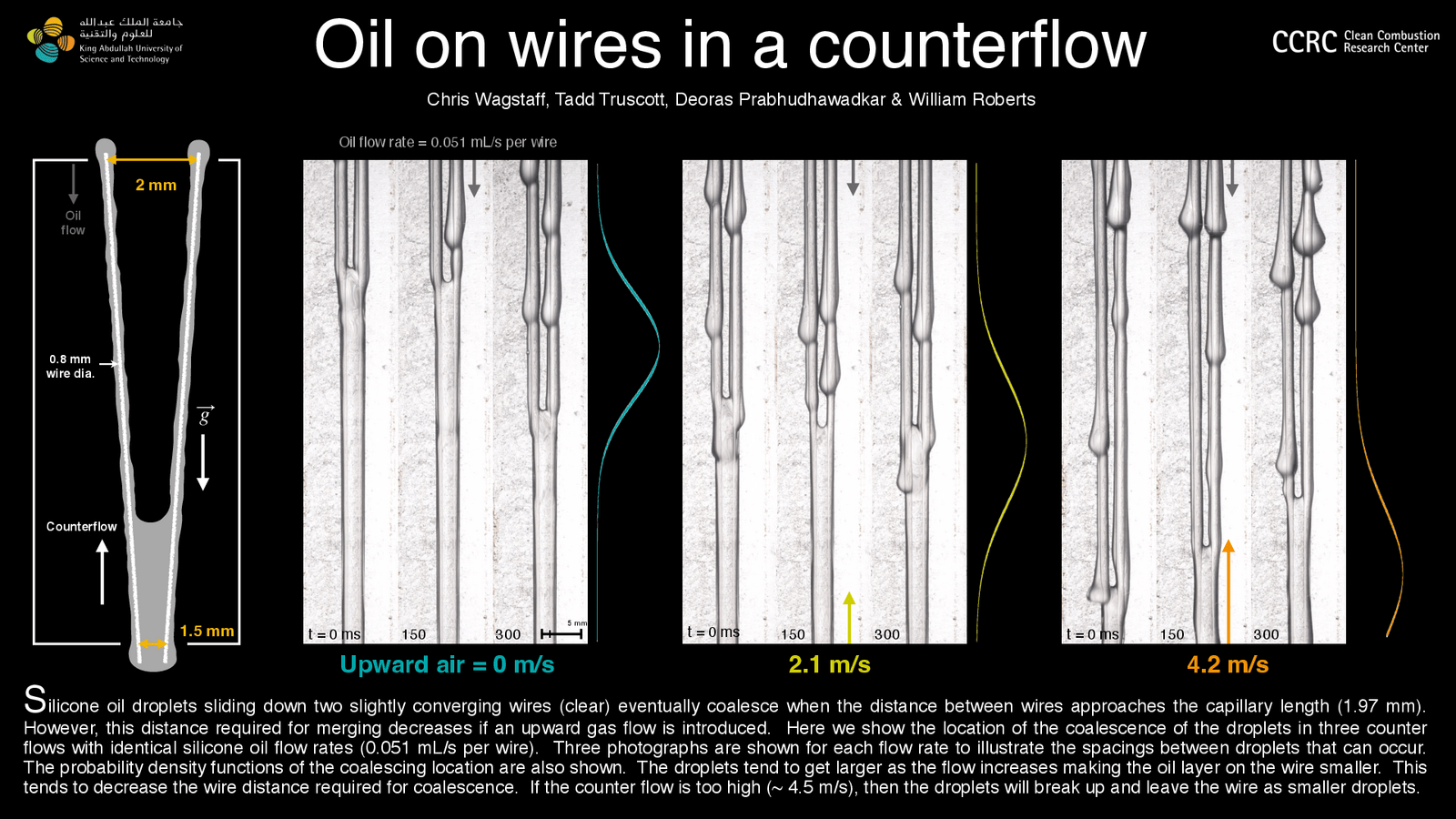
Merging Along Wires
As oil slides down two slowly converging wires, the droplets will merge into a sheet that stretches between both wires. When this happens can vary somewhat but occurs somewhere around the liquid’s capillary length.
In the poster above, the leftmost image (not the illustration) shows three possible merger points. To the right of the image, is a teal curve; this is a probability density function. Essentially, this curve shows where the merger is most likely to occur. The peak of the curve corresponds to the most probable point of merger.
The following two composite images show the same system — same oil flow rate, same wire spacing — with gas blowing upward along the wires. As the gas’s flow rate increase, the oil drops get larger, making the oil films thinner. The result? The wires have to get closer to one another before the oil merges. That’s reflected in the yellow and orange probability density functions, which have peaks further along the wires than the no-gas-flow case. (Image credit: C. Wagstaff et al.)

Acidic Aerosols
As ocean waves crash, they generate aerosols — tiny liquid and solid particulates — that interact with the atmosphere. Curious about the chemistry of these tiny drops, researchers set out to measure their acidity. That’s easier said than done. Over time, aerosol droplets acidify as they interact with acidic gases in the atmosphere and capturing fresh aerosols in the field is next to impossible.
To tackle these challenges, researchers instead moved the aerosols to the laboratory, filling a wave channel with seawater and agitating it to generate aerosols they could then measure. They found that the smallest aerosols become a million times more acidic than the bulk ocean in only two minutes! Find out more about their experiment and its implications over at Physics Today. (Image credit: E. Jepsen; research credit: K. Angle et al.)

Self-Stopping Leaks
A leak can actually stop itself, as shown in this video. To demonstrate, the team used a tube pierced with a small hole. When filled, water initially shoots out the hole in a jet. The pressure driving the jet comes from the weight of the fluid sitting above the hole. As the water level drops, the pressure drops, causing the jet to sag and eventually form a rivulet that wets the side of the tube. As the water level and driving pressure continue to fall, the rivulet breaks up into discrete droplets, whose exact behavior depends on how hydrophobic the tube is. Eventually, a final droplet forms a cap over the hole and the leak stops. At this point, the flow’s driving pressure is smaller than the pressure formed by the curvature of the capping droplet. (Image and video credit: C. Tally et al.)

Dripping Impact
How does water drip, drip, dripping onto stones erode a crater? Water is so much more deformable that it seems impossible for it to wear harder materials away, even over thousands of impacts. To investigate this, a team of researchers developed a new measurement technique: high-speed stress microscopy. In the process, they found that water owes its incredible erosive power to three factors: 1) The drop’s impact creates surface shock waves along the material, which helps increase erosive power; 2) After the shock wave passes, a decompression wave in the material helps loosen surface matter; and 3) The spreading drop sends a non-uniform wave of stress across the material that simultaneously presses and scrubs at the surface. Together, these factors enable simple, repetitive droplet impacts to wear away at hard surfaces. (Image credit: cottonbro; research credit: T. Sun et al.; via Cosmos; submitted by Kam-Yung Soh)

Coalescence Symmetry
When droplets coalesce, they perform a wiggly dance, gyrating as the capillary waves on their surface interfere. When the droplets have matching surface tensions, like the two water droplets in the animation on the lower left, the coalescence dance is symmetric. But for differing droplets, like the water and ethanol droplets merging on the lower right, coalescence is decidedly asymmetric.

Two water droplets merge symmetrically. 
A water droplet and an ethanol droplet merge asymmetrically. The asymmetry arises from the droplets’ different surface tensions. The size and speed of the capillary waves that form on a droplet depend on surface tension, so droplets of different liquids have inherently different capillary waves. During merger, the interference of these capillary waves causes the asymmetry we see. (Image credit: top – enfantnocta, coalescence – M. Hack et al.; research credit: M. Hack et al.)

Raindrops on the Windshield
When I was a child, I was fascinated by the raindrops that shimmied along the windshield of our car. Some would slide up the glass. Some would run down. And some just seemed to wiggle in place, until the car’s speed changed. As common as this sight is, the physics of these droplets is quite complicated and not completely understood.
Each droplet has a host of forces on it: gravity flattening it or pulling it down an incline; a drag force from the wind flowing over it; and friction between the drop and the surface trying to pin it in place. Recently, scientists have developed a new mathematical model that captures some of the behaviors behind these drops. The work describes the wind speed necessary to move a drop of a given size sitting on a flat surface. The authors also explored how that critical wind speed changes when a drop sits on a tilted surface aligned or against the wind. (Image credit: P. Gupta; research credit: A. Hooshanginejad and S. Lee; via Science News; submitted by Kam-Yung Soh)

Within the Bubble’s Pop
To our eyes, a soap bubble appears to pop instantly, but when observed in high-speed video, the process is far more complex. In this video, the Slow Mo Guys pop human-sized bubbles, giving us an opportunity to appreciate the rupture process at speeds up to 50,000 frames per second.
Once the rupture starts, the hole spreads very symmetrically. But as the hole grows, the remaining soap film starts distorting. As Gav and Dan observe, the far side of the bubble actually wrinkles up before the rupture front arrives and tears the remaining fluid into droplets! (Image and video credit: The Slow Mo Guys)

Mixing the Immiscible
Immiscible liquids — like oil and water — do not combine easily. Typically, with enough effort, you can create an emulsion — a mixture formed from droplets of one liquid suspended in the other — like the one above. But a team of researchers have taken mixing immiscible liquids to a new level using their Vortex Fluid Device (VFD).
Longtime readers may remember the group from their Ig-Nobel-winning demonstration of unboiling an egg, but this time the team is used the VFD to mix and de-mix immiscible liquids. As shown in the video below, the VFD is essentially a fast-spinning tube tilted at a 45-degree angle. As it spins, the liquids inside are forced into thin films with very high shear rates — high enough that immiscible liquids like water and toluene are forced together without forming an emulsion. Essentially, the mechanical forces mixing the liquids are strong enough to overcome the chemistry that typically keeps them apart.
Impressively, the device manages this without using harsh surfactants or catalysts that other methods rely on. As a result, the technique offers a greener method for mixing chemicals for pharmaceuticals, cosmetics, food processing, and more. (Image credit: pisauikan; research credit: M. Jellicoe et al.; video credit: Flinders University; submitted by Marc A.)

Parametric Resonance
At first glance, Steve Mould’s video on parametric resonance has nothing whatsoever to do with fluid dynamics. He uses a pendulum suspended on a spring to demonstrate how driving a system at a frequency that’s a multiple of the system’s natural frequency can add energy through resonance. Although his examples don’t use fluids, this phenomenon happens there, too, especially in vibrated fluid systems. Take, for example, this droplet bouncing on a vibrating pool. Depending on the amplitude of the vibrations driving the system, the droplet may bounce in time with the vibration, in time with the waves, or at a frequency twice that of the vibration. (Image and video credit: S. Mould)

By pulling on the string each time the mass swings through its lowest point (i.e., twice per swing cycle), Steve adds energy to the system, which is reflected in the increasing amplitude of the pendulum’s swing. This is an example of parametric resonance.









