Over the past few years, we’ve seen lots of droplets bouncing and walking on waves. But today’s example is a little different. In this set-up, the wave is a large standing wave that sloshes from side-to-side in a narrow container. As it does, the wave catches and tosses a large ~3mm water droplet. The system is surprisingly stable, with this game of catch lasting for tens of thousands of cycles and up to 90 minutes before the droplet coalesces. The researchers found that, if the droplet tries to wander from its spot, the oscillating surface wave corrects it, guiding the droplet back to the optimal position. (Image and research credit: C. Sandivari et al.; via APS Physics; submitted by Kam-Yung Soh)
Search results for: “droplet”

Rocket-Like Supercooled Drops
Many droplets can self-propel, often through the Leidenfrost effect and evaporation. But now researchers have observed freezing droplets that self-propel, too. The discovery came when observing the freezing of supercooled water drops inside a vacuum chamber. The researchers kept losing track of drops that seemingly disappeared. Upon closer inspection, though, they found that the drops weren’t shattering; they were flying away as they froze.
Inside a drop, freezing starts at a point, the nucleation point, and spreads from there. But the nucleation point isn’t always at the center of the drop. This asymmetry, the researchers found, is at the heart of the drop’s propulsion. When ice nucleates, the phase change releases heat that increases the drop’s evaporation rate, which can impart momentum to the drop. For an off-center nucleation, that momentum is enough to send the drop shooting off at nearly 1 meter per second. (Image credit: SpaceX; research credit: C. Stan et al.; via APS Physics)

How Large Particles Get in Sea Spray
When bubbles burst at the ocean’s surface, they eject droplets that can carry high concentrations of contaminants like pollutants, viruses, and microplastics. Previous theories posited that only particles smaller than the microlayer surrounding the bubble could make their way into these drops, but new work shows otherwise.
As bubbles rise to the surface, they carry particles on their surface, collecting them to a concentration that’s even higher than the surrounding seawater. But which particles make it into the air depend on the details of what happens when the bubble pops. Previously, researchers assumed that the thin microlayer of fluid surrounding the bubble was uniform, but that turns out not to be the case. As the bubble pops, some regions of the microlayer stretch and thin, while others grow thicker. The thicker the microlayer, the larger the particles it can pull along. In their single-bubble experiments, the researchers found that 15- and 30-micrometer plastic beads — representing oceanic microplastics — appeared in high concentrations in ejected droplets.

This animated simulation shows how fluid along the edge of a bubble makes its way into ejected droplets. Green particles indicate fluid from the left half of the bubble; blue shows fluid from the right side. Environmental scientists are keen to understand these mechanisms because they link our oceans and atmosphere, potentially affecting rainfall, pollution spread, and epidemiology. (Image, video, and research credit: L. Dubitsky et al.; via APS Physics)

Leidenfrost Explosion
When a water drop hits a surface that’s much hotter than its boiling point, part of it will vaporize immediately. Depending on the temperature, this Leidenfrost effect can be a relatively gentle process — or not. Here, the surface is so hot that the entire drop is boiling before it’s even finished spreading from impact. The vapor in contact with the surface is trying to escape, bubbling up so violently that it rips the original droplet into a spray of tiny droplets. (Video and image credit: L. Gledhill)

The Optical Atom
Researchers applied a quantum mechanical technique to study an evaporating drop in extreme detail. The team trapped a spherical water drop and collected the light scattered off it as it evaporated. Using an analytic technique originally developed for an atom, they were able to study changes in the drop down to the nanometric level without relying on numerical simulations to interpret the results. The authors suggest that their method is well-suited to studying the concentration of chemical or biological contaminants on the surface of a drop as it evaporates. (Image credit: droplet – Z. Kaiyv, Fano combs – J. Marmolejo et al.; research credit: J. Marmolejo et al.; via APS Physics)
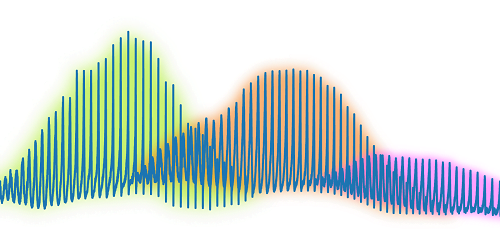
Illustration of the Fano combs seen by analyzing light scattered from an evaporating drop. 
Paint Ejection
Shaking paint on a speaker cone and filming it in high speed is an oldie but a goodie. Here, artist Linden Gledhill films paint ejection at 10,000 frames per second, giving us a glorious view of the process. As the paint flies upward, accelerated by the speaker, it stretches into long ligaments. As the ligaments thin, surface tension concentrates the paint into droplets, connected together by thinning strands. When those strands break, they snap back toward the remaining paint, imprinting swirling threads of different colors, thanks to their momentum. Eventually, surface tension wins the tug-of-war and transforms all the paint into droplets. (Video and image credit: L. Gledhill)

Hollow Drops
When a partially-air-filled drop hits a surface, it splashes and rebounds in a complex fashion. This video breaks down the physics of the process. Upon impact, a lamella spreads, eventually becoming wavy and unstable along its rim. At the same time, a counterjet forms, growing until it pierces the remaining bubble of the drop. The jet continues to stretch upward due to its momentum, pinching off and forming wobbly satellite drops that finally fall back to the surface. (Image and video credit: D. Naidu and S. Dash)

Toilet Plumes
Toilet flushes are gross. We’ve seen it before, though not in the same detail as this study. Here, researchers illuminate the spray from the flush of a typical commercial toilet, like those found in many public restrooms. They found that flushing generates a plume of droplets that reaches 1.5 meters in under 8 seconds, producing many thousands of droplets across a range of sizes.
The experiments were conducted in a ventilated lab space, and the flushes involved only clean water — no fecal matter or toilet paper — so they don’t perfectly mimic the confines of a public toilet stall. But the implications are still pretty gross. Without a lid to contain the flush’s spray, these energetic toilets are spraying droplets capable of carrying COVID, influenza, and other nastiness all over our bathrooms. (Image and research credit: J. Crimaldi et al.; via Gizmodo)

To Fizz or Not to Fizz
Place a drop of carbonated water on a superhydrophobic surface and it will slide almost frictionlessly, much the way Leidenfrost drops do. The drop behaves this way thanks to the self-produced layer of carbon dioxide vapor that it levitates on. As the gas escapes, the drop eventually settles back into contact with its surface. But until then, its levitation makes for some fun.

On the treated half of the glass (left), bubbles form a continuous film against the glass. On the untreated side (right), bubbles nucleate, grow, and rise as expected for a fizzy drink. Single droplets aren’t the only source of fun, however. In the images above, researchers coated the left half of a wine glass with a superhydrophobic treatment, while leaving the right half of the glass untouched. Once (dyed) carbonated water is poured into the glass, we see a bizarre dichotomy. In the right, untreated half of the glass, carbon dioxide bubbles nucleate, grow, and rise through the glass. But on the left side, the liquid appears still and bubble-less. In fact, the carbon dioxide gas on the left side is forming a continuous bubble film by the surface of the glass! (Image, video, and research credit: P. Bourrianne et al., see also)

Soapy Solutions
When a drop of soap falls into a pool of water, its surface-loving molecules spread out on the water’s surface. Exactly how the soap spreads depends on the local concentration of its surfactant molecules, which create areas with different surface tensions that cause flow. All in all, it’s a tough process to predict because it varies in time at every point on the pool. But a recent paper offers a new class of exact solutions for the problem.
The paper considers a surfactant-laden droplet spreading over a (relatively speaking) deep pool. Other researchers showed recently that this situation can be described with a complex version of the Burgers’s equation, which was originally developed to describe turbulent flows. The authors solved the equation for a variety of initial conditions and found that the time-dependent spread of the surfactants was sensitive to the initial surface distribution. The higher the initial surface concentration, the faster the surfactants spread. (Image credit: T. Despeyroux; research credit: T. Bickel and F. Detcheverry; via APS Physics; submitted by Kam-Yung Soh)









